Europe Magnetic Resonance Imaging Mri Devices Market
Market Size in USD Billion
CAGR :
% 
 USD
2.67 Billion
USD
3.89 Billion
2024
2032
USD
2.67 Billion
USD
3.89 Billion
2024
2032
| 2025 –2032 | |
| USD 2.67 Billion | |
| USD 3.89 Billion | |
|
|
|
|
Magnetic Resonance Imaging (MRI) Devices Market Size
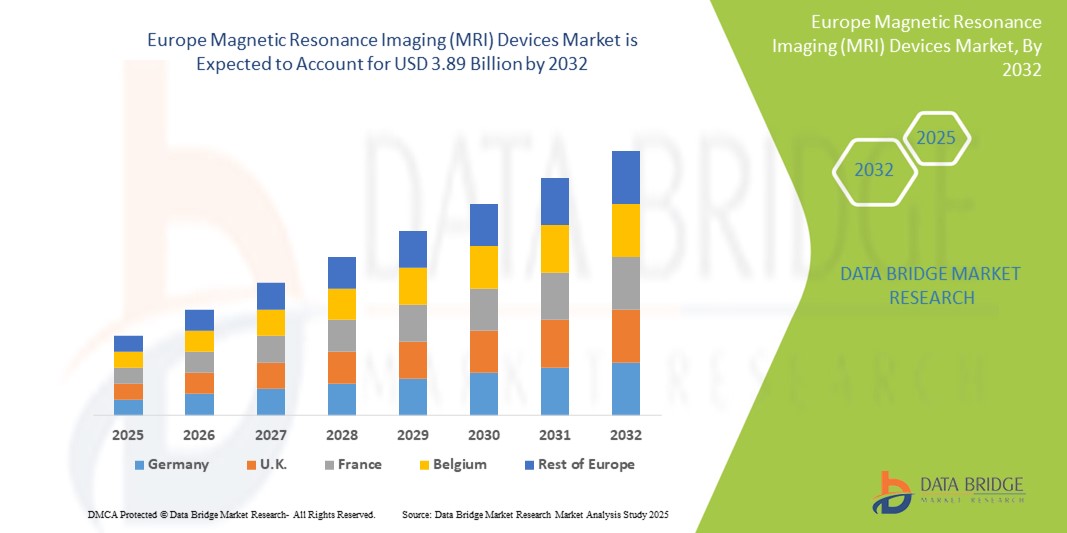
- The Europe Magnetic Resonance Imaging (MRI) Devices market size was valued at USD 2.67 billion in 2024 and is expected to reach USD 3.89 billion by 2032, at a CAGR of 7.20% during the forecast period
- The market's growth is largely fueled by the increasing aging population, the rising prevalence of chronic diseases, and the growing emphasis on early diagnosis.
- Furthermore, the introduction of hybrid MRI systems and the availability of universal health coverage in Europe are encouraging people to undergo tests or treatments at regular intervals, significantly boosting the industry's growth.
Magnetic Resonance Imaging (MRI) Devices Market Analysis
- The market's growth is largely fueled by the increasing aging population, the rising prevalence of chronic diseases, and the growing emphasis on early diagnosis.
- Furthermore, the introduction of hybrid MRI systems and the availability of universal health coverage in Europe are encouraging people to undergo tests or treatments at regular intervals, significantly boosting the industry's growth.
- Germany dominates the Magnetic Resonance Imaging (MRI) Devices Market with the largest revenue share of 54.5% in 2024, due to The market's growth is largely fueled by the increasing aging population, the rising prevalence of chronic diseases, and the growing emphasis on early diagnosis.
- The Conventional segment is expected to dominate the Magnetic Resonance Imaging (MRI) Devices Market with a market share of 59.99% in 2024, due to the increasing use of MRI machines in various fields such as gynecology, urology, dentistry, cardiology, musculoskeletal, cancer, pelvic and abdominal. The country's diagnostic imaging services, including MRI scans, are also cost-effective compared to other European countries like the UK.
Report Scope and Magnetic Resonance Imaging (MRI) Devices Market Segmentation
|
Attributes |
Magnetic Resonance Imaging (MRI) Devices Key Market Insights |
|
Segments Covered |
|
|
Countries Covered |
Europe
|
|
Key Market Players |
|
|
Market Opportunities |
|
|
Value Added Data Infosets |
In addition to the insights on market scenarios such as market value, growth rate, segmentation, geographical coverage, and major players, the market reports curated by the Data Bridge Market Research also include in-depth expert analysis, pricing analysis, brand share analysis, consumer survey, demography analysis, supply chain analysis, value chain analysis, raw material/consumables overview, vendor selection criteria, PESTLE Analysis, Porter Analysis, and regulatory framework. |
Magnetic Resonance Imaging (MRI) Devices Market Trends
“Advancements in Digital and AI-Powered MRI Systems”
- A significant trend in the Europe Magnetic Resonance Resonance Imaging (MRI) devices market is the rapid advancement and adoption of digital and AI-powered MRI systems. These innovations are revolutionizing the industry by improving image quality, scanning speed, and diagnostic accuracy.
- For instance, in February 2024, Fujifilm Healthcare Europe launched its new 1.5 Tesla superconductive MRI scanner, Echelon Synergy, featuring AI-powered deep learning for enhanced image sharpness and scanning speed. Similarly, in November 2023, Royal Philips launched its inaugural mobile MRI system, BlueSeal MR Mobile, featuring helium-free operations, which prioritizes patient-centric MRI services and aims to extend accessible, sustainable, and helium-free MRI exams to diverse locations.
- The integration of AI in MRI systems facilitates automated image reconstruction, noise reduction, and intelligent workflow optimization, leading to more efficient examinations and better patient outcomes.
- Furthermore, remote scanning capabilities, as highlighted by Philips' Radiology Operations Command Center Console receiving its EU Medical Device Regulation (MDR) certificate in February 2025, are enabling expert radiologists to assist technologists on-site by remotely controlling scans, improving efficiency and access to specialized care across Europe.
Magnetic Resonance Imaging (MRI) Devices Market Dynamics
Driver
“Rising Prevalence of Chronic Diseases and Aging Population”
- The increasing prevalence of chronic diseases such as cancer, neurological disorders (e.g., dementia, Alzheimer's, Parkinson's), cardiovascular diseases, and musculoskeletal conditions, coupled with a rapidly aging population in Europe, is a major driver for the MRI devices market.
- For instance, as per Eurostats Statistics in February 2022, over one-fifth (20.8%) of European citizens were 65 years of age or older in 2021, and this proportion is projected to increase to 14.6% by 2100 for those aged 80 or older. This demographic shift significantly increases the demand for diagnostic imaging as older individuals are more susceptible to age-related health issues requiring MRI scans for accurate diagnosis and monitoring.
- MRI's ability to provide detailed soft tissue contrast makes it invaluable for the early detection and management of these complex conditions. Universal health coverage in many European countries also makes MRI scans more accessible and affordable, further encouraging their use.
- Furthermore, advancements in MRI technology, such as the introduction of hybrid systems and personalized care approaches, continue to expand its utility in diagnosing and managing a broader spectrum of chronic illnesses.
Restraint/Challenge
“High Cost of MRI Systems and Declining Helium Availability”
- The high initial acquisition and installation costs of MRI systems, coupled with ongoing maintenance expenses, pose a significant restraint on market growth, particularly for smaller healthcare facilities or those with limited budgets.
- For instance, advanced high-field MRI systems require substantial capital investment, making them less accessible in some regions or for certain healthcare providers. Moreover, the scarcity and fluctuating prices of helium, a critical cryogenic gas essential for cooling superconducting MRI magnets, present a significant challenge. This supply chain vulnerability can increase operating costs and potentially disrupt MRI services.
- While advancements in helium-free or low-helium MRI technologies are emerging to mitigate this issue, the transition can be costly and time-consuming.
- Additionally, stringent regulatory frameworks and the need for highly skilled personnel to operate and maintain MRI devices further contribute to the overall operational complexities and costs, challenging widespread adoption, especially in regions with limited healthcare resources.
Magnetic Resonance Imaging (MRI) Devices Market Scope
The market is segmented on the basis of type, field strength, architecture, modality, application, and end-user.
By Type
On the basis of type, the Europe magnetic resonance imaging (MRI) devices market is segmented into conventional and bio-based. The conventional segment is anticipated to hold the market share of 48.34% in 2025 driven by their increasing use in advanced research and specialized clinical applications, particularly in neurology and oncology.
The bio-based segment is anticipated to hold the market share of 38.45% in 2025 driven by the increasing demand for patient comfort, especially for claustrophobic, obese, geriatric, and pediatric patients.
By Field Strength
On the basis of field strength, the Europe magnetic resonance imaging (MRI) devices market is segmented into high-field magnetic resonance imaging (MRI) systems (1.5t to 3t), low-to-mid-field magnetic resonance imaging (MRI) systems (<1.5t), and very-high-field magnetic resonance imaging (MRI) systems (4t and above). The high-field MRI systems (1.5T to 3T) segment is estimated to hold a significant share due to their superior image quality and diagnostic capabilities, making them widely adopted for various clinical applications.
The very-high-field MRI systems (4T and above) segment is expected to witness substantial growth, driven by their increasing use in advanced research and specialized clinical applications, particularly in neurology and oncology.
By Architecture
On the basis of architecture, the Europe magnetic resonance imaging (MRI) devices market is segmented into closed magnetic resonance imaging (MRI), standard bore magnetic resonance imaging (MRI), wide-bore magnetic resonance imaging (MRI), and open magnetic resonance imaging (MRI). The closed MRI systems segment holds a significant share due to their widespread installation base and ability to produce high-quality images.
The open magnetic resonance imaging (MRI) segment is anticipated to grow with a high CAGR, driven by the increasing demand for patient comfort, especially for claustrophobic, obese, geriatric, and pediatric patients.
By Modality
On the basis of modality, the Europe magnetic resonance imaging (MRI) devices market is segmented into stationary system, portable/mobile system, and point-of-care (POC) system. The stationary system segment is expected to hold the largest share due to its established presence in hospitals and diagnostic centers for comprehensive imaging needs.
The portable/mobile system segment is anticipated to grow with a high CAGR, driven by the increasing need for flexible and accessible MRI services in various healthcare settings, including remote areas and emergency situations.
By Application
On the basis of application, the Europe magnetic resonance imaging (MRI) devices market is segmented into oncology, neurology, cardiology, gastroenterology, musculoskeletal, and other applications. The neurology segment is expected to dominate, owing to the rising prevalence of neurological disorders and the indispensable role of MRI in brain and spinal cord imaging.
The oncology segment is anticipated to grow with a high CAGR, driven by the increasing use of MRI for cancer detection, staging, and treatment monitoring.
By End-User
On the basis of end-user, the Europe magnetic resonance imaging (MRI) devices market is segmented into hospitals, imaging centers, ambulatory surgical centers, and other end users. The hospitals segment is expected to hold the largest market share, attributed to their extensive infrastructure, high patient volume, and the need for comprehensive diagnostic capabilities.
The imaging centers segment is anticipated to grow with a high CAGR, driven by their specialization in diagnostic services and increasing patient referrals for various imaging procedures.
Magnetic Resonance Imaging (MRI) Devices Market Regional Analysis
- Germany dominates the Magnetic Resonance Imaging (MRI) Devices market with the largest revenue share of 54.5% in 2024, due to its sophisticated healthcare system, strong emphasis on research and development activities, and a high volume of MRI procedures performed for various conditions.
- The country's extensive network of doctors and hospitals, even in remote areas, ensures widespread access to advanced medical imaging services.
France Magnetic Resonance Imaging (MRI) Devices Market Insight
The France Magnetic Resonance Imaging (MRI) Devices market is driven by significant healthcare expenditure and a focus on integrating advanced imaging technologies. The country's robust healthcare infrastructure and increasing awareness among the population regarding early disease diagnosis are key factors fueling market growth.
U.K. Magnetic Resonance Imaging (MRI) Devices Market Insight
The U.K. Magnetic Resonance Imaging (MRI) Devices market is driven by the presence of a sophisticated healthcare system, such as the National Health Services (NHS), coupled with a growing geriatric population and increasing prevalence of chronic diseases. The rising demand for early and accurate diagnosis contributes to market growth in the U.K.
Magnetic Resonance Imaging (MRI) Devices Market Share
The Magnetic Resonance Imaging (MRI) Devices industry is primarily led by well-established companies, including:
- BD (U.S.)
- bioMérieux SA (France)
- Thermo Fisher Scientific Inc. (U.S.)
- Bruker Corporation (U.S.)
- Danaher Corporation (U.S.)
- Roche Diagnostics (Switzerland)
- T2 Biosystems (U.S.)
- Luminex Corporation (U.S.)
- Siemens Healthineers AG (Germany)
- Abbott Laboratories (U.S.)
- Koninklijke Philips N.V. (Netherlands)
- GE Healthcare (U.S.)
- FUJIFILM Corporation (Japan)
- Canon Medical Systems Corporation (Japan)
Latest Developments in Europe Magnetic Resonance Imaging (MRI) Devices Market
- In November 2023 ,Koninklijke Philips N.V. expanded the partnership agreement with SyntheticMR, which aims to introduce SyMRI 3D. The development would further expand the company’s imaging product offering.
- In November 2023, GE Healthcare launched a 1.5T wide bore Magnetic Resonance Imaging (MRI) system, SIGNA Champion. The system is designed to improve the standard of care for patients by integrating advanced AI and innovative features that enable faster and more precise MRI scans.
- In August 2023, FUJIFILM Corporation launched a new 1.5Tesla MRI system, ECHELON Synergy, integrated with deep learning reconstruction technology. This advanced technology will enhance the image quality and patient satisfaction.
- In April 2023, Canon Medical Systems Corporation launched an AI-automated MRI planning solution to accelerate MRI planning time. The system utilizes machine learning and deep learning to improve diagnostic imaging of prostate, liver, and whole spine examinations.
SKU-
Get online access to the report on the World's First Market Intelligence Cloud
- Interactive Data Analysis Dashboard
- Company Analysis Dashboard for high growth potential opportunities
- Research Analyst Access for customization & queries
- Competitor Analysis with Interactive dashboard
- Latest News, Updates & Trend analysis
- Harness the Power of Benchmark Analysis for Comprehensive Competitor Tracking
Research Methodology
Data collection and base year analysis are done using data collection modules with large sample sizes. The stage includes obtaining market information or related data through various sources and strategies. It includes examining and planning all the data acquired from the past in advance. It likewise envelops the examination of information inconsistencies seen across different information sources. The market data is analysed and estimated using market statistical and coherent models. Also, market share analysis and key trend analysis are the major success factors in the market report. To know more, please request an analyst call or drop down your inquiry.
The key research methodology used by DBMR research team is data triangulation which involves data mining, analysis of the impact of data variables on the market and primary (industry expert) validation. Data models include Vendor Positioning Grid, Market Time Line Analysis, Market Overview and Guide, Company Positioning Grid, Patent Analysis, Pricing Analysis, Company Market Share Analysis, Standards of Measurement, Global versus Regional and Vendor Share Analysis. To know more about the research methodology, drop in an inquiry to speak to our industry experts.
Customization Available
Data Bridge Market Research is a leader in advanced formative research. We take pride in servicing our existing and new customers with data and analysis that match and suits their goal. The report can be customized to include price trend analysis of target brands understanding the market for additional countries (ask for the list of countries), clinical trial results data, literature review, refurbished market and product base analysis. Market analysis of target competitors can be analyzed from technology-based analysis to market portfolio strategies. We can add as many competitors that you require data about in the format and data style you are looking for. Our team of analysts can also provide you data in crude raw excel files pivot tables (Fact book) or can assist you in creating presentations from the data sets available in the report.