Europe Q Pcr And D Pcr Devices Market
Market Size in USD Billion
CAGR :
% 
 USD
9.30 Billion
USD
17.47 Billion
2024
2032
USD
9.30 Billion
USD
17.47 Billion
2024
2032
| 2025 –2032 | |
| USD 9.30 Billion | |
| USD 17.47 Billion | |
|
|
|
|
Q-PCR and D-PCR Devices Market Size
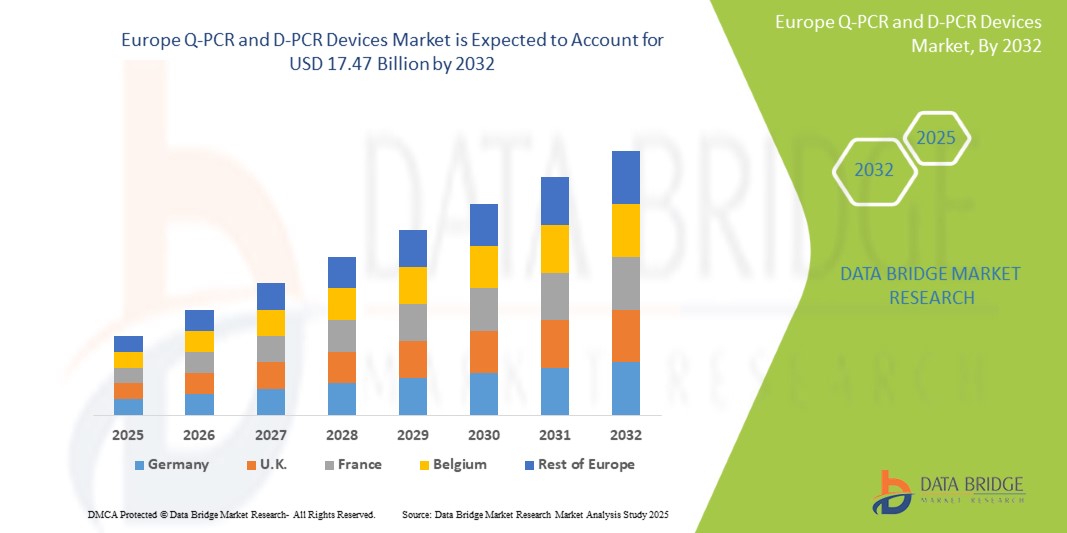
- The European Q-PCR and D-PCR Devices Market was valued at USD 9.3 billion in 2024 and is expected to reach USD 17.47 billion by 2032, at a CAGR of 8.1% during the forecast period.
- The drivers of the Q-PCR and D-PCR Devices Market include the growing demand for safe and effective diagnostic solutions, technological advancements in PCR devices, and the increasing prevalence of genetic disorders and infectious diseases, which require advanced diagnostic techniques.
Europe Q-PCR and D-PCR Devices Market Analysis
- Q-PCR and D-PCR Devices play a pivotal role in molecular biology and diagnostics by amplifying specific segments of DNA or RNA for detection, analysis, and research. These devices are essential in various applications such as genetic research, medical diagnostics, forensic analysis, and environmental monitoring.
- The demand for PCR devices in Europe is primarily driven by the rising prevalence of infectious diseases, advancements in molecular diagnostics, and increasing research in genetics and genomics. PCR technology is widely used in medical diagnostics, particularly for detecting genetic disorders, infectious diseases (e.g., COVID-19, HIV), and cancer.
- Europe is a leading region in the Europe PCR devices market, bolstered by strong healthcare infrastructure, significant investments in biotechnology and pharmaceutical research, and a high level of adoption of advanced diagnostic tools. The Germany, in particular, is a dominant player in the market, given its vast healthcare ecosystem, research funding, and increasing demand for diagnostic testing.
- The Europen PCR devices market is also influenced by regulatory support, including FDA approvals for novel PCR technologies, reimbursement policies, and growing research grants from both government and private sectors. Additionally, the increasing prevalence of chronic diseases and personalized medicine is accelerating the adoption of PCR devices in clinical diagnostics
Report Scope Q-PCR and D-PCR Devices Market Segmentation
|
Attributes |
Q-PCR and D-PCR Devices Key Market Insights |
|
Segments Covered |
|
|
Countries Covered |
|
|
Key Market Players |
|
|
Market Opportunities |
|
|
Value Added Data Infosets |
In addition to the insights on market scenarios such as market value, growth rate, segmentation, geographical coverage, and major players, the market reports curated by the Data Bridge Market Research also include import export analysis, production capacity overview, production consumption analysis, price trend analysis, climate change scenario, supply chain analysis, value chain analysis, raw material/consumables overview, vendor selection criteria, PESTLE Analysis, Porter Analysis, and regulatory framework. |
Q-PCR and D-PCR Devices Market Trends
“Advancements in PCR Technology and Growing Application Areas”
- Real-time PCR has become the gold standard in various diagnostic and research applications due to its ability to provide quantitative data. The growing use of real-time PCR in clinical diagnostics, especially for genetic testing and cancer detection, is significantly contributing to market growth.
- For instance, in 2024, the European Medicines Agency (EMA) approved a real-time PCR-based diagnostic developed by Qiagen for identifying EGFR mutations in non-small cell lung cancer, supporting targeted therapy decisions across EU countries.
- With the growing trend of personalized medicine, PCR devices are being used to tailor treatments based on an individual’s genetic makeup, particularly in oncology and genetic disorders.
Q-PCR and D-PCR Devices Market Dynamics
Driver
“High Adoption of Advanced Molecular Diagnostic Techniques”
- The Europe Q-PCR and D-PCR Devices Market is driven by the increasing reliance on molecular diagnostics for early and accurate disease detection, particularly in areas such as oncology, infectious diseases, and genetic disorders.
- Government-backed healthcare programs in the Germany and France, including CDC-supported disease surveillance initiatives and NIH-funded genomic research, are accelerating the adoption of PCR technologies in clinical and research settings.
- The presence of well-established diagnostic laboratories, growing integration of PCR in point-of-care testing, and rising investments in precision medicine are collectively supporting sustained market growth.
- Moreover, the post-COVID-19 era has reinforced the importance of PCR testing in healthcare infrastructure, with continued use in respiratory virus detection, including influenza and RSV.
For instance,
- According to the CDC, real-time PCR remains the gold standard for detecting various viral pathogens, contributing to its expanded use in routine diagnostic protocols.
- In February 2024, Thermo Fisher Scientific reported increased demand for its real-time PCR solutions driven by a rise in cancer screening initiatives and infectious disease monitoring in Europe
- This trend is further supported by growing public health awareness, advancements in automated PCR platforms, and government efforts to enhance laboratory testing capacities across the region.
Opportunity
“Integration of PCR Devices into Decentralized and Point-of-Care Testing Models”
- The increasing shift toward decentralized healthcare models, including clinics, urgent care centers, and home-based testing, is creating significant opportunities for portable and user-friendly PCR devices in Europe.
- Growing demand for rapid, accurate diagnostics in non-traditional settings such as pharmacies, workplaces, and remote areas is fueling interest in compact, automated PCR platforms that deliver real-time results.
For instance,
- In January 2024, according to a report by the U.S. Food and Drug Administration (FDA), there has been a surge in emergency use authorizations (EUAs) for point-of-care molecular diagnostic tools, including PCR-based assays, for respiratory infections and other emerging pathogens
- This trend is amplified by healthcare providers' need for faster clinical decision-making, particularly in infectious disease management, cancer screening, and genetic disorder detection—fostering growing investment in mobile and cloud-connected PCR solutions across Europe.
Restraint/Challenge
“High Equipment Costs and Complex Regulatory Approval Processes”
- Advanced PCR devices, especially those integrated with real-time capabilities and multiplex testing features, come with high procurement and maintenance costs. This poses a significant challenge for smaller laboratories, diagnostic clinics, and rural healthcare centers across Europe.
- The stringent and time-consuming regulatory requirements imposed by agencies such as the Germany and France create barriers for manufacturers aiming to launch or upgrade PCR technologies, delaying market entry and increasing development costs.
For instance,
- In October 2024, a report by the Medical Device Innovation Consortium (MDIC) highlighted that the cost of bringing a new molecular diagnostic device to market can exceed USD 100 million, with regulatory compliance accounting for a large share of this expenditure
- Consequently, these financial and procedural burdens can slow down innovation, limit accessibility in underserved areas, and create competitive pressure, especially for small and medium-sized enterprises operating in the Europe PCR devices market
Q-PCR and D-PCR Devices Market Scope
The market is segmented on the basis, product type, technology, application and distribution channel.
|
Segmentation |
Sub-Segmentation |
|
By Product Type |
|
|
By Technology |
|
|
By Application |
|
|
By End User
|
|
In 2025, the Real-Time PCR Devices is projected to dominate the market with a largest share in technology segment
The real-time PCR (qPCR) devices segment is expected to dominate the Europe Q-PCR and D-PCR Devices Market with 45.12% share in 2025, owing to its widespread use in diagnostic applications, including infectious disease detection, oncology, and genetic testing. The technology’s ability to deliver rapid, accurate, and quantifiable results makes it highly valuable in both clinical and research settings. Increasing adoption in personalized medicine, alongside strong investments in biotechnology and healthcare infrastructure across Germany and France[SS7] , further supports the segment’s leadership. Additionally, advancements in automation, user-friendly software, and integration with digital platforms are enhancing the usability and appeal of real-time PCR systems.
The Diagnostic Laboratories is expected to account for the largest share during the forecast period in end user market
In 2025, hospitals and diagnostic laboratories are anticipated to hold the largest share of the Europe PCR Devices Market by end user, with a 56.32% share in 2025 driven by the growing need for rapid and accurate diagnostic tools in managing infectious diseases, cancer, and genetic disorders. The high patient footfall, coupled with better reimbursement structures and access to advanced diagnostic technologies, enables these institutions to invest heavily in cutting-edge PCR systems. The post-pandemic emphasis on improving diagnostic capacities and the expansion of hospital laboratory services across Europe further bolster the dominance of this segment.
Q-PCR and D-PCR Devices Market Regional Analysis
“Germany is the Dominant Country in the Q-PCR and D-PCR Devices Market”
- Germany leads the Europe Q-PCR and D-PCR Devices market, with the United States accounting for the largest share due to its highly developed healthcare infrastructure, widespread use of molecular diagnostics, and substantial investments in life sciences research.
- The growing prevalence of infectious diseases, genetic disorders, and cancer is increasing the demand for accurate and rapid diagnostic tools, further driving PCR device adoption across hospitals, diagnostic labs, and research institutions in Germany[SS8] .
- The presence of key industry players such as Thermo Fisher Scientific, Bio-Rad Laboratories, and Agilent Technologies contributes to the region’s technological edge, offering advanced and user-friendly PCR systems.
- Favorable government funding, such as NIH research grants and public health initiatives targeting early disease detection, continues to strengthen the country’s leadership position in the market.
“U.K is Projected to Register the Highest Growth Rate”
- U.K is expected to witness the fastest growth in the Europe PCR Devices market, supported by its universal healthcare model and a strong national focus on improving disease surveillance and diagnostic capabilities.
- Strategic government investments in molecular diagnostics and research infrastructure—especially in response to public health challenges such as COVID-19 and antimicrobial resistance—are enhancing the adoption of PCR technologies.
- The expansion of genomics and personalized medicine programs, particularly through initiatives like Genome Germany, is boosting demand for real-time and digital PCR solutions.
- Increased collaborations between academic research centers and biotechnology firms, combined with growing awareness of early diagnosis and preventive care, are accelerating market growth across Canadian provinces
Q-PCR and D-PCR Devices Market Share
The market competitive landscape provides details by competitor. Details included are company overview, company financials, revenue generated, market potential, investment in research and development, new market initiatives, Europe presence, production sites and facilities, production capacities, company strengths and weaknesses, product launch, product width and breadth, application dominance. The above data points provided are only related to the companies' focus related to market.
The Major Market Leaders Operating in the Market Are:
- QIAGEN N.V. (Netherlands/Germany)
- bioMérieux S.A. (France)
- Roche Diagnostics (Switzerland)
- Oxford Nanopore Technologies (United Kingdom)
- Randox Laboratories (United Kingdom)
- Molzym GmbH & Co. KG (Germany)
- Genefirst Ltd. (United Kingdom)
- Altona Diagnostics GmbH (Germany)
- Eurofins Scientific (Luxembourg)
- Sysmex Inostics GmbH (Germany)
Latest Developments in Europe Q-PCR and D-PCR Devices Market
- In September 2023, Thermo Fisher Scientific introduced an advanced line of real-time PCR instruments, offering enhanced speed and high-throughput performance. Designed for both clinical diagnostics and research, the systems feature improved sensitivity, faster thermal cycling, and intuitive software, aiming to streamline workflows in laboratories handling large sample volumes.
- In January 2024, Bio-Rad Laboratories unveiled a new multiplex PCR system focused on improving diagnostic accuracy for infectious diseases. This system enables simultaneous detection of multiple pathogens in a single run, reducing turnaround time and reagent use. The innovation supports labs facing high testing demands and contributes to efficient disease surveillance
- In March 2024, Qiagen launched a digital PCR system tailored for precision oncology and rare mutation detection. The platform offers high sensitivity and quantification capabilities, catering to growing demand for personalized medicine. Its compact design and automated workflows are ideal for clinical labs and translational research centers.
- In February 2024, Agilent Technologies announced a new AI-powered software tool for analyzing PCR data, designed to improve result accuracy and reduce manual interpretation. The tool integrates with Agilent’s existing PCR platforms, enabling real-time analytics and predictive modeling. This innovation enhances efficiency in clinical diagnostics and life sciences research environments.
SKU-
Get online access to the report on the World's First Market Intelligence Cloud
- Interactive Data Analysis Dashboard
- Company Analysis Dashboard for high growth potential opportunities
- Research Analyst Access for customization & queries
- Competitor Analysis with Interactive dashboard
- Latest News, Updates & Trend analysis
- Harness the Power of Benchmark Analysis for Comprehensive Competitor Tracking
Research Methodology
Data collection and base year analysis are done using data collection modules with large sample sizes. The stage includes obtaining market information or related data through various sources and strategies. It includes examining and planning all the data acquired from the past in advance. It likewise envelops the examination of information inconsistencies seen across different information sources. The market data is analysed and estimated using market statistical and coherent models. Also, market share analysis and key trend analysis are the major success factors in the market report. To know more, please request an analyst call or drop down your inquiry.
The key research methodology used by DBMR research team is data triangulation which involves data mining, analysis of the impact of data variables on the market and primary (industry expert) validation. Data models include Vendor Positioning Grid, Market Time Line Analysis, Market Overview and Guide, Company Positioning Grid, Patent Analysis, Pricing Analysis, Company Market Share Analysis, Standards of Measurement, Global versus Regional and Vendor Share Analysis. To know more about the research methodology, drop in an inquiry to speak to our industry experts.
Customization Available
Data Bridge Market Research is a leader in advanced formative research. We take pride in servicing our existing and new customers with data and analysis that match and suits their goal. The report can be customized to include price trend analysis of target brands understanding the market for additional countries (ask for the list of countries), clinical trial results data, literature review, refurbished market and product base analysis. Market analysis of target competitors can be analyzed from technology-based analysis to market portfolio strategies. We can add as many competitors that you require data about in the format and data style you are looking for. Our team of analysts can also provide you data in crude raw excel files pivot tables (Fact book) or can assist you in creating presentations from the data sets available in the report.