Europe Trauma Devices Market
Market Size in USD Billion
CAGR :
% 
 USD
13.66 Billion
USD
24.54 Billion
2022
2030
USD
13.66 Billion
USD
24.54 Billion
2022
2030
| 2023 –2030 | |
| USD 13.66 Billion | |
| USD 24.54 Billion | |
|
|
|
Trauma Devices Market Analysis and Size
Injuries are a serious health issue everywhere in the world. There are 16000 injuries-related deaths per day, many of which have long-term effects. The market for trauma devices worldwide is boosted by this circumstance. The availability of more Essential Trauma Care (EsTC) facilities and a range of injury prevention initiatives are encouraging low-cost advancements in facility-based trauma care. The establishment of reasonable and attainable criteria for the treatment of injuries creates the global opportunity for the trauma devices market. The World Health Organization (WHO) offers emergency and trauma care training courses that enhance the number of specialists engaged in the management and fixation of trauma devices.
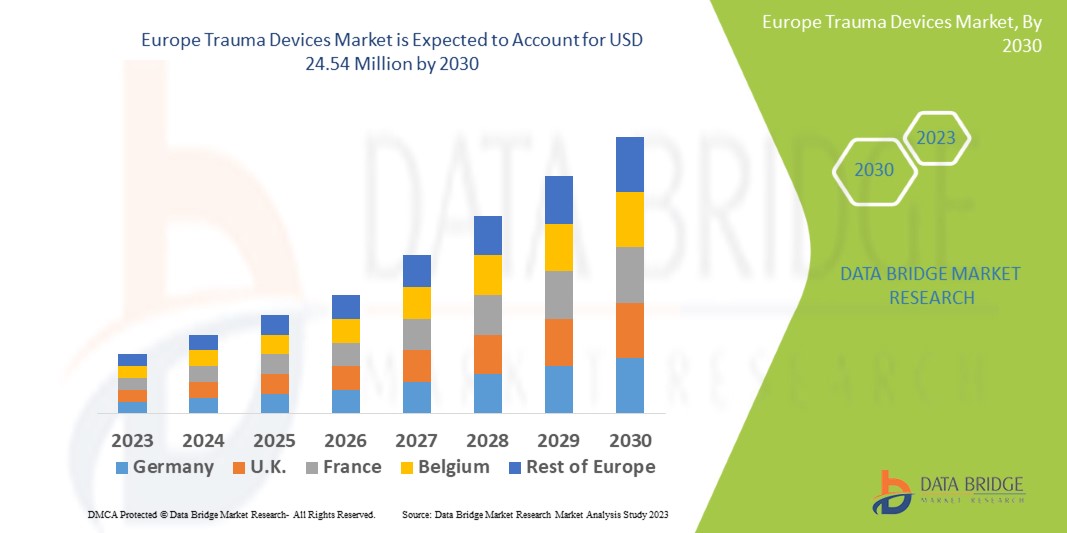
Data Bridge Market Research analyses that the trauma devices market which is USD 13.66 million in 2022, is expected to reach USD 24.54 million by 2030, at a CAGR of 7.6% during the forecast period 2023 to 2030. In addition to the insights on market scenarios such as market value, growth rate, segmentation, geographical coverage, and major players, the market reports curated by the Data Bridge Market Research also include depth expert analysis, patient epidemiology, pipeline analysis, pricing analysis, and regulatory framework.
Trauma Devices Market Scope and Segmentation
|
Report Metric |
Details |
|
Forecast Period |
2023 to 2030 |
|
Base Year |
2022 |
|
Historic Years |
2021 (Customizable to 2015 - 2020) |
|
Quantitative Units |
Revenue in USD Million, Volumes in Units, Pricing in USD |
|
Segments Covered |
Product (Internal Fixators Devices, External Fixators Devices and Others), Surgical Site (Lower Extremities and Upper Extremities), Tissue Type (Hard Tissue and Soft Tissue), Material Type (Non-Absorbable and Bio-Absorbable), Patient Age (Adults and Paediatric), End User (Hospitals, Trauma Centres, Ambulatory Surgical Centres and Other), Distribution Channel (Direct Tender and Retail Sales) |
|
Countries Covered |
Germany, France, U.K., Netherlands, Switzerland, Belgium, Russia, Italy, Spain, Turkey, Rest of Europe in Europe |
|
Market Players Covered |
Shanghai Kinetic Medical Co. Ltd (China), Weigao group (China), MicroPort Scientific Corporation (China), Orthofix US LLC (U.S.), CONMED Corporation (U.S.), Wright Medical Group N.V. (U.S.), NuVasive, Inc (U.S.), Corin Group (U.S.), Enovis (U.S.), OsteoMed (U.S.), Invibio Ltd. (U.S.), gpcmedical (U.S.), Medtronic (Ireland), Smith+Nephew (U.S.), Integra LifeSciences (U.S.), B. Braun SE (Germany), Stryker (U.S.) |
|
Market Opportunities |
|
Market Definition
Trauma devices are a clinical practise used in the treatment of trauma. Fractures, dislocations, sprains, strains and burns are instances of trauma exterior injuries. These technologies are particularly helpful in situations involving long bones, such as fractures. The upper and lower extremities parts of the body such as the knee, joint, leg, spine and others are also included in the trauma devices.
Trauma Devices Market Dynamics
Drivers
- Technological advancements
Technology advances are another major driver of the market, with top companies offering a wide range of products to their strong research skills and partnerships with other companies to diversify their product portfolios. For instance, Cardinal Health paid USD 6.1 billion to purchase the patient recovery business from Medtronic in July 2017. The acquisition added 23 new product categories to Cardinal Health's portfolio, including market-leading brands including Kendall, Curity, Kangaroo, Dover and Argyle.
- Increase in the aging population
The market will be driven by factors such an ageing population that is more prone to osteoarthritic fractures, a growth in the prevalence of osteoporosis and technological breakthroughs such as the usage of orthobiologic products and biodegradable materials. The industry will also grow as a result of a rise in traffic accidents, sports-related injuries and lifestyle changes among people.
- Injuries caused by trauma
Trauma can result in tissue injuries as well as bone fractures or breaks. Trauma devices are used to treat these wounds by replacing the injured bodily portion. Injuries from motor vehicle accidents frequently include trauma. An increase in the number of events in emerging nations has propelled the industry.
Opportunities
- Increased government attention on new medication discovery and development
The rapid spread of COVID-19 throughout the world, a rapidly growing patient base, significant changes in demand and supply of healthcare solutions during the COVID-19 outbreak and an increasing demand for advanced point-of-care diagnostics are the main factors propelling market revenue growth. Higher government funding for the COVID-19 vaccine programme, increased government focus on the discovery and development of innovative pharmaceuticals, and increased government focus on improving the healthcare supply chain system all have an impact on market revenue growth.
Restraints/Challenges
- High cost of trauma devices
The market expansion is anticipated to be hampered by the high cost of trauma devices, allergies related to internal fixation devices, the danger of infection, price fluctuations, and a lack of qualified professionals. Furthermore, regulatory agencies take their time to approve the materials used to make trauma devices due to worries about patient compatibility and allergies. The components of these implants should be very inert and biocompatible. As a result, it might be challenging for enterprises to get approval for these devices.
This trauma devices market report provides details of new recent developments, trade regulations, import-export analysis, production analysis, value chain optimization, market share, impact of domestic and localized market players, analyses opportunities in terms of emerging revenue pockets, changes in market regulations, strategic market growth analysis, market size, category market growths, application niches and dominance, product approvals, product launches, geographic expansions, technological innovations in the market. To gain more info on the trauma devices market contact Data Bridge Market Research for an Analyst Brief, our team will help you take an informed market decision to achieve market growth.
COVID-19 Impact on Trauma Devices Market
Global economy has been significantly impacted by the COVID-19 pandemic, with major losses expected across all sectors by 2020. The pandemic's stress on healthcare facilities to stop spread of virus which had a severe negative impact on business. The non-essential character of trauma fixation operations led to the reported suspension or permanent delay of numerous treatments and surgical procedures throughout the epidemic. The industry's expansion was restrained due to the limited demand for trauma fixation devices in the early months of the epidemic. Additionally, the growth of multispecialty hospitals will boost industry earnings by improving trauma care, especially in developing countries such as India. Similar to this, low-cost treatments offered by various government agencies will significantly stimulate the growth of the market in low-income countries.
Recent Developments
- In 2019, A private equity firm with numerous subsidiaries, including bracing and supports, surgical, footcare, healthcare solutions, recovery, and consumer, was acquired by Colfax Corporation. By raising income, this acquisition will enable the business to increase profits.
- In 2019, Titan Spine was acquired by Medtronic. This acquisition resulted in the expansion of Medtronic's titanium spine interbody implant and surface technology portfolios. This helped the business's trauma gadget selection as well.
Europe Trauma Devices Market Scope
The trauma devices market is segmented on the basis of type, surgical site and end-user. The growth amongst these segments will help you analyze meagre growth segments in the industries and provide the users with a valuable market overview and market insights to help them make strategic decisions for identifying core market applications.
Type
- Internal Fixators
- Plates and Screws
- Rods and Pins
- Others
- External Fixators
- Uniplanar and Biplanar Fixators
- Circular Fixators
- Hybrid Fixators
Surgical Site
- Upper Extremity
- Hand and Wrist
- Shoulder
- Arm
- Elbow
- Lower Extremity
- Hip and Pelvis
- Lower Leg
- Foot and Ankle
- Knee
- Thigh
End-User
- Hospitals
- Ambulatory Surgical Centers
Trauma Devices Market Regional Analysis/Insights
The trauma devices market is analyzed and market size insights and trends are provided by country, type, surgical site and end-user as referenced above.
The countries covered in the trauma devices market report are Germany, France, U.K., Netherlands, Switzerland, Belgium, Russia, Italy, Spain, Turkey, Rest of Europe in Europe.
Germany is dominating the trauma devices market due to a high number of businesses, R&D skills, and the expanding use of less invasive surgical techniques. With significant assistance from the government, the healthcare infrastructure is also being improved. Despite this, it has been shown that fewer traffic accidents occur in emerging nations. The market will rise, nevertheless, because countries with big populations are more prone to traffic accidents.
The country section of the report also provides individual market impacting factors and changes in regulation in the market domestically that impacts the current and future trends of the market. Data points like down-stream and upstream value chain analysis, technical trends and porter's five forces analysis, case studies are some of the pointers used to forecast the market scenario for individual countries. Also, the presence and availability of global brands and their challenges faced due to large or scarce competition from local and domestic brands, impact of domestic tariffs and trade routes are considered while providing forecast analysis of the country data.
Healthcare Infrastructure Growth Installed base and New Technology Penetration
The trauma devices market also provides you with detailed market analysis for every country growth in healthcare expenditure for capital equipment, installed base of different kind of products for trauma devices market, impact of technology using life line curves and changes in healthcare regulatory scenarios and their impact on the trauma devices market. The data is available for historic period 2011-2021.
Competitive Landscape and Trauma Devices Market Share Analysis
The trauma devices market competitive landscape provides details by competitor. Details included are company overview, company financials, revenue generated, market potential, investment in research and development, new market initiatives, global presence, production sites and facilities, production capacities, company strengths and weaknesses, product launch, product width and breadth, application dominance. The above data points provided are only related to the companies' focus related to trauma devices market.
Some of the major players operating in the trauma devices market are:
- Shanghai Kinetic Medical Co. Ltd (China)
- Weigao group (China)
- MicroPort Scientific Corporation (China)
- Orthofix US LLC (U.S.)
- CONMED Corporation (U.S.)
- Wright Medical Group N.V. (U.S.)
- NuVasive, Inc (U.S.)
- Corin Group (U.S.)
- Enovis (U.S.)
- OsteoMed (U.S.)
- Invibio Ltd. (U.S.)
- gpcmedical (U.S.)
- Medtronic (Ireland)
- Smith+Nephew (U.S.)
- Integra LifeSciences (U.S.)
- B. Braun SE (Germany)
- Stryker (U.S.)
SKU-
Get online access to the report on the World's First Market Intelligence Cloud
- Interactive Data Analysis Dashboard
- Company Analysis Dashboard for high growth potential opportunities
- Research Analyst Access for customization & queries
- Competitor Analysis with Interactive dashboard
- Latest News, Updates & Trend analysis
- Harness the Power of Benchmark Analysis for Comprehensive Competitor Tracking
Table of Content
1 INTRODUCTION
1.1 OBJECTIVES OF THE STUDY
1.2 MARKET DEFINITION
1.3 OVERVIEW OF EUROPE TRAUMA DEVICES MARKET
1.4 CURRENCY AND PRICING
1.5 LIMITATIONS
1.6 MARKETS COVERED
2 MARKET SEGMENTATION
2.1 MARKETS COVERED
2.2 GEOGRAPHICAL SCOPE
2.3 YEARS CONSIDERED FOR THE STUDY
2.4 DBMR TRIPOD DATA VALIDATION MODEL
2.5 PRIMARY INTERVIEWS WITH KEY OPINION LEADERS
2.6 MULTIVARIATE MODELLING
2.7 PRODUCTS LIFELINE CURVE
2.8 DBMR MARKET POSITION GRID
2.9 VENDOR SHARE ANALYSIS
2.1 MARKET END-USER COVERAGE GRID
2.11 SECONDARY SOURCES
2.12 ASSUMPTIONS
3 EXECUTIVE SUMMARY
4 PREMIUM INSIGHTS
5 MARKET OVERVIEW
5.1 DRIVERS
5.1.1 INCREASING INCIDENTS OF ROAD ACCIDENTS AND INJURIES WORLDWIDE
5.1.2 RISING GERIATRIC POPULATION
5.1.3 GROWING ADOPTION OF MINIMALLY INVASIVE SURGICAL PROCEDURES
5.1.4 INCREASING PREVALENCE OF DEGENERATIVE BONE DISEASES
5.2 RESTRAINTS
5.2.1 HIGH COST OF TRAUMA DEVICES AND SURGICAL PROCEDURES
5.2.2 STRINGENT REGULATION SCENARIO
5.2.3 HIGH RISK AND COMPLICATIONS OF TRAUMA SURGERY
5.3 OPPORTUNITIES
5.3.1 TECHNOLOGICAL ADVANCEMENTS IN THE TRAUMA DEVICES
5.3.2 INCREASING AWARENESS ABOUT TRAUMA DEVICES
5.4 CHALLENGE
5.4.1 DEARTH OF TECHNICALLY SKILLED PROFESSIONALS REQUIRED FOR SUCCESSFUL OPERATING
6 EUROPE TRAUMA DEVICES MARKET, BY PRODUCT
6.1 OVERVIEW
6.2 INTERNAL FIXATOR DEVICES
6.2.1 PLATES
6.2.2 SCREW
6.2.2.1 CORTICAL SCREWS
6.2.2.2 CANCELLOUS SCREWS
6.2.2.3 OTHERS
6.2.3 RODS AND PINS
6.2.4 NAILING
6.2.5 SUTURE
6.2.6 STAPLES
6.2.7 FILLERS
6.2.8 WIRES
6.2.9 ARROWS AND DARTS
6.2.10 BUTTONS
6.2.11 TACKS
6.2.12 OTHERS
6.3 EXTERNAL FIXATOR DEVICES
6.3.1 AXIAL
6.3.2 RADIUS
6.3.3 HYBRID
6.3.4 UNILATERAL & BILATERAL
6.3.5 SCREWS & PINS
6.3.6 CIRCULAR
6.3.7 OTHERS
6.4 OTHERS
7 EUROPE TRAUMA DEVICES MARKET, BY SURGICAL SITE
7.1 OVERVIEW
7.2 LOWER EXTREMITIES
7.2.1 FOOT & ANKLE
7.2.2 OTHERS
7.3 UPPER EXTREMITIES
7.3.1 HAND & WRIST
7.3.2 SHOULDER
7.3.3 ELBOW
7.3.4 OTHERS
8 EUROPE TRAUMA DEVICES MARKET, BY TISSUE
8.1 OVERVIEW
8.2 HARD TISSUE
8.3 SOFT TISSUE
9 EUROPE TRAUMA DEVICES MARKET, BY MATERIAL
9.1 OVERVIEW
9.2 NON-ABSORBABLE
9.2.1 METALS
9.2.1.1 STAINLESS STEEL
9.2.1.2 TITANIUM ALLOYS
9.2.1.3 CO-CR ALLOYS
9.2.1.4 OTHERS
9.2.2 POLYMERS
9.2.3 CERAMICS
9.2.3.1 HYDROXYAPATITE
9.2.3.2 ZIRCONIA
9.2.3.3 ALUMINA
9.2.3.4 OTHERS
9.2.4 COMPOSITE
9.2.5 OTHERS
9.3 BIO-ABSORBABLE
9.3.1 POLYGLYCOLIC ACID POLYMER
9.3.2 POLYLACTIC ACID POLYMER
9.3.3 OTHERS
10 EUROPE TRAUMA DEVICES MARKET, BY PATIENT AGE
10.1 OVERVIEW
10.2 ADULTS
10.3 PEDIATRIC
11 EUROPE TRAUMA DEVICES MARKET, BY END USER
11.1 OVERVIEW
11.2 HOSPITALS
11.3 TRAUMA CENTERS
11.4 AMBULATORY SURGICAL CENTERS
11.5 OTHERS
12 EUROPE TRAUMA DEVICES MARKET, BY DISTRIBUTION CHANNEL
12.1 OVERVIEW
12.2 DIRECT TENDER
12.3 RETAIL SALES
13 EUROPE TRAUMA DEVICES MARKET, BY GEOGRAPHY
13.1 EUROPE
13.1.1 GERMANY
13.1.2 FRANCE
13.1.3 ITALY
13.1.4 U.K.
13.1.5 SPAIN
13.1.6 RUSSIA
13.1.7 NETHERLANDS
13.1.8 SWITZERLAND
13.1.9 BELGIUM
13.1.10 TURKEY
13.1.11 REST OF EUROPE
14 EUROPE TRAUMA DEVICES MARKET, COMPANY LANDSCAPE
14.1 COMPANY SHARE ANALYSIS: EUROPE
15 COMPANY PROFILE
15.1 DEPUY SYNTHES COMPANIES (A SUBSIDIARY OF JOHNSON & JOHNSON SERVICES, INC.)
15.1.1 COMPANY SNAPSHOT
15.1.2 REVENUE ANALYSIS
15.1.3 COMPANY SHARE ANALYSIS
15.1.4 PRODUCT PORTFOLIO
15.1.5 RECENT DEVELOPMENTS
15.2 STRYKER
15.2.1 COMPANY SNAPSHOT
15.2.2 REVENUE ANALYSIS
15.2.3 COMPANY SHARE ANALYSIS
15.2.4 PRODUCT PORTFOLIO
15.2.5 RECENT DEVELOPMENT
15.3 ZIMMER BIOMET
15.3.1 COMPANY SNAPSHOT
15.3.2 REVENUE ANALYSIS
15.3.3 COMPANY SHARE ANALYSIS
15.3.4 PRODUCT PORTFOLIO
15.3.5 RECENT DEVELOPMENTS
15.4 SMITH & NEPHEW
15.4.1 COMPANY SNAPSHOT
15.4.2 REVENUE ANALYSIS
15.4.3 COMPANY SHARE ANALYSIS
15.4.4 PRODUCT PORTFOLIO
15.4.5 RECENT DEVELOPMENTS
15.5 B. BRAUN MELSUNGEN AG
15.5.1 COMPANY SNAPSHOT
15.5.2 REVENUE ANALYSIS
15.5.3 PRODUCT PORTFOLIO
15.5.4 RECENT DEVELOPMENT
15.6 CONMED CORPORATION
15.6.1 COMPANY SNAPSHOT
15.6.2 REVENUE ANALYSIS
15.6.3 PRODUCT PORTFOLIO
15.6.4 RECENT DEVELOPMENT
15.7 CORIN
15.7.1 COMPANY SNAPSHOT
15.7.2 PRODUCT PORTFOLIO
15.7.3 RECENT DEVELOPMENTS
15.8 DJO EUROPE, INC (A SUBSIDIARY OF COLFAX CORPORATION)
15.8.1 COMPANY SNAPSHOT
15.8.2 REVENUE ANALYSIS
15.8.3 PRODUCT PORTFOLIO
15.8.4 RECENT DEVELOPMENT
15.9 GPCMEDICAL.COM
15.9.1 COMPANY SNAPSHOT
15.9.2 PRODUCT PORTFOLIO
15.9.3 RECENT DEVELOPMENTS
15.1 INVIBIO LTD
15.10.1 COMPANY SNAPSHOT
15.10.2 PRODUCT PORTFOLIO
15.10.3 RECENT DEVELOPMENT
15.11 INTEGRA LIFESCIENCES CORPORATION
15.11.1 COMPANY SNAPSHOT
15.11.2 REVENUE ANALYSIS
15.11.3 PRODUCT PORTFOLIO
15.11.4 RECENT DEVELOPMENT
15.12 MEDTRONIC
15.12.1 COMPANY SNAPSHOT
15.12.2 REVENUE ANALYSIS
15.12.3 PRODUCT PORTFOLIO
15.12.4 RECENT DEVELOPMENTS
15.13 MICROPORT SCIENTIFIC CORPORATION
15.13.1 COMPANY SNAPSHOT
15.13.2 REVENUE ANALYSIS
15.13.3 PRODUCT PORTFOLIO
15.13.4 RECENT DEVELOPMENTS
15.14 NUVASIVE, INC
15.14.1 COMPANY SNAPSHOT
15.14.2 REVENUE ANALYSIS
15.14.3 PRODUCT PORTFOLIO
15.14.4 RECENT DEVELOPMENT
15.15 ORTHOFIX MEDICAL INC.
15.15.1 COMPANY SNAPSHOT
15.15.2 REVENUE ANALYSIS
15.15.3 PRODUCT PORTFOLIO
15.15.4 RECENT DEVELOPMENT
15.16 OSTEOMED
15.16.1 COMPANY SNAPSHOT
15.16.2 PRODUCT PORTFOLIO
15.16.3 RECENT DEVELOPMENT
15.17 SHANGHAI KINETIC MEDICAL CO., LTD.
15.17.1 COMPANY SNAPSHOT
15.17.2 PRODUCT PORTFOLIO
15.17.3 RECENT DEVELOPMENT
15.18 WEIGAO GROUP
15.18.1 COMPANY SNAPSHOT
15.18.2 REVENUE ANALYSIS
15.18.3 PRODUCT PORTFOLIO
15.18.4 RECENT DEVELOPMENT
15.19 WRIGHT MEDICAL GROUP N.V.
15.19.1 COMPANY SNAPSHOT
15.19.2 REVENUE ANALYSIS
15.19.3 PRODUCT PORTFOLIO
15.19.4 RECENT DEVELOPMENTS
16 SWOT & DATA BRIDGE MARKET RESEARCH ANALYSIS
16.1 SWOT ANALYSIS
16.1.1 STRENGTH: - SIGNIFICANT GROWTH OF SHARES IN MARKET FOCUSED SEGMENT
16.1.2 STRENGTH: - STRONG EUROPE PRESENCE
16.1.3 WEAKNESS: - STRINGENT REGULATION FOR COMMERCIALIZATION OF THE PRODUCT
16.1.4 OPPORTUNITY: - STRATEGIC BUSINESS EXPANISION THOROUGH THEIR SUBSIDIARIES
16.1.5 THREAT: - CURRENCY FLUCTATUATION
16.2 DATA BRIDGE MARKET RESEARCH ANALYSIS
17 CONCLUSION
18 QUESTIONNAIRE
19 RELATED REPORTS
List of Table
LIST OF TABLES
TABLE 1 MEDICAL DEVICE REIMBURSEMENT EXPENDITURE RATIO BY PRODUCT CATEGORY (%)
TABLE 2 SOURCE OF HEALTH FUNDS IN LGUS (IN MILLION PHP), 2003–2007
TABLE 3 PERCENTAGE OF PRIVATE INSURED PATIENTS VS UNDER PUBLIC FOR EACH ASIA PACIFIC COUNTRIES
TABLE 4 LIST OF COMPETITORS IN DIFFERENT REGIONS OF CHINA
TABLE 5 REIMBURSEMENT SCENARIO IN CHINA
TABLE 6 QUANTITY, TYPE, AND LOCATION OF HOSPITALS THAT ARE ABLE TO USE/AFFORD IMPORTED PRODUCTS IN DIFFERENT REGIONS OF CHINA
TABLE 7 CAUSES OF ROAD TRAFFIC ACCIDENTS
TABLE 8 DEMOGRAPHIC, INJURY AND EVENT CHARACTERISTICS OF OLDER ADULTS WITH MAJOR TRAUMA
TABLE 9 EXISTENCE OF SPINAL AND SUPRASPINAL NEUROINFLAMMATION IN JOINT DISEASES
TABLE 10 TOP 10” INSTRUMENT PARTS SOLD IN TRAUMA CASES
TABLE 11 EUROPE TRAUMA DEVICES MARKET, BY PRODUCT, 2018-2027 (USD MILLION)
TABLE 12 EUROPE TRAUMA DEVICES MARKET, BY PRODUCT, 2018-2027 (UNITS)
TABLE 13 EUROPE INTERNAL FIXATOR DEVICES IN TRAUMA DEVICES MARKET, BY REGION, 2018-2027 (USD MILLION)
TABLE 14 EUROPE INTERNAL FIXATION DEVICES IN TRAUMA DEVICES MARKET, BY PRODUCT, 2018-2027 (USD MILLION)
TABLE 15 EUROPE SCREW IN TRAUMA DEVICES MARKET, BY PRODUCT, 2018-2027 (USD MILLION)
TABLE 16 EUROPE EXTERNAL FIXATOR DEVICES IN TRAUMA DEVICES MARKET, BY REGION, 2018-2027 (USD MILLION)
TABLE 17 EUROPE EXTERNAL FIXATOR DEVICES IN TRAUMA DEVICES MARKET, BY PRODUCT, 2018-2027 (USD MILLION)
TABLE 18 EUROPE OTHERS IN TRAUMA DEVICES MARKET, BY REGION, 2018-2027 (USD MILLION)
TABLE 19 EUROPE TRAUMA DEVICES MARKET, BY SURGICAL SITES, 2018-2027 (USD MILLION)
TABLE 20 EUROPE LOWER EXTREMITIES IN TRAUMA DEVICES MARKET, BY REGION, 2018-2027 (USD MILLION)
TABLE 21 EUROPE LOWER EXTREMITIES IN TRAUMA DEVICES MARKET, BY SURGICAL SITES, 2018-2027 (USD MILLION)
TABLE 22 EUROPE UPPER EXTREMITIES IN TRAUMA DEVICES MARKET, BY REGION, 2018-2027 (USD MILLION)
TABLE 23 EUROPE UPPER EXTREMITIES IN TRAUMA DEVICES MARKET, BY SURGICAL SITES, 2018-2027 (USD MILLION)
TABLE 24 EUROPE TRAUMA DEVICES MARKET, BY TISSUE, 2018-2027 (USD MILLION)
TABLE 25 EUROPE HARD TISSUE IN TRAUMA DEVICES MARKET, BY REGION, 2018-2027 (USD MILLION)
TABLE 26 EUROPE SOFT TISSUE IN TRAUMA DEVICES MARKET, BY REGION, 2018-2027 (USD MILLION)
TABLE 27 EUROPE TRAUMA DEVICES MARKET, BY MATERIAL, 2018-2027 (USD MILLION)
TABLE 28 EUROPE NON-ABSORBABLE IN TRAUMA DEVICES MARKET, BY REGION, 2018-2027 (USD MILLION)
TABLE 29 EUROPE NON-ABSORBABLE IN TRAUMA DEVICES MARKET, BY MATERIAL, 2018-2027 (USD MILLION)
TABLE 30 EUROPE METALS IN TRAUMA DEVICES MARKET, BY MATERIAL, 2018-2027 (USD MILLION)
TABLE 31 EUROPE CERAMICS IN TRAUMA DEVICES MARKET, BY MATERIAL, 2018-2027 (USD MILLION)
TABLE 32 EUROPE BIO-ABSORBABLE IN TRAUMA DEVICES MARKET, BY REGION, 2018-2027 (USD MILLION)
TABLE 33 EUROPE BIO-ABSORBABLE IN TRAUMA DEVICES MARKET, BY MATERIAL, 2018-2027 (USD MILLION)
TABLE 34 EUROPE TRAUMA DEVICES MARKET, BY PATIENT AGE, 2018-2027 (USD MILLION)
TABLE 35 EUROPE ADULT IN TRAUMA DEVICES MARKET, BY REGION, 2018-2027 (USD MILLION)
TABLE 36 EUROPE PEDIATRIC IN TRAUMA DEVICES MARKET, BY REGION, 2018-2027 (USD MILLION)
TABLE 37 EUROPE TRAUMA DEVICES MARKET, BY END USER, 2018-2027 (USD MILLION)
TABLE 38 EUROPE HOSPITALS IN TRAUMA DEVICES MARKET, BY REGION, 2018-2027 (USD MILLION)
TABLE 39 EUROPE TRAUMA CENTERS IN TRAUMA DEVICES MARKET, BY REGION, 2018-2027 (USD MILLION)
TABLE 40 EUROPE AMBULATORY SURGICAL CENTERS IN TRAUMA DEVICES MARKET, BY REGION, 2018-2027 (USD MILLION)
TABLE 41 EUROPE OTHERS IN TRAUMA DEVICES MARKET, BY REGION, 2018-2027 (USD MILLION)
TABLE 42 EUROPE TRAUMA DEVICES MARKET, BY DISTRIBUTION CHANNEL , 2018-2027 (USD MILLION)
TABLE 43 EUROPE DIRECT TENDER IN TRAUMA DEVICES MARKET, BY REGION, 2018-2027 (USD MILLION)
TABLE 44 EUROPE RETAIL SALES IN TRAUMA DEVICES MARKET, BY REGION, 2018-2027 (USD MILLION)
TABLE 45 EUROPE TRAUMA DEVICES MARKET, BY COUNTRY, 2018-2027 (USD MILLION)
TABLE 46 EUROPE TRAUMA DEVICES MARKET, BY PRODUCT, 2018-2027 (USD MILLION)
TABLE 47 EUROPE TRAUMA DEVICES MARKET, BY PRODUCT, 2018-2027 (UNITS)
TABLE 48 EUROPE INTERNAL FIXATOR DEVICES IN TRAUMA DEVICES MARKET, BY PRODUCT, 2018-2027 (USD MILLION)
TABLE 49 EUROPE SCREW IN TRAUMA DEVICES MARKET, BY PRODUCT, 2018-2027 (USD MILLION)
TABLE 50 EUROPE EXTERNAL FIXATOR DEVICES IN TRAUMA DEVICES MARKET, BYPRODUCT, 2018-2027 (USD MILLION)
TABLE 51 EUROPE TRAUMA DEVICES MARKET, BY SURGICAL SITE, 2018-2027 (USD MILLION)
TABLE 52 EUROPE LOWER EXTREMITIES IN TRAUMA DEVICES MARKET, BY SURGICAL SITE, 2018-2027 (USD MILLION)
TABLE 53 EUROPE UPPER EXTREMITIES IN TRAUMA DEVICES TRAUMA DEVICES MARKET, BY SURGICAL SITE, 2018-2027 (USD MILLION)
TABLE 54 EUROPE TRAUMA DEVICES MARKET, BY TISSUE, 2018-2027 (USD MILLION)
TABLE 55 EUROPE TRAUMA DEVICES MARKET, BY MATERIAL, 2018-2027 (USD MILLION)
TABLE 56 EUROPE NON-ABSORBABLE IN TRAUMA DEVICES MARKET, BY MATERIAL, 2018-2027 (USD MILLION)
TABLE 57 EUROPE METALS IN TRAUMA DEVICES MARKET, BY MATERIAL, 2018-2027 (USD MILLION)
TABLE 58 EUROPE CERAMICS IN TRAUMA DEVICES MARKET, BY MATERIAL, 2018-2027 (USD MILLION)
TABLE 59 EUROPE BIO-ABSORBABLE IN TRAUMA DEVICES MARKET, BY MATERIAL, 2018-2027 (USD MILLION)
TABLE 60 EUROPE TRAUMA DEVICES MARKET, BY PATIENT AGE, 2018-2027 (USD MILLION)
TABLE 61 EUROPE TRAUMA DEVICES MARKET, BY END USER, 2018-2027 (USD MILLION)
TABLE 62 EUROPE TRAUMA DEVICES MARKET, BY DISTRIBUTION CHANNEL, 2018-2027 (USD MILLION)
TABLE 63 GERMANY TRAUMA DEVICES MARKET, BY PRODUCT, 2018-2027 (USD MILLION)
TABLE 64 GERMANY TRAUMA DEVICES MARKET, BY PRODUCT, 2018-2027 (UNITS)
TABLE 65 GERMANY INTERNAL FIXATOR DEVICES IN TRAUMA DEVICES MARKET, BY PRODUCT, 2018-2027 (USD MILLION)
TABLE 66 GERMANY SCREW IN TRAUMA DEVICES MARKET, BY PRODUCT, 2018-2027 (USD MILLION)
TABLE 67 GERMANY EXTERNAL FIXATOR DEVICES IN TRAUMA DEVICES MARKET, BYPRODUCT, 2018-2027 (USD MILLION)
TABLE 68 GERMANY TRAUMA DEVICES MARKET, BY SURGICAL SITE, 2018-2027 (USD MILLION)
TABLE 69 GERMANY LOWER EXTREMITIES IN TRAUMA DEVICES MARKET, BY SURGICAL SITE, 2018-2027 (USD MILLION)
TABLE 70 GERMANY UPPER EXTREMITIES IN TRAUMA DEVICES TRAUMA DEVICES MARKET, BY SURGICAL SITE, 2018-2027 (USD MILLION)
TABLE 71 GERMANY TRAUMA DEVICES MARKET, BY TISSUE, 2018-2027 (USD MILLION)
TABLE 72 GERMANY TRAUMA DEVICES MARKET, BY MATERIAL, 2018-2027 (USD MILLION)
TABLE 73 GERMANY NON-ABSORBABLE IN TRAUMA DEVICES MARKET, BY MATERIAL, 2018-2027 (USD MILLION)
TABLE 74 GERMANY METALS IN TRAUMA DEVICES MARKET, BY MATERIAL, 2018-2027 (USD MILLION)
TABLE 75 GERMANY CERAMICS IN TRAUMA DEVICES MARKET, BY MATERIAL, 2018-2027 (USD MILLION)
TABLE 76 GERMANY BIO-ABSORBABLE IN TRAUMA DEVICES MARKET, BY MATERIAL, 2018-2027 (USD MILLION)
TABLE 77 GERMANY TRAUMA DEVICES MARKET, BY PATIENT AGE, 2018-2027 (USD MILLION)
TABLE 78 GERMANY TRAUMA DEVICES MARKET, BY END USER, 2018-2027 (USD MILLION)
TABLE 79 GERMANY TRAUMA DEVICES MARKET, BY DISTRIBUTION CHANNEL, 2018-2027 (USD MILLION)
TABLE 80 FRANCE TRAUMA DEVICES MARKET, BY PRODUCT, 2018-2027 (USD MILLION)
TABLE 81 FRANCE TRAUMA DEVICES MARKET, BY PRODUCT, 2018-2027 (UNITS)
TABLE 82 FRANCE INTERNAL FIXATOR DEVICES IN TRAUMA DEVICES MARKET, BY PRODUCT, 2018-2027 (USD MILLION)
TABLE 83 FRANCE SCREW IN TRAUMA DEVICES MARKET, BY PRODUCT, 2018-2027 (USD MILLION)
TABLE 84 FRANCE EXTERNAL FIXATOR DEVICES IN TRAUMA DEVICES MARKET, BYPRODUCT, 2018-2027 (USD MILLION)
TABLE 85 FRANCE TRAUMA DEVICES MARKET, BY SURGICAL SITE, 2018-2027 (USD MILLION)
TABLE 86 FRANCE LOWER EXTREMITIES IN TRAUMA DEVICES MARKET, BY SURGICAL SITE, 2018-2027 (USD MILLION)
TABLE 87 FRANCE UPPER EXTREMITIES IN TRAUMA DEVICES TRAUMA DEVICES MARKET, BY SURGICAL SITE, 2018-2027 (USD MILLION)
TABLE 88 FRANCE TRAUMA DEVICES MARKET, BY TISSUE, 2018-2027 (USD MILLION)
TABLE 89 FRANCE TRAUMA DEVICES MARKET, BY MATERIAL, 2018-2027 (USD MILLION)
TABLE 90 FRANCE NON-ABSORBABLE IN TRAUMA DEVICES MARKET, BY MATERIAL, 2018-2027 (USD MILLION)
TABLE 91 FRANCE METALS IN TRAUMA DEVICES MARKET, BY MATERIAL, 2018-2027 (USD MILLION)
TABLE 92 FRANCE CERAMICS IN TRAUMA DEVICES MARKET, BY MATERIAL, 2018-2027 (USD MILLION)
TABLE 93 FRANCE BIO-ABSORBABLE IN TRAUMA DEVICES MARKET, BY MATERIAL, 2018-2027 (USD MILLION)
TABLE 94 FRANCE TRAUMA DEVICES MARKET, BY PATIENT AGE, 2018-2027 (USD MILLION)
TABLE 95 FRANCE TRAUMA DEVICES MARKET, BY END USER, 2018-2027 (USD MILLION)
TABLE 96 FRANCE TRAUMA DEVICES MARKET, BY DISTRIBUTION CHANNEL, 2018-2027 (USD MILLION)
TABLE 97 ITALY TRAUMA DEVICES MARKET, BY PRODUCT, 2018-2027 (USD MILLION)
TABLE 98 ITALY TRAUMA DEVICES MARKET, BY PRODUCT, 2018-2027 (UNITS)
TABLE 99 ITALY INTERNAL FIXATOR DEVICES IN TRAUMA DEVICES MARKET, BY PRODUCT, 2018-2027 (USD MILLION)
TABLE 100 ITALY SCREW IN TRAUMA DEVICES MARKET, BY PRODUCT, 2018-2027 (USD MILLION)
TABLE 101 ITALY EXTERNAL FIXATOR DEVICES IN TRAUMA DEVICES MARKET, BYPRODUCT, 2018-2027 (USD MILLION)
TABLE 102 ITALY TRAUMA DEVICES MARKET, BY SURGICAL SITE, 2018-2027 (USD MILLION)
TABLE 103 ITALY LOWER EXTREMITIES IN TRAUMA DEVICES MARKET, BY SURGICAL SITE, 2018-2027 (USD MILLION)
TABLE 104 ITALY UPPER EXTREMITIES IN TRAUMA DEVICES TRAUMA DEVICES MARKET, BY SURGICAL SITE, 2018-2027 (USD MILLION)
TABLE 105 ITALY TRAUMA DEVICES MARKET, BY TISSUE, 2018-2027 (USD MILLION)
TABLE 106 ITALY TRAUMA DEVICES MARKET, BY MATERIAL, 2018-2027 (USD MILLION)
TABLE 107 ITALY NON-ABSORBABLE IN TRAUMA DEVICES MARKET, BY MATERIAL, 2018-2027 (USD MILLION)
TABLE 108 ITALY METALS IN TRAUMA DEVICES MARKET, BY MATERIAL, 2018-2027 (USD MILLION)
TABLE 109 ITALY CERAMICS IN TRAUMA DEVICES MARKET, BY MATERIAL, 2018-2027 (USD MILLION)
TABLE 110 ITALY BIO-ABSORBABLE IN TRAUMA DEVICES MARKET, BY MATERIAL, 2018-2027 (USD MILLION)
TABLE 111 ITALY TRAUMA DEVICES MARKET, BY PATIENT AGE, 2018-2027 (USD MILLION)
TABLE 112 ITALY TRAUMA DEVICES MARKET, BY END USER, 2018-2027 (USD MILLION)
TABLE 113 ITALY TRAUMA DEVICES MARKET, BY DISTRIBUTION CHANNEL, 2018-2027 (USD MILLION)
TABLE 114 U.K. TRAUMA DEVICES MARKET, BY PRODUCT, 2018-2027 (USD MILLION)
TABLE 115 U.K. TRAUMA DEVICES MARKET, BY PRODUCT, 2018-2027 (UNITS)
TABLE 116 U.K. INTERNAL FIXATOR DEVICES IN TRAUMA DEVICES MARKET, BY PRODUCT, 2018-2027 (USD MILLION)
TABLE 117 U.K. SCREW IN TRAUMA DEVICES MARKET, BY PRODUCT, 2018-2027 (USD MILLION)
TABLE 118 U.K. EXTERNAL FIXATOR DEVICES IN TRAUMA DEVICES MARKET, BYPRODUCT, 2018-2027 (USD MILLION)
TABLE 119 U.K. TRAUMA DEVICES MARKET, BY SURGICAL SITE, 2018-2027 (USD MILLION)
TABLE 120 U.K. LOWER EXTREMITIES IN TRAUMA DEVICES MARKET, BY SURGICAL SITE, 2018-2027 (USD MILLION)
TABLE 121 U.K. UPPER EXTREMITIES IN TRAUMA DEVICES TRAUMA DEVICES MARKET, BY SURGICAL SITE, 2018-2027 (USD MILLION)
TABLE 122 U.K. TRAUMA DEVICES MARKET, BY TISSUE, 2018-2027 (USD MILLION)
TABLE 123 U.K. TRAUMA DEVICES MARKET, BY MATERIAL, 2018-2027 (USD MILLION)
TABLE 124 U.K. NON-ABSORBABLE IN TRAUMA DEVICES MARKET, BY MATERIAL, 2018-2027 (USD MILLION)
TABLE 125 U.K. METALS IN TRAUMA DEVICES MARKET, BY MATERIAL, 2018-2027 (USD MILLION)
TABLE 126 U.K. CERAMICS IN TRAUMA DEVICES MARKET, BY MATERIAL, 2018-2027 (USD MILLION)
TABLE 127 U.K. BIO-ABSORBABLE IN TRAUMA DEVICES MARKET, BY MATERIAL, 2018-2027 (USD MILLION)
TABLE 128 U.K. TRAUMA DEVICES MARKET, BY PATIENT AGE, 2018-2027 (USD MILLION)
TABLE 129 U.K. TRAUMA DEVICES MARKET, BY END USER, 2018-2027 (USD MILLION)
TABLE 130 U.K. TRAUMA DEVICES MARKET, BY DISTRIBUTION CHANNEL, 2018-2027 (USD MILLION)
TABLE 131 SPAIN TRAUMA DEVICES MARKET, BY PRODUCT, 2018-2027 (USD MILLION)
TABLE 132 SPAIN TRAUMA DEVICES MARKET, BY PRODUCT, 2018-2027 (UNITS)
TABLE 133 SPAIN INTERNAL FIXATOR DEVICES IN TRAUMA DEVICES MARKET, BY PRODUCT, 2018-2027 (USD MILLION)
TABLE 134 SPAIN SCREW IN TRAUMA DEVICES MARKET, BY PRODUCT, 2018-2027 (USD MILLION)
TABLE 135 SPAIN EXTERNAL FIXATOR DEVICES IN TRAUMA DEVICES MARKET, BYPRODUCT, 2018-2027 (USD MILLION)
TABLE 136 SPAIN TRAUMA DEVICES MARKET, BY SURGICAL SITE, 2018-2027 (USD MILLION)
TABLE 137 SPAIN LOWER EXTREMITIES IN TRAUMA DEVICES MARKET, BY SURGICAL SITE, 2018-2027 (USD MILLION)
TABLE 138 SPAIN UPPER EXTREMITIES IN TRAUMA DEVICES TRAUMA DEVICES MARKET, BY SURGICAL SITE, 2018-2027 (USD MILLION)
TABLE 139 SPAIN TRAUMA DEVICES MARKET, BY TISSUE, 2018-2027 (USD MILLION)
TABLE 140 SPAIN TRAUMA DEVICES MARKET, BY MATERIAL, 2018-2027 (USD MILLION)
TABLE 141 SPAIN NON-ABSORBABLE IN TRAUMA DEVICES MARKET, BY MATERIAL, 2018-2027 (USD MILLION)
TABLE 142 SPAIN METALS IN TRAUMA DEVICES MARKET, BY MATERIAL, 2018-2027 (USD MILLION)
TABLE 143 SPAIN CERAMICS IN TRAUMA DEVICES MARKET, BY MATERIAL, 2018-2027 (USD MILLION)
TABLE 144 SPAIN BIO-ABSORBABLE IN TRAUMA DEVICES MARKET, BY MATERIAL, 2018-2027 (USD MILLION)
TABLE 145 SPAIN TRAUMA DEVICES MARKET, BY PATIENT AGE, 2018-2027 (USD MILLION)
TABLE 146 SPAIN TRAUMA DEVICES MARKET, BY END USER, 2018-2027 (USD MILLION)
TABLE 147 SPAIN TRAUMA DEVICES MARKET, BY DISTRIBUTION CHANNEL, 2018-2027 (USD MILLION)
TABLE 148 RUSSIA TRAUMA DEVICES MARKET, BY PRODUCT, 2018-2027 (USD MILLION)
TABLE 149 RUSSIA TRAUMA DEVICES MARKET, BY PRODUCT, 2018-2027 (UNITS)
TABLE 150 RUSSIA INTERNAL FIXATOR DEVICES IN TRAUMA DEVICES MARKET, BY PRODUCT, 2018-2027 (USD MILLION)
TABLE 151 RUSSIA SCREW IN TRAUMA DEVICES MARKET, BY PRODUCT, 2018-2027 (USD MILLION)
TABLE 152 RUSSIA EXTERNAL FIXATOR DEVICES IN TRAUMA DEVICES MARKET, BYPRODUCT, 2018-2027 (USD MILLION)
TABLE 153 RUSSIA TRAUMA DEVICES MARKET, BY SURGICAL SITE, 2018-2027 (USD MILLION)
TABLE 154 RUSSIA LOWER EXTREMITIES IN TRAUMA DEVICES MARKET, BY SURGICAL SITE, 2018-2027 (USD MILLION)
TABLE 155 RUSSIA UPPER EXTREMITIES IN TRAUMA DEVICES TRAUMA DEVICES MARKET, BY SURGICAL SITE, 2018-2027 (USD MILLION)
TABLE 156 RUSSIA TRAUMA DEVICES MARKET, BY TISSUE, 2018-2027 (USD MILLION)
TABLE 157 RUSSIA TRAUMA DEVICES MARKET, BY MATERIAL, 2018-2027 (USD MILLION)
TABLE 158 RUSSIA NON-ABSORBABLE IN TRAUMA DEVICES MARKET, BY MATERIAL, 2018-2027 (USD MILLION)
TABLE 159 RUSSIA METALS IN TRAUMA DEVICES MARKET, BY MATERIAL, 2018-2027 (USD MILLION)
TABLE 160 RUSSIA CERAMICS IN TRAUMA DEVICES MARKET, BY MATERIAL, 2018-2027 (USD MILLION)
TABLE 161 RUSSIA BIO-ABSORBABLE IN TRAUMA DEVICES MARKET, BY MATERIAL, 2018-2027 (USD MILLION)
TABLE 162 RUSSIA TRAUMA DEVICES MARKET, BY PATIENT AGE, 2018-2027 (USD MILLION)
TABLE 163 RUSSIA TRAUMA DEVICES MARKET, BY END USER, 2018-2027 (USD MILLION)
TABLE 164 RUSSIA TRAUMA DEVICES MARKET, BY DISTRIBUTION CHANNEL, 2018-2027 (USD MILLION)
TABLE 165 NETHERLANDS TRAUMA DEVICES MARKET, BY PRODUCT, 2018-2027 (USD MILLION)
TABLE 166 NETHERLANDS TRAUMA DEVICES MARKET, BY PRODUCT, 2018-2027 (UNITS)
TABLE 167 NETHERLANDS INTERNAL FIXATOR DEVICES IN TRAUMA DEVICES MARKET, BY PRODUCT, 2018-2027 (USD MILLION)
TABLE 168 NETHERLANDS SCREW IN TRAUMA DEVICES MARKET, BY PRODUCT, 2018-2027 (USD MILLION)
TABLE 169 NETHERLANDS EXTERNAL FIXATOR DEVICES IN TRAUMA DEVICES MARKET, BYPRODUCT, 2018-2027 (USD MILLION)
TABLE 170 NETHERLANDS TRAUMA DEVICES MARKET, BY SURGICAL SITE, 2018-2027 (USD MILLION)
TABLE 171 NETHERLANDS LOWER EXTREMITIES IN TRAUMA DEVICES MARKET, BY SURGICAL SITE, 2018-2027 (USD MILLION)
TABLE 172 NETHERLANDS UPPER EXTREMITIES IN TRAUMA DEVICES TRAUMA DEVICES MARKET, BY SURGICAL SITE, 2018-2027 (USD MILLION)
TABLE 173 NETHERLANDS TRAUMA DEVICES MARKET, BY TISSUE, 2018-2027 (USD MILLION)
TABLE 174 NETHERLANDS TRAUMA DEVICES MARKET, BY MATERIAL, 2018-2027 (USD MILLION)
TABLE 175 NETHERLANDS NON-ABSORBABLE IN TRAUMA DEVICES MARKET, BY MATERIAL, 2018-2027 (USD MILLION)
TABLE 176 NETHERLANDS METALS IN TRAUMA DEVICES MARKET, BY MATERIAL, 2018-2027 (USD MILLION)
TABLE 177 NETHERLANDS CERAMICS IN TRAUMA DEVICES MARKET, BY MATERIAL, 2018-2027 (USD MILLION)
TABLE 178 NETHERLANDS BIO-ABSORBABLE IN TRAUMA DEVICES MARKET, BY MATERIAL, 2018-2027 (USD MILLION)
TABLE 179 NETHERLANDS TRAUMA DEVICES MARKET, BY PATIENT AGE, 2018-2027 (USD MILLION)
TABLE 180 NETHERLANDS TRAUMA DEVICES MARKET, BY END USER, 2018-2027 (USD MILLION)
TABLE 181 NETHERLANDS TRAUMA DEVICES MARKET, BY DISTRIBUTION CHANNEL, 2018-2027 (USD MILLION)
TABLE 182 SWITZERLAND TRAUMA DEVICES MARKET, BY PRODUCT, 2018-2027 (USD MILLION)
TABLE 183 SWITZERLAND TRAUMA DEVICES MARKET, BY PRODUCT, 2018-2027 (UNITS)
TABLE 184 SWITZERLAND INTERNAL FIXATOR DEVICES IN TRAUMA DEVICES MARKET, BY PRODUCT, 2018-2027 (USD MILLION)
TABLE 185 SWITZERLAND SCREW IN TRAUMA DEVICES MARKET, BY PRODUCT, 2018-2027 (USD MILLION)
TABLE 186 SWITZERLAND EXTERNAL FIXATOR DEVICES IN TRAUMA DEVICES MARKET, BYPRODUCT, 2018-2027 (USD MILLION)
TABLE 187 SWITZERLAND TRAUMA DEVICES MARKET, BY SURGICAL SITE, 2018-2027 (USD MILLION)
TABLE 188 SWITZERLAND LOWER EXTREMITIES IN TRAUMA DEVICES MARKET, BY SURGICAL SITE, 2018-2027 (USD MILLION)
TABLE 189 SWITZERLAND UPPER EXTREMITIES IN TRAUMA DEVICES TRAUMA DEVICES MARKET, BY SURGICAL SITE, 2018-2027 (USD MILLION)
TABLE 190 SWITZERLAND TRAUMA DEVICES MARKET, BY TISSUE, 2018-2027 (USD MILLION)
TABLE 191 SWITZERLAND TRAUMA DEVICES MARKET, BY MATERIAL, 2018-2027 (USD MILLION)
TABLE 192 SWITZERLAND NON-ABSORBABLE IN TRAUMA DEVICES MARKET, BY MATERIAL, 2018-2027 (USD MILLION)
TABLE 193 SWITZERLAND METALS IN TRAUMA DEVICES MARKET, BY MATERIAL, 2018-2027 (USD MILLION)
TABLE 194 SWITZERLAND CERAMICS IN TRAUMA DEVICES MARKET, BY MATERIAL, 2018-2027 (USD MILLION)
TABLE 195 SWITZERLAND BIO-ABSORBABLE IN TRAUMA DEVICES MARKET, BY MATERIAL, 2018-2027 (USD MILLION)
TABLE 196 SWITZERLAND TRAUMA DEVICES MARKET, BY PATIENT AGE, 2018-2027 (USD MILLION)
TABLE 197 SWITZERLAND TRAUMA DEVICES MARKET, BY END USER, 2018-2027 (USD MILLION)
TABLE 198 SWITZERLAND TRAUMA DEVICES MARKET, BY DISTRIBUTION CHANNEL, 2018-2027 (USD MILLION)
TABLE 199 BELGIUM TRAUMA DEVICES MARKET, BY PRODUCT, 2018-2027 (USD MILLION)
TABLE 200 BELGIUM TRAUMA DEVICES MARKET, BY PRODUCT, 2018-2027 (UNITS)
TABLE 201 BELGIUM INTERNAL FIXATOR DEVICES IN TRAUMA DEVICES MARKET, BY PRODUCT, 2018-2027 (USD MILLION)
TABLE 202 BELGIUM SCREW IN TRAUMA DEVICES MARKET, BY PRODUCT, 2018-2027 (USD MILLION)
TABLE 203 BELGIUM EXTERNAL FIXATOR DEVICES IN TRAUMA DEVICES MARKET, BYPRODUCT, 2018-2027 (USD MILLION)
TABLE 204 BELGIUM TRAUMA DEVICES MARKET, BY SURGICAL SITE, 2018-2027 (USD MILLION)
TABLE 205 BELGIUM LOWER EXTREMITIES IN TRAUMA DEVICES MARKET, BY SURGICAL SITE, 2018-2027 (USD MILLION)
TABLE 206 BELGIUM UPPER EXTREMITIES IN TRAUMA DEVICES TRAUMA DEVICES MARKET, BY SURGICAL SITE, 2018-2027 (USD MILLION)
TABLE 207 BELGIUM TRAUMA DEVICES MARKET, BY TISSUE, 2018-2027 (USD MILLION)
TABLE 208 BELGIUM TRAUMA DEVICES MARKET, BY MATERIAL, 2018-2027 (USD MILLION)
TABLE 209 BELGIUM NON-ABSORBABLE IN TRAUMA DEVICES MARKET, BY MATERIAL, 2018-2027 (USD MILLION)
TABLE 210 BELGIUM METALS IN TRAUMA DEVICES MARKET, BY MATERIAL, 2018-2027 (USD MILLION)
TABLE 211 BELGIUM CERAMICS IN TRAUMA DEVICES MARKET, BY MATERIAL, 2018-2027 (USD MILLION)
TABLE 212 BELGIUM BIO-ABSORBABLE IN TRAUMA DEVICES MARKET, BY MATERIAL, 2018-2027 (USD MILLION)
TABLE 213 BELGIUM TRAUMA DEVICES MARKET, BY PATIENT AGE, 2018-2027 (USD MILLION)
TABLE 214 BELGIUM TRAUMA DEVICES MARKET, BY END USER, 2018-2027 (USD MILLION)
TABLE 215 BELGIUM TRAUMA DEVICES MARKET, BY DISTRIBUTION CHANNEL, 2018-2027 (USD MILLION)
TABLE 216 TURKEY TRAUMA DEVICES MARKET, BY PRODUCT, 2018-2027 (USD MILLION)
TABLE 217 TURKEY TRAUMA DEVICES MARKET, BY PRODUCT, 2018-2027 (UNITS)
TABLE 218 TURKEY INTERNAL FIXATOR DEVICES IN TRAUMA DEVICES MARKET, BY PRODUCT, 2018-2027 (USD MILLION)
TABLE 219 TURKEY SCREW IN TRAUMA DEVICES MARKET, BY PRODUCT, 2018-2027 (USD MILLION)
TABLE 220 TURKEY EXTERNAL FIXATOR DEVICES IN TRAUMA DEVICES MARKET, BYPRODUCT, 2018-2027 (USD MILLION)
TABLE 221 TURKEY TRAUMA DEVICES MARKET, BY SURGICAL SITE, 2018-2027 (USD MILLION)
TABLE 222 TURKEY LOWER EXTREMITIES IN TRAUMA DEVICES MARKET, BY SURGICAL SITE, 2018-2027 (USD MILLION)
TABLE 223 TURKEY UPPER EXTREMITIES IN TRAUMA DEVICES TRAUMA DEVICES MARKET, BY SURGICAL SITE, 2018-2027 (USD MILLION)
TABLE 224 TURKEY TRAUMA DEVICES MARKET, BY TISSUE, 2018-2027 (USD MILLION)
TABLE 225 TURKEY TRAUMA DEVICES MARKET, BY MATERIAL, 2018-2027 (USD MILLION)
TABLE 226 TURKEY NON-ABSORBABLE IN TRAUMA DEVICES MARKET, BY MATERIAL, 2018-2027 (USD MILLION)
TABLE 227 TURKEY METALS IN TRAUMA DEVICES MARKET, BY MATERIAL, 2018-2027 (USD MILLION)
TABLE 228 TURKEY CERAMICS IN TRAUMA DEVICES MARKET, BY MATERIAL, 2018-2027 (USD MILLION)
TABLE 229 TURKEY BIO-ABSORBABLE IN TRAUMA DEVICES MARKET, BY MATERIAL, 2018-2027 (USD MILLION)
TABLE 230 TURKEY TRAUMA DEVICES MARKET, BY PATIENT AGE, 2018-2027 (USD MILLION)
TABLE 231 TURKEY TRAUMA DEVICES MARKET, BY END USER, 2018-2027 (USD MILLION)
TABLE 232 TURKEY TRAUMA DEVICES MARKET, BY DISTRIBUTION CHANNEL, 2018-2027 (USD MILLION)
TABLE 233 REST OF EUROPE TRAUMA DEVICES MARKET, BY PRODUCT, 2018-2027 (USD MILLION)
TABLE 234 REST OF EUROPE TRAUMA DEVICES MARKET, BY PRODUCT, 2018-2027 (UNITS)
TABLE 235 LIST OF MEDTRONIC SUBSIDIARIES
List of Figure
LIST OF FIGURES
FIGURE 1 EUROPE TRAUMA DEVICES MARKET : SEGMENTATION
FIGURE 2 EUROPE TRAUMA DEVICES MARKET : DATA TRIANGULATION
FIGURE 3 EUROPE TRAUMA DEVICES MARKET : DROC ANALYSIS
FIGURE 4 EUROPE TRAUMA DEVICES MARKET : EUROPE VS REGIONAL MARKET ANALYSIS
FIGURE 5 EUROPE TRAUMA DEVICES MARKET : COMPANY RESEARCH ANALYSIS
FIGURE 6 EUROPE TRAUMA DEVICES MARKET : INTERVIEW DEMOGRAPHICS
FIGURE 7 EUROPE TRAUMA DEVICES MARKET: DBMR MARKET POSITION GRID
FIGURE 8 EUROPE TRAUMA DEVICES MARKET: VENDOR SHARE ANALYSIS
FIGURE 9 EUROPE TRAUMA DEVICES MARKET: MARKET END-USER COVERAGE GRID
FIGURE 10 EUROPE TRAUMA DEVICES MARKET : SEGMENTATION
FIGURE 11 GROWING ADOPTION OF MINIMALLY INVASIVE SURGICAL PROCEDURES AND INCREASING PREVALENCE OF DEGENERATIVE BONE DISEASES ARE EXPECTED TO DRIVE THE EUROPE TRAUMA DEVICES MARKET IN THE FORECAST PERIOD OF 2020 TO 2027
FIGURE 12 INTERNAL FIXATOR DEVICES IS EXPECTED TO ACCOUNT FOR THE LARGEST SHARE OF THE EUROPE TRAUMA DEVICES MARKET IN 2020 & 2027
FIGURE 13 ASIA-PACIFIC IS THE MAXIMUM GROWING REGION FOR EUROPE TRAUMA DEVICES MARKET MANUFACTURERS IN THE FORECAST PERIOD OF 2020 TO 2027
FIGURE 14 DRIVERS, RESTRAINTS, OPPORTUNITIES AND CHALLENGE OF EUROPE TRAUMA DEVICES MARKET
FIGURE 15 ROAD TRAFFIC DEATH WORLDWIDE BY AGE AND SEX GROUP, IN 2002
FIGURE 16 MEDICAL DEVICES LISTED WITH FDA
FIGURE 17 EUROPE TRAUMA DEVICES MARKET: BY PRODUCT, 2019
FIGURE 18 EUROPE TRAUMA DEVICES MARKET: BY PRODUCT, 2019-2027 (USD MILLION)
FIGURE 19 EUROPE TRAUMA DEVICES MARKET: BY PRODUCT, CAGR (2020-2027)
FIGURE 20 EUROPE TRAUMA DEVICES MARKET: BY PRODUCT, LIFELINE CURVE
FIGURE 21 EUROPE TRAUMA DEVICES MARKET: BY SURGICAL SITES, 2019
FIGURE 22 EUROPE TRAUMA DEVICES MARKET: BY SURGICAL SITES, 2019-2027 (USD MILLION)
FIGURE 23 EUROPE TRAUMA DEVICES MARKET: BY SURGICAL SITES, CAGR (2020-2027)
FIGURE 24 EUROPE TRAUMA DEVICES MARKET: BY SURGICAL SITES, LIFELINE CURVE
FIGURE 25 EUROPE TRAUMA DEVICES MARKET: BY TISSUE, 2019
FIGURE 26 EUROPE TRAUMA DEVICES MARKET: BY TISSUE, 2019-2027 (USD MILLION)
FIGURE 27 EUROPE TRAUMA DEVICES MARKET: BY TISSUE, CAGR (2020-2027)
FIGURE 28 EUROPE TRAUMA DEVICES MARKET: BY TISSUE, LIFELINE CURVE
FIGURE 29 EUROPE TRAUMA DEVICES MARKET: BY MATERIAL, 2019
FIGURE 30 EUROPE TRAUMA DEVICES MARKET: BY MATERIAL, 2019-2027 (USD MILLION)
FIGURE 31 EUROPE TRAUMA DEVICES MARKET: BY MATERIAL, CAGR (2020-2027)
FIGURE 32 EUROPE TRAUMA DEVICES MARKET: BY MATERIAL, LIFELINE CURVE
FIGURE 33 EUROPE TRAUMA DEVICES MARKET: BY PATIENT AGE, 2019
FIGURE 34 EUROPE TRAUMA DEVICES MARKET, BY PATIENT AGE, 2019-2027 (USD MILLION)
FIGURE 35 EUROPE TRAUMA DEVICES MARKET: BY PATIENT AGE, CAGR (2020-2027)
FIGURE 36 EUROPE TRAUMA DEVICES MARKET: BY PATIENT AGE, LIFELINE CURVE
FIGURE 37 EUROPE TRAUMA DEVICES MARKET: BY END USER, 2019
FIGURE 38 EUROPE TRAUMA DEVICES MARKET: BY END USER, 2019-2027 (USD MILLION)
FIGURE 39 EUROPE TRAUMA DEVICES MARKET: BY END USER, CAGR (2020-2027)
FIGURE 40 EUROPE TRAUMA DEVICES MARKET: BY END USER, LIFELINE CURVE
FIGURE 41 EUROPE TRAUMA DEVICES MARKET: BY DISTRIBUTION CHANNEL, 2019
FIGURE 42 EUROPE TRAUMA DEVICES MARKET: BY DISTRIBUTION CHANNEL, 2019-2027 (USD MILLION)
FIGURE 43 EUROPE TRAUMA DEVICES MARKET: BY DISTRIBUTION CHANNEL , CAGR (2020-2027)
FIGURE 44 EUROPE TRAUMA DEVICES MARKET: BY DISTRIBUTION CHANNEL, LIFELINE CURVE
FIGURE 45 EUROPE TRAUMA DEVICES MARKET: SNAPSHOT (2019)
FIGURE 46 EUROPE TRAUMA DEVICES MARKET: BY COUNTRY (2019)
FIGURE 47 EUROPE TRAUMA DEVICES MARKET: BY COUNTRY (2020 & 2027)
FIGURE 48 EUROPE TRAUMA DEVICES MARKET: BY COUNTRY (2019 & 2027)
FIGURE 49 EUROPE TRAUMA DEVICES MARKET: BY PRODUCT (2020-2027)
FIGURE 50 EUROPE TRAUMA DEVICES MARKET: COMPANY SHARE 2019 (%)

Research Methodology
Data collection and base year analysis are done using data collection modules with large sample sizes. The stage includes obtaining market information or related data through various sources and strategies. It includes examining and planning all the data acquired from the past in advance. It likewise envelops the examination of information inconsistencies seen across different information sources. The market data is analysed and estimated using market statistical and coherent models. Also, market share analysis and key trend analysis are the major success factors in the market report. To know more, please request an analyst call or drop down your inquiry.
The key research methodology used by DBMR research team is data triangulation which involves data mining, analysis of the impact of data variables on the market and primary (industry expert) validation. Data models include Vendor Positioning Grid, Market Time Line Analysis, Market Overview and Guide, Company Positioning Grid, Patent Analysis, Pricing Analysis, Company Market Share Analysis, Standards of Measurement, Global versus Regional and Vendor Share Analysis. To know more about the research methodology, drop in an inquiry to speak to our industry experts.
Customization Available
Data Bridge Market Research is a leader in advanced formative research. We take pride in servicing our existing and new customers with data and analysis that match and suits their goal. The report can be customized to include price trend analysis of target brands understanding the market for additional countries (ask for the list of countries), clinical trial results data, literature review, refurbished market and product base analysis. Market analysis of target competitors can be analyzed from technology-based analysis to market portfolio strategies. We can add as many competitors that you require data about in the format and data style you are looking for. Our team of analysts can also provide you data in crude raw excel files pivot tables (Fact book) or can assist you in creating presentations from the data sets available in the report.