Global Brafmek Combination Therapy Market
Market Size in USD Billion
CAGR :
% 
 USD
4.21 Billion
USD
8.63 Billion
2025
2033
USD
4.21 Billion
USD
8.63 Billion
2025
2033
| 2026 - 2033 | |
| USD 4.21 Billion | |
| USD 8.63 Billion | |
|
|
|
|
BRAF/MEK Combination Therapy Market Size
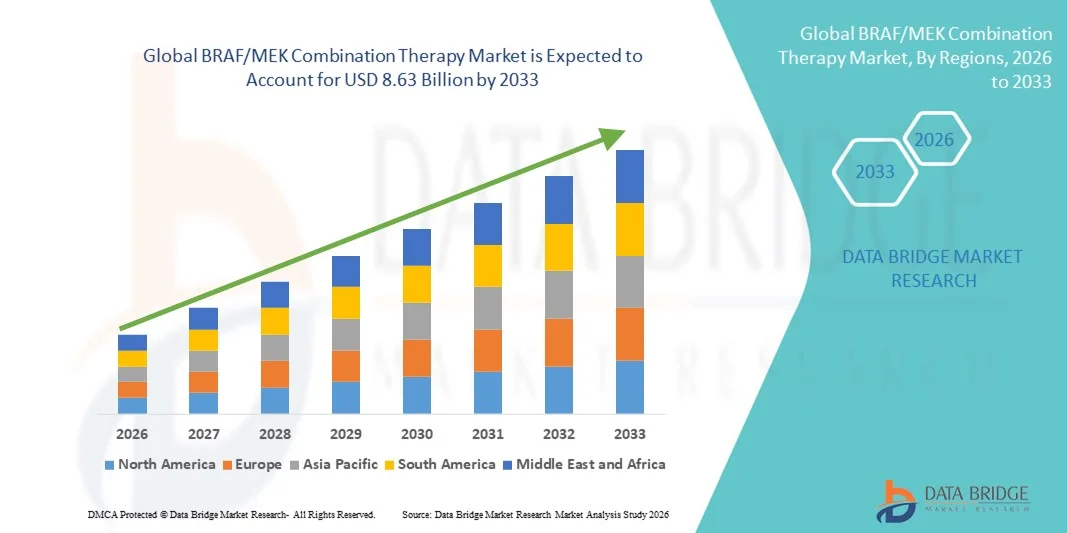
- The global BRAF/MEK combination therapy market size was valued at USD 4.21 billion in 2025 and is expected to reach USD 8.63 billion by 2033, at a CAGR of 9.40% during the forecast period
- The market growth is largely fueled by the increasing prevalence of BRAF-mutated cancers, growing adoption of targeted therapies, and advances in precision oncology that enable combination treatment approaches to improve efficacy and patient outcomes
- Furthermore, rising demand for personalized cancer treatment, favorable reimbursement policies, and ongoing clinical research supporting the synergistic effects of BRAF and MEK inhibitors are establishing BRAF/MEK combination therapies as a preferred treatment option for melanoma and other solid tumors. These converging factors are accelerating the uptake of BRAF/MEK Combination Therapy solutions, thereby significantly boosting the industry’s growth
BRAF/MEK Combination Therapy Market Analysis
- BRAF/MEK combination therapies, which target the MAPK/ERK signaling pathway in BRAF-mutated cancers, are increasingly vital in modern oncology due to their ability to improve response rates, delay resistance, and enhance overall survival in patients with melanoma, colorectal cancer, and other solid tumors
- The escalating demand for BRAF/MEK combination therapies is primarily fueled by the rising prevalence of BRAF-mutated cancers, increasing adoption of precision oncology, ongoing clinical trials demonstrating synergistic efficacy, and growing awareness among oncologists and patients regarding targeted treatment options
- North America dominated the BRAF/MEK Combination Therapy market with the largest revenue share of 38.6% in 2025, driven by high incidence of melanoma and other BRAF-mutated cancers, strong healthcare infrastructure, early adoption of targeted therapies, and robust presence of key pharmaceutical companies in the region, with the U.S. leading growth
- Asia-Pacific is expected to be the fastest-growing region in the BRAF/MEK Combination Therapy market during the forecast period, expanding at a CAGR of 10.3% from 2026 to 2033, due to increasing cancer incidence, rising awareness of precision medicine, improving healthcare infrastructure, and expanding access to novel oncology therapies in countries such as China, Japan, and India
- The Oral segment dominated the largest market revenue share of 61.3% in 2025, driven by patient preference for oral therapies, ease of administration at home, and improved compliance
Report Scope and BRAF/MEK Combination Therapy Market Segmentation
|
Attributes |
BRAF/MEK Combination Therapy Key Market Insights |
|
Segments Covered |
|
|
Countries Covered |
North America
Europe
Asia-Pacific
Middle East and Africa
South America
|
|
Key Market Players |
|
|
Market Opportunities |
|
|
Value Added Data Infosets |
In addition to the insights on market scenarios such as market value, growth rate, segmentation, geographical coverage, and major players, the market reports curated by the Data Bridge Market Research also include in-depth expert analysis, patient epidemiology, pipeline analysis, pricing analysis, and regulatory framework. |
BRAF/MEK Combination Therapy Market Trends
Enhanced Treatment Efficacy and Targeted Therapy Integration
- A significant and accelerating trend in the global BRAF/MEK Combination Therapy market is the increasing adoption of precision oncology approaches that combine BRAF and MEK inhibitors for enhanced treatment efficacy in patients with BRAF-mutated cancers
- This combination strategy is significantly improving patient outcomes by targeting multiple points in the MAPK signaling pathway, reducing the likelihood of drug resistance, and improving progression-free survival rates
- For instance, combinations such as Dabrafenib plus Trametinib and Vemurafenib plus Cobimetinib have demonstrated improved clinical outcomes in melanoma patients with BRAF V600 mutations, leading to faster adoption in oncology centers. The integration of these combination therapies with companion diagnostic testing allows clinicians to identify eligible patients accurately and tailor treatment regimens accordingly
- Furthermore, ongoing clinical trials are exploring expanded indications, including non-small cell lung cancer and colorectal cancer, which is broadening the application of BRAF/MEK combination regimens
- The trend towards personalized, combination-based cancer therapies is fundamentally reshaping oncology treatment protocols. Pharmaceutical companies are developing newer BRAF/MEK inhibitors with improved safety profiles and combination regimens that optimize therapeutic windows while minimizing adverse effects. Increasing collaboration between biotech firms, research institutions, and healthcare providers is also driving innovation in this space
- The growing focus on targeted therapy and patient-centric approaches is influencing prescribing patterns and encouraging healthcare systems to integrate molecular diagnostics and combination therapy strategies into standard care pathways
BRAF/MEK Combination Therapy Market Dynamics
Driver
Increasing Incidence of BRAF-Mutated Cancers and Improved Clinical Outcomes
- The rising prevalence of BRAF-mutated cancers, particularly melanoma, non-small cell lung cancer, and colorectal cancer, is a key driver for the growing demand for BRAF/MEK combination therapy. Patients and healthcare providers are increasingly seeking targeted therapies that offer improved efficacy compared to traditional chemotherapy
- For instance, the combination of Dabrafenib and Trametinib has demonstrated significantly improved progression-free survival in patients with BRAF V600E-mutated melanoma, according to clinical studies published by Novartis. Similarly, Vemurafenib plus Cobimetinib has shown superior response rates in metastatic melanoma patients, driving higher adoption in oncology centers
- Growing awareness among oncologists about the benefits of dual inhibition of BRAF and MEK, along with positive clinical guidelines from organizations such as NCCN and ESMO, is encouraging earlier adoption of combination therapies in treatment protocols
- The expansion of healthcare infrastructure, increasing availability of targeted therapies, and greater investment in oncology R&D are further supporting market growth. Pharmaceutical companies are actively investing in clinical trials, real-world evidence studies, and patient assistance programs to ensure accessibility and optimize treatment outcomes
- In addition, the potential for BRAF/MEK combination therapy to be used in combination with immunotherapy or other targeted treatments is opening new avenues for market expansion, as clinicians look for personalized and more effective cancer treatment strategies
Restraint/Challenge
High Treatment Costs and Safety Concerns
- Concerns regarding the high cost and potential adverse effects of BRAF/MEK combination therapy pose a significant challenge to broader market penetration
- As these therapies rely on patented drugs and require careful patient monitoring, the high price and safety management can limit adoption, especially in emerging regions and among price-sensitive patients
- For instance, branded therapies such as Dabrafenib plus Trametinib and Vemurafenib plus Cobimetinib can cost tens of thousands of dollars annually, and some patients experience severe side effects such as pyrexia, rash, fatigue, or gastrointestinal issues, leading to therapy discontinuation or dose adjustment
- Addressing these challenges through patient support programs, insurance coverage, financial assistance schemes, and improved management of adverse events is crucial for enhancing market access. Pharmaceutical companies such as Novartis and Roche are increasingly providing patient education, monitoring protocols, and cost-mitigation initiatives to facilitate therapy adoption
- While some generic and biosimilar options are entering the market, the perceived premium for combination therapy compared to single-agent treatments can still hinder widespread adoption, particularly for patients without adequate insurance coverage or in regions with limited healthcare infrastructure
- Overcoming these barriers through enhanced patient support, safety management, and cost-reduction strategies will be vital for sustained growth in the BRAF/MEK combination therapy market
BRAF/MEK Combination Therapy Market Scope
The market is segmented on the basis of drug type, indication, route of administration, and end user.
- By Drug Type
On the basis of drug type, the BRAF/MEK Combination Therapy market is segmented into BRAF Inhibitors, MEK Inhibitors, and Combination Formulations. The Combination Formulations segment dominated the largest market revenue share of 48.5% in 2025, driven by the synergistic effect of targeting both BRAF and MEK pathways, which improves treatment efficacy and delays resistance in cancer patients. Combination formulations are widely adopted in melanoma and other BRAF-mutant cancers due to higher response rates compared to monotherapies. Physicians prefer combination therapy for improved progression-free survival and better overall patient outcomes. Clinical guidelines and recommendations from oncology societies reinforce its adoption. Pharmaceutical companies are increasingly investing in new combination regimens with improved safety profiles. Regulatory approvals across developed and emerging markets have expanded accessibility. Patient adherence is enhanced through fixed-dose oral combinations. The availability of supportive patient assistance programs further strengthens its market position. Real-world evidence from large patient cohorts supports superior efficacy. Market dominance is also aided by increasing BRAF mutation testing and early diagnosis programs. Overall, combination formulations are the standard of care in BRAF/MEK-mutated cancers, securing the largest market share.
The MEK Inhibitors segment is expected to witness the fastest CAGR of 11.9% from 2026 to 2033, driven by their increasing use as adjunct therapy in BRAF-mutated cancers and expanding application in other cancer types. MEK inhibitors are increasingly used in NSCLC, colorectal, and thyroid cancers where MEK pathway mutations are identified. Development of next-generation MEK inhibitors with improved tolerability and reduced side effects supports adoption. Rising clinical trial activity globally fuels rapid growth. Increased awareness among oncologists about mutation-specific therapies accelerates utilization. Expansion of biomarker-guided therapy strategies supports precision oncology. Adoption is also fueled by the need for sequential therapy post-BRAF monotherapy resistance. Growth is further enhanced by favorable reimbursement policies and healthcare infrastructure improvements. Emerging markets show significant uptake due to growing oncology awareness. Pharmaceutical pipeline expansion ensures continuous innovation. Patient preference for targeted therapies drives faster adoption. Overall, MEK inhibitors represent the fastest-growing drug type due to expanding clinical applications and strong R&D support.
- By Indication
On the basis of indication, the market is segmented into Melanoma, Non-Small Cell Lung Cancer (NSCLC), Colorectal Cancer, Thyroid Cancer, and Other Cancers. The Melanoma segment accounted for the largest market revenue share of 52.1% in 2025, driven by the high prevalence of BRAF mutations in melanoma patients and the strong clinical evidence supporting combination therapy. Clinical guidelines recommend combination therapy as first-line treatment for BRAF-mutant melanoma. The segment benefits from increasing skin cancer incidence and growing awareness about early diagnosis. Availability of specialized oncology centers and multidisciplinary care supports widespread adoption. Oral combination formulations enable outpatient treatment and improve quality of life. Patient assistance programs and financial support schemes facilitate therapy access. Regulatory approvals across major markets enhance availability. Ongoing research demonstrates improved progression-free survival compared to monotherapies. Real-world data confirm efficacy across diverse patient populations. Pharmaceutical companies continue to launch optimized combination regimens. Hospitals and oncology clinics are the primary points of administration. Marketing campaigns targeting physicians strengthen adoption further.
The NSCLC segment is expected to witness the fastest CAGR of 12.4% from 2026 to 2033, fueled by the rising incidence of BRAF-mutant NSCLC and the growing use of precision medicine in lung cancer therapy. Combination therapy improves patient response rates and delays acquired resistance in NSCLC. Adoption is supported by increased molecular testing for BRAF/MEK mutations. Expansion of targeted therapy awareness among oncologists accelerates uptake. Clinical trials continue to demonstrate efficacy in diverse patient cohorts. Development of next-generation inhibitors enhances tolerability and safety. Health authorities and insurance reimbursement policies in developed regions promote adoption. Emerging markets show significant growth due to increased oncology infrastructure. Outpatient treatment options and oral therapy preference further boost utilization. Pharmaceutical companies are actively investing in NSCLC-focused therapy programs. Overall, NSCLC represents the fastest-growing indication segment due to precision-targeted therapies and expanding patient populations.
- By Route of Administration
On the basis of route of administration, the market is segmented into Oral and Injectable. The Oral segment dominated the largest market revenue share of 61.3% in 2025, driven by patient preference for oral therapies, ease of administration at home, and improved compliance. Oral BRAF/MEK inhibitors reduce hospital visits and support long-term outpatient therapy. Fixed-dose oral combination regimens simplify treatment and enhance adherence. Pharmaceutical companies focus on convenient oral formulations with optimized dosing schedules. Oral therapy allows for continuous treatment without hospitalization. Guidelines recommend oral regimens for most BRAF/MEK-mutated cancers. Patients and caregivers prefer oral over injectable administration due to convenience and comfort. Oral therapies enable broader adoption in emerging markets with limited hospital infrastructure. Real-world data confirm sustained efficacy and tolerability of oral regimens. Patient assistance programs further increase accessibility. Healthcare providers prioritize oral options for better workflow and resource management.
The Injectable segment is expected to witness the fastest CAGR of 10.7% from 2026 to 2033, driven by novel formulations for targeted delivery, reduced systemic toxicity, and hospital-based use in advanced cancers. Injectable therapies are often used in specialized care settings and clinical trials. Development of next-generation injectable inhibitors supports faster adoption. Hospitals and oncology centers provide monitoring for safety and efficacy. Injectable formulations address patients with absorption issues or specific treatment protocols. Investment in oncology infrastructure accelerates market penetration. Growing clinical evidence supports their efficacy in combination with other therapies. Regulatory approvals and guidelines facilitate broader adoption. Emerging markets are witnessing increasing adoption of injectable targeted therapies. Hospitals leverage injectables for controlled and precise dosing. Overall, injectables represent the fastest-growing route of administration due to targeted delivery and specialized care adoption.
- By End User
On the basis of end user, the market is segmented into Hospitals, Oncology Clinics, Ambulatory Care Centers, and Research & Academic Institutes. The Hospitals segment held the largest market revenue share of 56.8% in 2025, due to high patient volumes, comprehensive oncology services, and availability of multidisciplinary care for administering combination therapy. Hospitals offer integrated services including diagnostics, treatment, and follow-up. Advanced hospital infrastructure supports both oral and injectable administration. Presence of experienced oncology specialists ensures proper therapy management. Hospitals are key in early adoption of newly approved therapies. Multispecialty collaboration enables higher patient coverage. Patient support programs and adherence monitoring are implemented effectively in hospital settings. Hospitals provide specialized outpatient and inpatient facilities. Financial reimbursement support is more accessible in hospitals. Hospitals remain the primary point of care for BRAF/MEK therapy due to capacity, infrastructure, and treatment expertise. Pharmaceutical companies focus marketing efforts on hospitals to maximize penetration.
The Ambulatory Care Centers segment is expected to witness the fastest CAGR of 11.6% from 2026 to 2033, fueled by the trend of outpatient management of stable cancer patients and preference for cost-effective care. Ambulatory centers provide convenient access to oral therapies and routine monitoring. The shift from inpatient to outpatient treatment drives adoption. Ambulatory care allows reduced hospital stay and lower treatment costs. Centers are expanding services for molecular testing and follow-up care. Patient convenience and improved workflow enhance therapy compliance. Growth is further supported by regulatory encouragement for outpatient oncology care. Centers adopt advanced oral therapy regimens to manage multiple patients efficiently. The segment benefits from rising awareness of precision medicine and mutation-targeted treatments. Pharmaceutical companies target ambulatory centers for new therapy launches. Overall, ambulatory care centers represent the fastest-growing end-user segment due to convenience, cost-effectiveness, and outpatient treatment trends.
BRAF/MEK Combination Therapy Market Regional Analysis
- North America dominated the BRAF/MEK combination therapy market with the largest revenue share of 38.6% in 2025, driven by a high incidence of melanoma and other BRAF-mutated cancers. The region benefits from strong healthcare infrastructure, early adoption of targeted therapies, and the robust presence of key pharmaceutical companies. The U.S. led the market growth, fueled by awareness of precision oncology, increasing R&D investments, and the adoption of novel combination therapies
- Advanced diagnostic capabilities, early cancer screening programs, and supportive reimbursement policies further strengthen the region’s position. Patients and healthcare providers increasingly prefer targeted BRAF/MEK therapies due to their improved efficacy and tolerability. The adoption of combination therapy in clinical guidelines, alongside rising oncology research initiatives, continues to drive market expansion
- Technological advancements in drug formulation and personalized treatment approaches also contribute to market dominance. Ongoing clinical trials in melanoma and NSCLC, coupled with patient advocacy efforts, enhance adoption. The North American market also sees increasing collaboration between pharmaceutical companies and oncology centers. The U.S. market captured a significant portion of this revenue, accounting for the largest share within the region
U.S. BRAF/MEK Combination Therapy Market Insight
The U.S. BRAF/MEK combination therapy market captured the largest revenue share in 2025 within North America, driven by early adoption of advanced oncology treatments and the availability of well-established cancer care centers. Rapid adoption of precision medicine, strong clinical trial activity, and patient awareness of targeted therapy benefits propel market growth. The integration of BRAF/MEK therapy into standard-of-care treatment guidelines for melanoma, NSCLC, and other BRAF-mutated cancers further drives uptake. High healthcare expenditure, favorable insurance coverage, and access to novel combination drugs enhance accessibility. The presence of leading pharmaceutical manufacturers ensures continuous product innovation. In addition, increasing collaboration between hospitals, research institutes, and biotech companies supports clinical adoption. Growing preference for outpatient treatment and combination therapy protocols adds to the demand. Patient-centric treatment approaches, along with physician familiarity with BRAF/MEK regimens, strengthen market penetration.
Europe BRAF/MEK Combination Therapy Market Insight
The Europe BRAF/MEK combination therapy market is projected to expand at a substantial CAGR during the forecast period, driven by rising incidence of melanoma and targeted therapy adoption. Stringent regulatory frameworks ensure the availability of approved BRAF/MEK combination regimens, while healthcare infrastructure improvements enhance access to specialized oncology centers. Countries like Germany, France, and Italy are witnessing steady growth due to high patient awareness and physician adoption of evidence-based therapies. Early detection programs, oncology awareness campaigns, and the presence of key pharmaceutical players support market expansion. European healthcare systems increasingly prioritize personalized medicine, further promoting combination therapy adoption. Hospitals and specialized cancer centers are central to driving treatment uptake, with reimbursement support facilitating patient access. Real-world clinical data demonstrating efficacy in BRAF-mutated cancers enhances confidence among clinicians.
U.K. BRAF/MEK Combination Therapy Market Insight
The U.K. BRAF/MEK combination therapy market is anticipated to grow at a noteworthy CAGR, supported by rising melanoma incidence and early adoption of precision oncology. The NHS and private hospitals are increasingly integrating BRAF/MEK combination therapies into treatment protocols. Awareness campaigns for skin cancer, coupled with effective diagnostic programs, drive patient demand. The country benefits from a well-developed oncology infrastructure, strong R&D activities, and collaborations between pharmaceutical companies and academic research centers. Adoption of targeted therapies is also accelerated by clinical guideline recommendations. The U.K. market sees rising preference for outpatient combination therapy administration, alongside increasing enrollment in clinical trials for novel indications. E-commerce and specialty pharmacy availability contribute to improved access, particularly in urban centers.
Germany BRAF/MEK Combination Therapy Market Insight
The Germany BRAF/MEK combination therapy market is expected to expand at a considerable CAGR during the forecast period, driven by increasing awareness of melanoma and other BRAF-mutated cancers. Germany’s robust healthcare system ensures high accessibility to targeted therapies. Government support, reimbursement policies, and well-established oncology centers further contribute to market growth. Physicians are adopting BRAF/MEK combination therapy based on strong clinical evidence, while ongoing clinical trials boost awareness. Patients increasingly prefer outpatient administration in specialized cancer care facilities. Adoption is supported by collaborations between biotech firms, hospitals, and academic institutions. Focus on personalized medicine, along with patient advocacy programs, enhances therapy penetration.
Asia-Pacific BRAF/MEK Combination Therapy Market Insight
The Asia-Pacific BRAF/MEK combination therapy market is expected to be the fastest-growing region during the forecast period, expanding at a CAGR of 10.3% from 2026 to 2033. Increasing cancer incidence, rising awareness of precision medicine, improving healthcare infrastructure, and expanding access to novel oncology therapies in countries such as China, Japan, and India are key growth drivers. Urbanization, government health initiatives, and growing private healthcare investments further boost adoption. Availability of skilled oncology specialists and improved diagnostic capabilities facilitate early therapy initiation. Cost-effective therapy options and increasing insurance coverage improve market penetration. Regional collaborations between pharmaceutical companies and hospitals support clinical trial enrollment. Adoption in private and tertiary care hospitals is expanding, while government-supported cancer programs enhance access. Rising patient education and awareness of targeted therapy benefits further drive growth.
Japan BRAF/MEK Combination Therapy Market Insight
The Japan BRAF/MEK combination therapy market is gaining momentum due to a high prevalence of melanoma, a tech-savvy population, and the country’s focus on advanced oncology care. Healthcare providers emphasize precision oncology, leading to increased adoption of BRAF/MEK combination therapy. Strong clinical trial activity, supportive insurance schemes, and hospital infrastructure enhance accessibility. Aging population trends increase demand for safe, effective, and convenient outpatient therapies. Integration with digital health platforms facilitates patient monitoring and adherence. Growing awareness among oncologists and patients ensures sustained uptake.
China BRAF/MEK Combination Therapy Market Insight
The China BRAF/MEK combination therapy market accounted for the largest market revenue share in Asia-Pacific in 2025, driven by rising cancer prevalence, rapid urbanization, and growing adoption of precision medicine. Increased access to modern oncology centers, government initiatives promoting cancer care, and affordability of BRAF/MEK combination therapies further stimulate market growth. China’s expanding middle class, widespread awareness programs, and local pharmaceutical production enhance availability. The push toward smart healthcare and hospital digitalization supports rapid therapy adoption. Strong hospital networks and ongoing clinical trials further fuel market expansion.
BRAF/MEK Combination Therapy Market Share
The BRAF/MEK Combination Therapy industry is primarily led by well-established companies, including:
- Novartis AG (Switzerland)
- GlaxoSmithKline plc (U.K.)
- Daiichi Sankyo Company, Limited (Japan)
- Roche Holding AG (Switzerland)
- Pfizer Inc. (U.S.)
- Amgen Inc. (U.S.)
- Bristol-Myers Squibb Company (U.S.)
- Merck & Co., Inc. (U.S.)
- Eli Lilly and Company (U.S.)
- Sanofi S.A. (France)
- AstraZeneca plc (U.K.)
- Array BioPharma, Inc. (U.S.)
- Pierre Fabre Group (France)
- Incyte Corporation (U.S.)
- Beacon Pharmaceuticals Ltd. (Bangladesh)
- Hanmi Pharmaceutical Co., Ltd. (South Korea)
- BeiGene, Ltd. (China)
- Symphogen A/S (Denmark)
- Genentech, Inc. (U.S.)
- Medivir AB (Sweden)
Latest Developments in Global BRAF/MEK Combination Therapy Market
- In October 2023, Pfizer’s BRAFTOVI (encorafenib) + MEKTOVI (binimetinib) combination therapy was approved by the U.S. FDA for the treatment of adult patients with metastatic non‑small cell lung cancer (NSCLC) harboring a BRAF V600E mutation, providing a new targeted precision medicine option beyond melanoma indications and expanding the clinical reach of BRAF/MEK combinations. This approval was based on data from the PHAROS Phase 2 trial showing significant objective response rates and duration of response in treated patients
- In April 2023, regulatory bodies updated approvals for dabrafenib + trametinib (Tafinlar + Mekinist) to include pediatric indications for low‑grade gliomas in patients with BRAF V600E mutations, expanding established BRAF/MEK combinations into new age groups and cancer types
- In September 2024, at the European Society for Medical Oncology (ESMO) Congress 2024, updated clinical findings confirmed the benefit of BRAF/MEK inhibitor combinations (such as dabrafenib + trametinib and encorafenib + binimetinib) in BRAF V600E‑mutated non‑small cell lung cancer, reaffirming the therapeutic value of BRAF/MEK combinations beyond melanoma indications
- In December 2024, the U.S. FDA granted accelerated approval to encorafenib in combination with cetuximab and mFOLFOX6 for metastatic colorectal cancer (mCRC) with a BRAF V600E mutation, supporting the role of BRAF/MEK combinations (with additional targeted agents) as effective first‑line regimens in hard‑to‑treat cancers
- In March 2025, the EORTC‑2139‑MG/Columbus‑AD Phase 3 trial results for encorafenib + binimetinib in high‑risk stage IIB/C BRAF V600‑mutant melanoma showed that the combination improved 12‑month recurrence‑free survival compared to placebo and was well‑tolerated. These data were presented at the 2025 ASCO Annual Meeting, highlighting the expanded use of BRAF/MEK combinations into earlier melanoma disease settings
- In March 2025, Pfizer announced updated survival data from the Phase 3 BREAKWATER trial, reporting that the BRAFTOVI combination regimen (encorafenib with cetuximab and mFOLFOX6) reduced the risk of death by 51% and disease progression or death by 47% compared to standard chemotherapy regimens in patients with BRAF V600E‑mutant metastatic colorectal cancer. These practice‑changing results were presented at the 2025 ASCO Annual Meeting and published in leading oncology outlets, underscoring the efficacy of advanced BRAF/MEK‑based combinations in solid tumors
SKU-
Get online access to the report on the World's First Market Intelligence Cloud
- Interactive Data Analysis Dashboard
- Company Analysis Dashboard for high growth potential opportunities
- Research Analyst Access for customization & queries
- Competitor Analysis with Interactive dashboard
- Latest News, Updates & Trend analysis
- Harness the Power of Benchmark Analysis for Comprehensive Competitor Tracking
Research Methodology
Data collection and base year analysis are done using data collection modules with large sample sizes. The stage includes obtaining market information or related data through various sources and strategies. It includes examining and planning all the data acquired from the past in advance. It likewise envelops the examination of information inconsistencies seen across different information sources. The market data is analysed and estimated using market statistical and coherent models. Also, market share analysis and key trend analysis are the major success factors in the market report. To know more, please request an analyst call or drop down your inquiry.
The key research methodology used by DBMR research team is data triangulation which involves data mining, analysis of the impact of data variables on the market and primary (industry expert) validation. Data models include Vendor Positioning Grid, Market Time Line Analysis, Market Overview and Guide, Company Positioning Grid, Patent Analysis, Pricing Analysis, Company Market Share Analysis, Standards of Measurement, Global versus Regional and Vendor Share Analysis. To know more about the research methodology, drop in an inquiry to speak to our industry experts.
Customization Available
Data Bridge Market Research is a leader in advanced formative research. We take pride in servicing our existing and new customers with data and analysis that match and suits their goal. The report can be customized to include price trend analysis of target brands understanding the market for additional countries (ask for the list of countries), clinical trial results data, literature review, refurbished market and product base analysis. Market analysis of target competitors can be analyzed from technology-based analysis to market portfolio strategies. We can add as many competitors that you require data about in the format and data style you are looking for. Our team of analysts can also provide you data in crude raw excel files pivot tables (Fact book) or can assist you in creating presentations from the data sets available in the report.