Global Cell Therapy Manufacturing Platforms Market
Market Size in USD Billion
CAGR :
% 
 USD
3.48 Billion
USD
11.97 Billion
2025
2033
USD
3.48 Billion
USD
11.97 Billion
2025
2033
| 2026 - 2033 | |
| USD 3.48 Billion | |
| USD 11.97 Billion | |
|
|
|
|
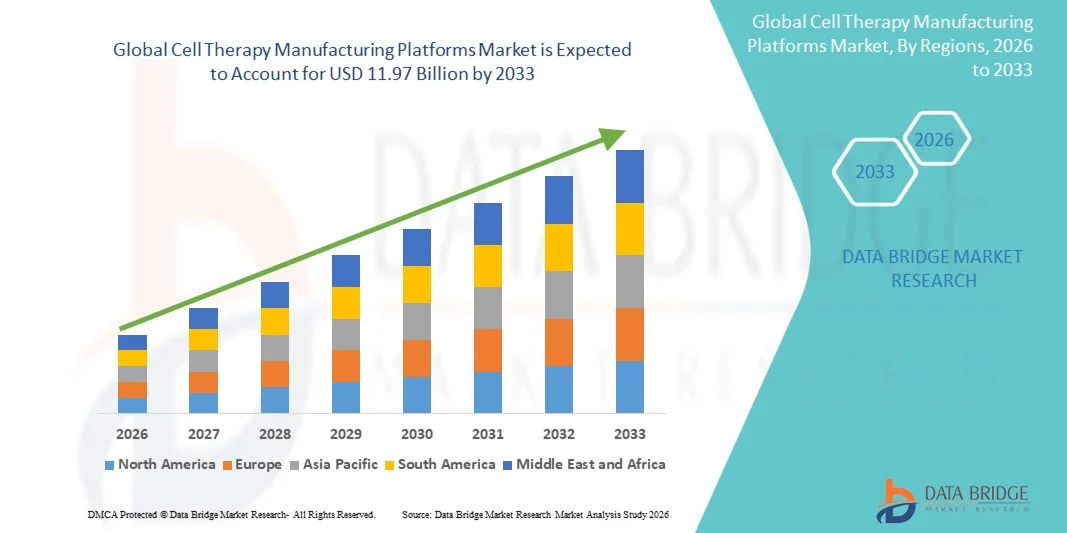
Cell Therapy Manufacturing Platforms Market Size
- The global Cell Therapy Manufacturing Platforms market size was valued at USD 3.48 billion in 2025and is expected to reach USD 11.97 billion by 2033, at a CAGR of 16.70% during the forecast period
- The market growth is largely fueled by the increasing adoption of advanced bioprocessing technologies and automation in biopharmaceutical manufacturing, leading to improved scalability, efficiency, and consistency in cell-based therapy production across research and commercial settings
- Furthermore, rising demand for personalized medicine, growing investment in regenerative medicine research, and expanding clinical applications of cell therapies are establishing automated and scalable production systems as the preferred manufacturing approach for Cell Therapy Manufacturing Platforms. These converging factors are accelerating the uptake of cell therapy manufacturing platforms, thereby significantly boosting the industry's growth
Cell Therapy Manufacturing Platforms Market Analysis
- Cell Therapy Manufacturing Platforms, offering automated, scalable, and closed-system bioprocess solutions for the production of advanced cell-based therapies, are increasingly vital components of modern biopharmaceutical manufacturing systems due to their enhanced efficiency, reduced contamination risk, and seamless integration with digital bioprocess control technologies
- The escalating demand for Cell Therapy Manufacturing Platforms is primarily fueled by the rapid growth of regenerative medicine, increasing clinical adoption of cell and gene therapies, and rising need for scalable, cost-efficient, and standardized biomanufacturing solutions
- North America dominated the cell therapy manufacturing platforms market with the largest revenue share of 41.6% in 2025, characterized by strong biopharmaceutical infrastructure, early adoption of advanced manufacturing technologies, and a high concentration of leading biotech and pharmaceutical companies, with the U.S. experiencing substantial growth in automated cell therapy manufacturing facilities driven by increasing clinical trials, regulatory approvals, and expansion of GMP-compliant production capacity
- Asia-Pacific is expected to be the fastest growing region in the cell therapy manufacturing platforms market during the forecast period due to increasing investments in biotechnology, rising government support for regenerative medicine, expanding clinical research activities, and growing presence of contract manufacturing organizations (CMOs)
- The oncology segment dominated the largest market revenue share of 52.3% in 2025, driven by the rapid adoption of cell-based immunotherapies such as CAR-T cell therapy for cancer treatment
Report Scope and Cell Therapy Manufacturing Platforms Market Segmentation
|
Attributes |
Cell Therapy Manufacturing Platforms Key Market Insights |
|
Segments Covered |
|
|
Countries Covered |
North America · U.S. · Canada · Mexico Europe · Germany · France · U.K. · Netherlands · Switzerland · Belgium · Russia · Italy · Spain · Turkey · Rest of Europe Asia-Pacific · China · Japan · India · South Korea · Singapore · Malaysia · Australia · Thailand · Indonesia · Philippines · Rest of Asia-Pacific Middle East and Africa · Saudi Arabia · U.A.E. · South Africa · Egypt · Israel · Rest of Middle East and Africa South America · Brazil · Argentina · Rest of South America |
|
Key Market Players |
· Thermo Fisher Scientific Inc. (U.S.) · Danaher Corporation (U.S.) · Merck KGaA (Germany) · Sartorius AG (Germany) · Lonza Group AG (Switzerland) · Miltenyi Biotec (Germany) · GE HealthCare (U.S.) · BD (U.S.) · Fresenius Kabi AG (Germany) · Charles River Laboratories (U.S.) · STEMCELL Technologies Inc. (Canada) · CellGenix GmbH (Germany) · Terumo Corporation (Japan) · Hitachi High-Tech Corporation (Japan) · Repligen Corporation (U.S.) · Catalent, Inc. (U.S.) · WuXi AppTec (China) · Fujifilm Holdings Corporation (Japan) · Beckman Coulter (Danaher) (U.S.) · Bio-Techne Corporation (U.S.) |
|
Market Opportunities |
· Rising adoption of automated, closed-system bioprocessing and AI-driven manufacturing optimization · Rising Demand in Emerging Markets |
|
Value Added Data Infosets |
In addition to the insights on market scenarios such as market value, growth rate, segmentation, geographical coverage, and major players, the market reports curated by the Data Bridge Market Research also include in-depth expert analysis, patient epidemiology, pipeline analysis, pricing analysis, and regulatory framework. |
Cell Therapy Manufacturing Platforms Market Trends
“Rising Adoption of Automated, Closed-System and Digitally Integrated Manufacturing Platforms”
- A significant and accelerating trend in the global Cell Therapy Manufacturing Platforms market is the shift toward automated, closed-system, and digitally integrated manufacturing solutions aimed at improving scalability, consistency, and regulatory compliance in advanced therapy production
- Biopharmaceutical and biotechnology companies are increasingly adopting modular and automated manufacturing platforms to streamline complex cell therapy workflows, including cell isolation, expansion, genetic modification, and final product formulation
- For instance, closed-system bioreactors and automated cell processing systems are being widely used to reduce contamination risks, improve batch-to-batch consistency, and enhance overall manufacturing efficiency for therapies such as CAR-T cell therapy and stem cell-based treatments
- The integration of digital technologies such as real-time monitoring, process analytics, and data-driven manufacturing controls is enabling better process optimization, quality assurance, and regulatory traceability across production stages
- Furthermore, advancements in single-use technologies and scalable manufacturing systems are significantly reducing production time and operational complexity while supporting flexible and cost-efficient manufacturing models
- The increasing focus on decentralized manufacturing models, including point-of-care and near-patient production facilities, is also transforming the traditional centralized production approach in the cell therapy industry
- This trend toward automation, digitalization, and scalable manufacturing infrastructure is significantly improving efficiency, reducing costs, and accelerating the commercialization of advanced cell therapies globally
Cell Therapy Manufacturing Platforms Market Dynamics
Driver
“Growing Need Due to Rising Demand for Advanced Cell and Gene Therapies”
- The increasing prevalence of cancer, genetic disorders, and chronic diseases is a major factor driving the growth of the global Cell Therapy Manufacturing Platforms market
- Rising demand for advanced therapies such as CAR-T cell therapies, stem cell therapies, and regenerative medicine solutions is significantly increasing the need for efficient, scalable, and standardized manufacturing platforms
- For instance, the expanding number of approved cell-based therapies and ongoing clinical trials across oncology and rare diseases are driving pharmaceutical companies to invest heavily in advanced manufacturing infrastructure
- Furthermore, increasing government funding, regulatory support for advanced therapy medicinal products (ATMPs), and growing investments in biopharmaceutical R&D are accelerating market expansion
- The complexity of cell therapy production, which requires highly controlled environments and precise manufacturing processes, is also driving the adoption of specialized platforms to ensure product safety and efficacy
- In addition, the rising trend of personalized medicine is increasing demand for patient-specific therapies, further boosting the need for flexible and scalable manufacturing solutions
- Expanding collaborations between biotech companies, contract development and manufacturing organizations (CDMOs), and research institutes are further supporting innovation and capacity expansion in the market
Restraint/Challenge
“High Manufacturing Costs and Complex Regulatory Requirements”
- The high cost associated with cell therapy manufacturing infrastructure, equipment, and operational processes remains a significant challenge restraining the growth of the Cell Therapy Manufacturing Platforms market
- The production of cell-based therapies requires specialized facilities, highly skilled personnel, and stringent quality control measures, all of which contribute to elevated manufacturing costs
- For instance, maintaining GMP-compliant facilities and implementing advanced automation systems can significantly increase capital expenditure for biopharmaceutical companies and manufacturing organizations
- In addition, complex regulatory requirements for cell and gene therapies, including strict approval pathways, documentation standards, and quality assurance protocols, can delay product development and commercialization timelines
- The variability of biological starting materials also adds complexity to manufacturing processes, leading to challenges in achieving consistent product quality and large-scale production efficiency
- Furthermore, limited manufacturing capacity and supply chain constraints for specialized raw materials and consumables can hinder production scalability
- Addressing these challenges through cost-efficient manufacturing technologies, regulatory harmonization, workforce training, and expansion of global production capacity will be essential for sustaining long-term growth in the Cell Therapy Manufacturing Platforms market
Cell Therapy Manufacturing Platforms Market Scope
The market is segmented on the basis of product type and application.
- By Product Type
On the basis of product type, the Cell Therapy Manufacturing Platforms market is segmented into consumables & reagents, equipment, and software & services. The consumables & reagents segment dominated the largest market revenue share of 47.6% in 2025, driven by their recurring usage in every stage of cell therapy production, including cell culture, processing, and expansion. These products are essential for ensuring consistency, sterility, and scalability in manufacturing workflows. Rising demand for personalized medicine and increasing clinical trials in cell-based therapies are significantly supporting segment growth. In addition, growing adoption of advanced culture media, cytokines, and growth factors is strengthening market expansion. Pharmaceutical and biotechnology companies heavily rely on consumables for continuous production processes, further reinforcing segment dominance. Expanding investment in regenerative medicine and increasing commercialization of CAR-T and stem cell therapies are also contributing to sustained demand globally.
The software & services segment is anticipated to witness the fastest growth rate of 22.8% from 2026 to 2033, driven by increasing adoption of digital manufacturing platforms, automation, and data-driven process optimization. Advanced software solutions enable real-time monitoring, quality control, and regulatory compliance in cell therapy production. Growing integration of artificial intelligence, machine learning, and cloud-based platforms is significantly enhancing manufacturing efficiency. In addition, rising demand for scalable and standardized production systems is accelerating adoption of end-to-end service solutions. Increasing outsourcing of manufacturing processes to specialized CDMOs (Contract Development and Manufacturing Organizations) is further boosting segment growth. The shift toward smart biomanufacturing and digital transformation in life sciences is expected to strongly drive market expansion.
- By Application
On the basis of application, the Cell Therapy Manufacturing Platforms market is segmented into oncology, cardiovascular diseases, musculoskeletal disorders, neurological disorders, and others. The oncology segment dominated the largest market revenue share of 52.3% in 2025, driven by the rapid adoption of cell-based immunotherapies such as CAR-T cell therapy for cancer treatment. Increasing global cancer prevalence and rising demand for targeted and personalized treatment approaches are significantly contributing to segment growth. Cell therapy has shown high efficacy in treating hematological malignancies and is increasingly being explored for solid tumors. Expanding clinical trials and regulatory approvals for advanced oncology therapies are further supporting market expansion. In addition, strong investments from pharmaceutical companies and biotechnology firms in cancer immunotherapy research are strengthening segment dominance. Growing awareness of personalized cancer treatment and increasing healthcare expenditure are also boosting demand globally.
The neurological disorders segment is expected to witness the fastest CAGR of 23.4% from 2026 to 2033, driven by increasing research into cell-based therapies for conditions such as Parkinson’s disease, Alzheimer’s disease, and spinal cord injuries. Advancements in regenerative medicine and stem cell research are significantly improving treatment potential for neurodegenerative diseases. Rising prevalence of neurological disorders due to aging populations is further accelerating demand. In addition, ongoing clinical trials and breakthroughs in neuronal cell regeneration are supporting segment growth. Increasing funding for neuroscience research and growing focus on innovative therapeutic approaches are expected to drive strong market expansion during the forecast period.
Cell Therapy Manufacturing Platforms Market Regional Analysis
- North America dominated the cell therapy manufacturing platforms market with the largest revenue share of 41.6% in 2025, characterized by advanced biopharmaceutical infrastructure, early adoption of automated manufacturing technologies, and a strong presence of leading biotechnology firms. The region continues to lead global innovation in cell and gene therapy manufacturing, supported by robust investments in scalable and GMP-compliant production platforms
- The market is further driven by increasing clinical trial activity, rising regulatory approvals for cell and gene therapies, and the rapid expansion of commercial-scale manufacturing facilities, particularly in the United States. In addition, strong collaboration between biotech companies, research institutes, and contract manufacturing organizations is accelerating the development of next-generation cell therapy production systems
- The presence of well-established biomanufacturing hubs, high healthcare R&D spending, and continuous technological advancements in automated and closed-system manufacturing platforms continues to reinforce North America’s dominance in the Cell Therapy Manufacturing Platforms market
U.S. Cell Therapy Manufacturing Platforms Market Insight
The U.S. cell therapy manufacturing platforms market captured the largest revenue share in 2025 within North America, driven by rapid expansion of cell therapy pipelines and increasing demand for scalable manufacturing solutions. The country is witnessing substantial growth in cell therapy production facilities, supported by rising clinical trials, accelerating regulatory approvals, and strong investment in regenerative medicine. Furthermore, increasing adoption of automated bioprocessing technologies and expansion of GMP-compliant manufacturing infrastructure are significantly contributing to market growth in the U.S.
Europe Cell Therapy Manufacturing Platforms Market Insight
The Europe cell therapy manufacturing platforms market is projected to expand at a substantial CAGR throughout the forecast period, primarily driven by growing investments in advanced biomanufacturing infrastructure and increasing focus on regenerative medicine. The region benefits from strong regulatory frameworks supporting cell and gene therapy development, along with rising collaboration between academic institutions and biotech companies. Additionally, expanding clinical research activity and increasing demand for scalable manufacturing solutions are supporting market growth across Europe.
U.K. Cell Therapy Manufacturing Platforms Market Insight
The U.K. cell therapy manufacturing platforms market is anticipated to grow at a noteworthy CAGR during the forecast period, driven by strong government support for advanced therapies and increasing investment in biotechnology innovation. The country’s growing clinical trial ecosystem and expanding cell therapy research initiatives are supporting demand for advanced manufacturing platforms. Moreover, rising collaborations between academic research centers and biopharmaceutical companies are accelerating market development in the U.K.
Germany Cell Therapy Manufacturing Platforms Market Insight
The Germany cell therapy manufacturing platforms market is expected to expand at a considerable CAGR during the forecast period, fueled by strong pharmaceutical manufacturing capabilities and increasing focus on biotechnology innovation. Germany’s well-established biopharma industry and growing adoption of automated and digitalized manufacturing systems are supporting market growth. Additionally, rising investment in GMP-compliant facilities and increasing research in regenerative medicine are further enhancing industry expansion.
Asia-Pacific Cell Therapy Manufacturing Platforms Market Insight
The Asia-Pacific cell therapy manufacturing platforms market is expected to be the fastest growing region during the forecast period due to rising investments in biotechnology, expanding clinical research activities, increasing government support for regenerative medicine, and growing establishment of contract manufacturing organizations (CMOs). Rapid development of biopharmaceutical infrastructure in countries such as China, Japan, South Korea, and India is enabling large-scale adoption of advanced manufacturing platforms. Additionally, increasing demand for cost-effective production and growing participation in global clinical trials are driving strong regional growth.
Japan Cell Therapy Manufacturing Platforms Market Insight
The Japan cell therapy manufacturing platforms market is gaining momentum due to the country’s advanced healthcare system, strong focus on regenerative medicine, and increasing approvals of cell-based therapies. Japan’s emphasis on automated and closed-system manufacturing technologies is supporting efficient production of advanced therapies. Moreover, growing investments in clinical research and increasing collaboration between academia and industry are contributing to market expansion.
China Cell Therapy Manufacturing Platforms Market Insight
The China cell therapy manufacturing platforms market accounted for the largest market revenue share in Asia-Pacific in 2025, attributed to rapid expansion of biotechnology infrastructure, increasing government support for regenerative medicine, and rising investment in advanced manufacturing facilities. China is witnessing strong growth in contract manufacturing organizations and cell therapy production capacity, supported by increasing clinical trials and expanding domestic biotech capabilities. In addition, favorable regulatory reforms and growing focus on innovation in biologics manufacturing are key factors propelling market growth in the country.
Cell Therapy Manufacturing Platforms Market Share
The Cell Therapy Manufacturing Platforms industry is primarily led by well-established companies, including:
- Thermo Fisher Scientific Inc. (U.S.)
- Danaher Corporation (U.S.)
- Merck KGaA (Germany)
- Sartorius AG (Germany)
- Lonza Group AG (Switzerland)
- Miltenyi Biotec (Germany)
- GE HealthCare (U.S.)
- BD (U.S.)
- Fresenius Kabi AG (Germany)
- Charles River Laboratories (U.S.)
- STEMCELL Technologies Inc. (Canada)
- CellGenix GmbH (Germany)
- Terumo Corporation (Japan)
- Hitachi High-Tech Corporation (Japan)
- Repligen Corporation (U.S.)
- Catalent, Inc. (U.S.)
- WuXi AppTec (China)
- Fujifilm Holdings Corporation (Japan)
- Beckman Coulter (Danaher) (U.S.)
- Bio-Techne Corporation (U.S.)
Latest Developments in Global Cell Therapy Manufacturing Platforms Market
- In February 2021, Lonza Group announced expansion of its Cocoon® closed automated cell therapy manufacturing platform, strengthening decentralized and scalable CAR-T manufacturing capabilities. The Cocoon system enabled fully closed, patient-scale cell therapy production, reducing contamination risks and improving GMP compliance for commercial cell therapy workflows
- In October 2023, Sartorius AG and Cytiva expanded their single-use bioprocessing and modular cell therapy manufacturing platforms, integrating automation, digital monitoring, and closed-system workflows to improve scalability and reduce manual intervention in GMP cell therapy production environments
- In January 2024, Thermo Fisher Scientific launched Gibco CTS Cellmation Software, an off-the-shelf validated automation platform designed to digitally connect cell therapy instruments, reduce manual steps, and ensure GMP-compliant manufacturing workflows for CAR-T and other advanced therapies
- In February 2024, Thermo Fisher Scientific expanded its collaboration with Multiply Labs, integrating robotics with cell therapy manufacturing systems to achieve fully automated GMP-compliant production workflows, significantly reducing labor dependency and increasing scalability in cell therapy manufacturing platforms
- In June 2024, Cytiva launched the SEFIA cell therapy manufacturing platform, designed to accelerate production of CAR-T and other cell-based therapies using higher automation, closed processing, and cost-efficient manufacturing architecture
- In 2024, Lonza expanded its viral vector and cell therapy manufacturing capacity in the U.S. and Europe, enhancing large-scale production capabilities for allogeneic and autologous cell therapies using automated, closed-system manufacturing suites
- In January 2025, Thermo Fisher Scientific introduced next-generation closed-loop bioreactor systems for T-cell expansion, integrating inline analytics and automated process control for GMP-grade cell therapy manufacturing
- In April 2025, Miltenyi Biotec launched CliniMACS Prodigy Connect, an advanced closed-system platform integrating AI-based process control for magnetic separation, cell washing, and expansion in cell therapy manufacturing workflows
- In May 2025, Lonza announced an USD 85 million expansion of its Houston cell therapy manufacturing facility, increasing capacity for next-generation automated closed-system platforms supporting commercial CAR-T and gene-modified therapies
- In June 2025, Xcell Biosciences and Thermo Fisher Scientific collaborated to advance automated separation and expansion workflows, improving reproducibility and scalability of GMP-compliant cell therapy production platforms globally
SKU-
Get online access to the report on the World's First Market Intelligence Cloud
- Interactive Data Analysis Dashboard
- Company Analysis Dashboard for high growth potential opportunities
- Research Analyst Access for customization & queries
- Competitor Analysis with Interactive dashboard
- Latest News, Updates & Trend analysis
- Harness the Power of Benchmark Analysis for Comprehensive Competitor Tracking
Research Methodology
Data collection and base year analysis are done using data collection modules with large sample sizes. The stage includes obtaining market information or related data through various sources and strategies. It includes examining and planning all the data acquired from the past in advance. It likewise envelops the examination of information inconsistencies seen across different information sources. The market data is analysed and estimated using market statistical and coherent models. Also, market share analysis and key trend analysis are the major success factors in the market report. To know more, please request an analyst call or drop down your inquiry.
The key research methodology used by DBMR research team is data triangulation which involves data mining, analysis of the impact of data variables on the market and primary (industry expert) validation. Data models include Vendor Positioning Grid, Market Time Line Analysis, Market Overview and Guide, Company Positioning Grid, Patent Analysis, Pricing Analysis, Company Market Share Analysis, Standards of Measurement, Global versus Regional and Vendor Share Analysis. To know more about the research methodology, drop in an inquiry to speak to our industry experts.
Customization Available
Data Bridge Market Research is a leader in advanced formative research. We take pride in servicing our existing and new customers with data and analysis that match and suits their goal. The report can be customized to include price trend analysis of target brands understanding the market for additional countries (ask for the list of countries), clinical trial results data, literature review, refurbished market and product base analysis. Market analysis of target competitors can be analyzed from technology-based analysis to market portfolio strategies. We can add as many competitors that you require data about in the format and data style you are looking for. Our team of analysts can also provide you data in crude raw excel files pivot tables (Fact book) or can assist you in creating presentations from the data sets available in the report.