Global Crispr Gene Editing Therapeutics Market
Market Size in USD Billion
CAGR :
% 
 USD
3.21 Billion
USD
12.90 Billion
2025
2033
USD
3.21 Billion
USD
12.90 Billion
2025
2033
| 2026 - 2033 | |
| USD 3.21 Billion | |
| USD 12.90 Billion | |
|
|
|
|
CRISPR Gene Editing Therapeutics Market Size
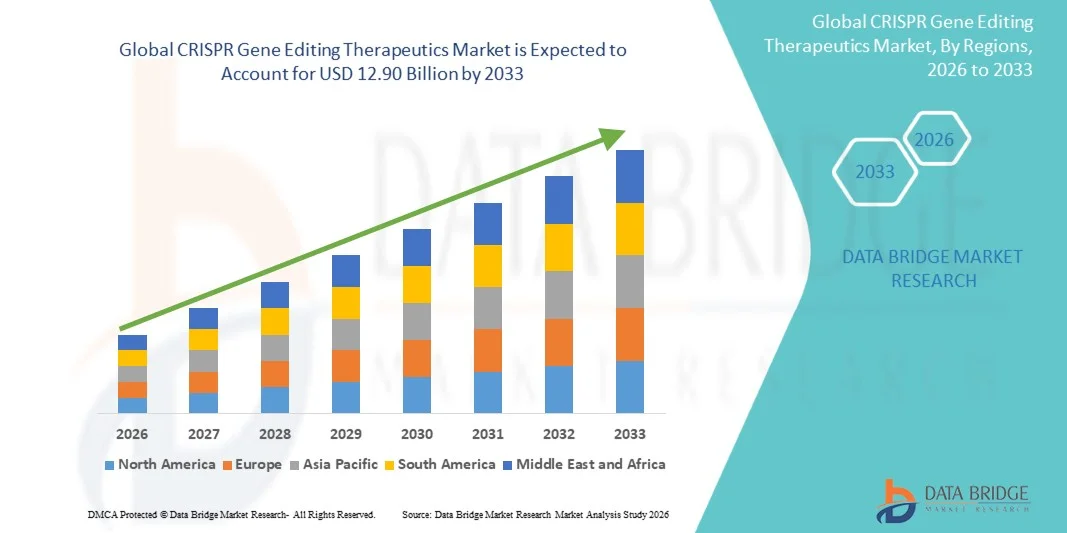
- The global CRISPR gene editing therapeutics market size was valued at USD 3.21 billion in 2025 and is expected to reach USD 12.90 billion by 2033, at a CAGR of 19.00% during the forecast period
- The market growth is largely driven by the increasing advancements in gene editing technologies and the rising focus on precision medicine, enabling targeted treatment of genetic and chronic diseases across multiple therapeutic areas
- Furthermore, growing clinical trial activity, expanding investment in biotechnology research, and rising demand for innovative, curative therapies are positioning CRISPR-based therapeutics as a transformative solution in modern healthcare, thereby significantly accelerating market growth
CRISPR Gene Editing Therapeutics Market Analysis
- CRISPR gene editing therapeutics, enabling precise modification of DNA sequences for the treatment of genetic and chronic diseases, are increasingly becoming essential tools in advanced healthcare applications such as gene therapy development, cell therapy, and drug discovery due to their high accuracy and potential for long-term therapeutic benefits
- The escalating demand for CRISPR-based therapeutics is primarily fueled by rapid technological advancements including CRISPR-Cas9, base editing, and prime editing, along with increasing investment in biotechnology research and the growing need for targeted and personalized treatment solutions
- North America dominated the CRISPR gene editing therapeutics market with the largest revenue share of 41.3% in 2025, characterized by strong presence of leading biotech firms, advanced research infrastructure, and high adoption of innovative technologies across gene therapy and drug development applications
- Asia-Pacific is expected to be the fastest growing region in the CRISPR gene editing therapeutics market during the forecast period due to rising investments in genomic research, expanding biopharmaceutical sector, and increasing adoption of advanced gene editing platforms
- CRISPR-Cas9 segment dominated the market with a share of 45.7% in 2025, driven by its widespread adoption, efficiency, and versatility across multiple applications including gene therapy development, functional genomics, and disease modeling
Report Scope and CRISPR Gene Editing Therapeutics Market Segmentation
|
Attributes |
CRISPR Gene Editing Therapeutics Key Market Insights |
|
Segments Covered |
|
|
Countries Covered |
North America
Europe
Asia-Pacific
Middle East and Africa
South America
|
|
Key Market Players |
|
|
Market Opportunities |
|
|
Value Added Data Infosets |
In addition to the insights on market scenarios such as market value, growth rate, segmentation, geographical coverage, and major players, the market reports curated by the Data Bridge Market Research also include in-depth expert analysis, patient epidemiology, pipeline analysis, pricing analysis, and regulatory framework |
CRISPR Gene Editing Therapeutics Market Trends
“Advancement of Precision Editing Through Next-Generation Technologies”
- A significant and accelerating trend in the global CRISPR gene editing therapeutics market is the advancement of next-generation technologies such as base editing and prime editing, enabling higher precision and efficiency in genetic modifications. This evolution is significantly enhancing therapeutic outcomes and expanding treatment possibilities
- For instance, base editing technologies allow direct, irreversible conversion of one DNA base into another without double-strand breaks, improving safety and reducing unintended mutations. Similarly, prime editing enables highly targeted DNA corrections with minimal off-target effects, offering a versatile platform for complex gene corrections
- Integration of advanced CRISPR technologies is enabling features such as improved targeting accuracy, reduced off-target activity, and enhanced delivery efficiency. For instance, some emerging platforms utilize engineered Cas variants to optimize editing precision and therapeutic efficacy while minimizing risks. Furthermore, these innovations offer researchers the ability to address a broader range of genetic mutations with greater confidence
- The seamless integration of CRISPR technologies with gene therapy and cell therapy platforms facilitates comprehensive treatment approaches across multiple disease areas. Through a unified framework, researchers can develop therapies that combine genome editing with cellular engineering, creating more effective and durable treatment solutions
- This trend towards more precise, efficient, and versatile gene editing systems is fundamentally transforming the therapeutic landscape. Consequently, companies such as CRISPR Therapeutics are developing advanced editing platforms with improved specificity and scalability for clinical applications
- The demand for CRISPR therapeutics that offer enhanced precision, safety, and broad applicability is growing rapidly across multiple therapeutic areas, as healthcare systems increasingly prioritize innovative and curative treatment approaches
- Growing collaboration between biotechnology companies and research institutions is accelerating innovation in CRISPR platforms, leading to faster clinical translation and commercialization of next-generation gene editing therapies
CRISPR Gene Editing Therapeutics Market Dynamics
Driver
“Rising Demand Driven by Increasing Genetic Disorders and Precision Medicine Adoption”
- The increasing prevalence of genetic disorders and chronic diseases, coupled with the growing adoption of precision medicine approaches, is a significant driver for the heightened demand for CRISPR gene editing therapeutics
- For instance, in 2024, Vertex Pharmaceuticals Incorporated, in collaboration with CRISPR Therapeutics, advanced CRISPR-based therapies targeting sickle cell disease and beta-thalassemia, highlighting the growing clinical adoption of gene editing solutions. Such strategies by key companies are expected to drive the CRISPR therapeutics market growth in the forecast period
- As healthcare providers seek more effective and long-term treatment options, CRISPR therapeutics offer the potential for one-time curative interventions, providing a compelling alternative to conventional therapies that require lifelong management
- Furthermore, the growing focus on personalized medicine and advancements in genomic research are accelerating the integration of CRISPR technologies into clinical pipelines, enabling tailored treatments based on individual genetic profiles
- The ability to precisely target and modify disease-causing genes, along with increasing regulatory support for advanced therapies, is a key factor propelling the adoption of CRISPR-based treatments across multiple therapeutic areas. The expansion of clinical trials and increasing success rates further contribute to market growth
- Increasing funding from governments and private investors is supporting large-scale research initiatives and accelerating the development of innovative CRISPR-based therapeutics across diverse disease areas
- Rising strategic partnerships and licensing agreements among pharmaceutical and biotechnology companies are enhancing resource sharing, speeding up product development, and strengthening market expansion efforts
Restraint/Challenge
“Ethical Concerns and Regulatory Complexity Hurdle”
- Concerns surrounding ethical implications and regulatory complexities associated with gene editing technologies pose a significant challenge to broader market adoption. As CRISPR therapeutics involve direct modification of genetic material, they raise questions about long-term safety and ethical boundaries
- For instance, debates around germline editing and potential unintended genetic consequences have led to strict regulatory scrutiny in multiple regions, slowing down approval processes and clinical advancements
- Addressing these concerns through robust regulatory frameworks, transparent clinical practices, and long-term safety studies is crucial for building stakeholder confidence. Companies such as Editas Medicine emphasize stringent clinical validation and compliance strategies to address regulatory requirements. In addition, the high cost associated with CRISPR therapy development and treatment can be a barrier to accessibility, particularly in low- and middle-income regions
- While technological advancements are gradually improving efficiency and reducing costs, the complexity of gene editing procedures and associated infrastructure requirements continue to limit widespread adoption. Ensuring affordability and scalability remains a key challenge for market players
- Overcoming these challenges through regulatory harmonization, ethical governance, and continued innovation in cost-effective delivery technologies will be vital for sustained market growth
- Limited long-term clinical data on safety and efficacy of CRISPR-based therapies creates uncertainty among stakeholders, potentially delaying regulatory approvals and wider adoption
- Technical challenges related to off-target effects and efficient delivery mechanisms remain critical barriers, requiring continuous innovation to ensure precise, safe, and reliable therapeutic outcomes
CRISPR Gene Editing Therapeutics Market Scope
The market is segmented on the basis of product type, technology type, application, and end user
- By Product Type
On the basis of product type, the CRISPR gene editing therapeutics market is segmented into Cas Proteins, Guide RNAs, Delivery Systems, CRISPR Libraries, Kits & Reagents, and Detection & Screening Kits. The Cas Proteins segment dominated the market with the largest revenue share in 2025, driven by their fundamental role as the core enzyme responsible for DNA cleavage and gene editing activity. These proteins, particularly Cas9, are essential for nearly all CRISPR-based therapeutic applications, making them indispensable in both research and clinical settings. The increasing adoption of CRISPR technologies across gene therapy and drug development pipelines has significantly boosted the demand for high-quality and engineered Cas proteins. In addition, continuous innovation in protein engineering to enhance specificity and reduce off-target effects further strengthens the dominance of this segment. The scalability and adaptability of Cas proteins across multiple therapeutic applications also contribute to their widespread utilization.
The Delivery Systems segment is anticipated to witness the fastest growth rate from 2026 to 2033, fueled by the critical need for efficient and safe delivery of CRISPR components into target cells. Delivery remains one of the most significant challenges in gene editing, driving innovation in viral vectors, lipid nanoparticles, and non-viral methods. Advancements in in vivo delivery technologies are expanding the therapeutic scope of CRISPR, enabling treatment of previously inaccessible tissues. Increasing investment in improving delivery efficiency and reducing immune responses is further accelerating segment growth. Moreover, the growing focus on precision medicine and targeted therapies is pushing the demand for advanced delivery platforms. As clinical trials progress, the importance of reliable delivery systems is expected to drive rapid adoption.
- By Technology Type
On the basis of technology type, the market is segmented into CRISPR-Cas9, CRISPR-Cas12 (Cpf1), CRISPR-Cas13, Base Editing, Prime Editing, and Epigenome Editing. The CRISPR-Cas9 segment dominated the market with the largest revenue share of 45.7% in 2025, owing to its widespread adoption, proven efficiency, and extensive validation across multiple research and therapeutic applications. Cas9 technology has become the standard platform for gene editing due to its simplicity, versatility, and cost-effectiveness. It is extensively used in gene therapy development, functional genomics, and disease modeling, making it the backbone of CRISPR-based innovations. Continuous improvements in Cas9 variants to enhance specificity and reduce off-target effects further support its dominance. In addition, the large number of ongoing clinical trials utilizing CRISPR-Cas9 reinforces its leading position in the market.
The Prime Editing segment is expected to witness the fastest CAGR from 2026 to 2033, driven by its ability to perform highly precise DNA modifications without inducing double-strand breaks. This advanced technology offers significant advantages in terms of safety and accuracy compared to traditional CRISPR methods. Increasing research focus on correcting complex genetic mutations is accelerating the adoption of prime editing. The technology’s potential to address a broader range of genetic disorders is attracting significant investment from biotechnology companies. Furthermore, ongoing advancements aimed at improving efficiency and scalability are expected to drive its rapid growth. As clinical applications expand, prime editing is likely to become a key driver of next-generation gene therapies.
- By Application
On the basis of application, the market is segmented into Gene Therapy Development, Cell Therapy, Drug Discovery & Development, Functional Genomics, and Disease Modeling. The Gene Therapy Development segment dominated the market with the largest revenue share in 2025, driven by the increasing focus on developing curative treatments for genetic disorders and chronic diseases. CRISPR technology plays a crucial role in enabling precise gene correction, making it highly suitable for therapeutic applications. The growing number of clinical trials targeting diseases such as sickle cell anemia and beta-thalassemia is significantly contributing to segment growth. In addition, regulatory support for advanced therapies and increasing investment in gene therapy research are further strengthening its dominance. The potential for one-time curative treatments continues to attract both investors and healthcare providers.
The Cell Therapy segment is anticipated to witness the fastest growth rate from 2026 to 2033, fueled by the rising adoption of CRISPR in engineering immune cells for cancer treatment. Technologies such as CAR-T cell therapy are increasingly incorporating CRISPR for enhanced efficacy and precision. The ability to modify cells ex vivo and reintroduce them into patients offers significant therapeutic advantages. Growing interest in personalized medicine and immunotherapy is further accelerating the demand for CRISPR-enabled cell therapies. In addition, advancements in cell engineering techniques are expanding the range of treatable conditions. As research progresses, cell therapy is expected to emerge as a major growth driver in the market.
- By End User
On the basis of end user, the market is segmented into pharmaceutical & biotechnology companies, academic & research institutes, and contract research organizations. The Pharmaceutical & Biotechnology Companies segment dominated the market with the largest revenue share in 2025, driven by their strong financial capabilities and active involvement in drug development and commercialization. These companies are heavily investing in CRISPR technologies to develop innovative therapies and expand their product pipelines. Strategic collaborations, partnerships, and acquisitions are further enhancing their market position. The increasing number of clinical trials and regulatory approvals is also contributing to segment dominance. In addition, their ability to scale production and bring therapies to market efficiently supports their leading role.
The Contract Research Organizations segment is expected to witness the fastest CAGR from 2026 to 2033, fueled by the growing trend of outsourcing research and development activities. CROs provide specialized expertise and cost-effective solutions, enabling companies to accelerate drug development timelines. The increasing complexity of CRISPR-based research is driving demand for external support services. Furthermore, CROs are investing in advanced technologies and infrastructure to cater to the evolving needs of biotechnology firms. The expansion of global clinical trials and research activities is further boosting their growth. As outsourcing becomes more prevalent, CROs are expected to play a crucial role in market expansion.
CRISPR Gene Editing Therapeutics Market Regional Analysis
- North America dominated the CRISPR gene editing therapeutics market with the largest revenue share of 41.3% in 2025, characterized by strong presence of leading biotech firms, advanced research infrastructure, and high adoption of innovative technologies across gene therapy and drug development applications
- Healthcare systems and research institutions in the region highly value the precision, innovation, and transformative potential offered by CRISPR-based therapies in treating genetic disorders and chronic diseases
- This widespread adoption is further supported by advanced healthcare infrastructure, presence of leading biotechnology companies, and a strong focus on clinical research and regulatory support, establishing CRISPR therapeutics as a preferred solution for next-generation treatment development
U.S. CRISPR Gene Editing Therapeutics Market Insight
The U.S. CRISPR gene editing therapeutics market captured the largest revenue share within North America in 2025, fueled by the rapid advancement of biotechnology research and the growing number of clinical trials focused on gene-based therapies. Healthcare providers and researchers are increasingly prioritizing innovative treatment approaches for genetic and chronic diseases. The growing focus on precision medicine, combined with strong investment from both public and private sectors, further propels the market. Moreover, the increasing collaboration between biotechnology companies and research institutions is significantly contributing to the market's expansion.
Europe CRISPR Gene Editing Therapeutics Market Insight
The Europe CRISPR gene editing therapeutics market is projected to expand at a substantial CAGR throughout the forecast period, primarily driven by supportive regulatory frameworks and the increasing demand for advanced treatment solutions. The rise in genomic research, coupled with growing healthcare investments, is fostering the adoption of CRISPR technologies. European healthcare systems are also focusing on innovation and patient-centric care. The region is experiencing notable growth across research and therapeutic applications, with CRISPR technologies being incorporated into both clinical trials and advanced therapy development programs.
U.K. CRISPR Gene Editing Therapeutics Market Insight
The U.K. CRISPR gene editing therapeutics market is anticipated to grow at a noteworthy CAGR during the forecast period, driven by increasing investment in life sciences and a strong focus on genomic medicine. In addition, rising prevalence of genetic disorders is encouraging healthcare providers and researchers to adopt advanced gene editing solutions. The country’s well-established research ecosystem, alongside its growing biotechnology sector, is expected to continue to stimulate market growth.
Germany CRISPR Gene Editing Therapeutics Market Insight
The Germany CRISPR gene editing therapeutics market is expected to expand at a considerable CAGR during the forecast period, fueled by increasing focus on biomedical innovation and the demand for advanced therapeutic solutions. Germany’s strong research infrastructure, combined with its emphasis on technological advancement, promotes the adoption of CRISPR-based therapies. The integration of gene editing technologies into clinical research is also becoming increasingly prevalent, with a strong preference for safe and effective treatment solutions aligning with local healthcare priorities.
Asia-Pacific CRISPR Gene Editing Therapeutics Market Insight
The Asia-Pacific CRISPR gene editing therapeutics market is poised to grow at the fastest CAGR during the forecast period of 2026 to 2033, driven by increasing healthcare investments, rising prevalence of chronic diseases, and advancements in biotechnology in countries such as China, Japan, and India. The region's growing focus on precision medicine, supported by government initiatives promoting genomic research, is driving the adoption of CRISPR therapeutics. Furthermore, as Asia-Pacific emerges as a hub for biotechnology innovation and clinical research, accessibility and development of gene editing therapies are expanding significantly.
Japan CRISPR Gene Editing Therapeutics Market Insight
The Japan CRISPR gene editing therapeutics market is gaining momentum due to the country’s advanced healthcare system, strong focus on innovation, and increasing demand for targeted therapies. The Japanese market places a significant emphasis on precision medicine, and the adoption of CRISPR technologies is driven by the growing number of research initiatives and clinical studies. The integration of gene editing with other advanced therapeutic approaches is fueling growth. Moreover, Japan's aging population is likely to spur demand for effective and long-term treatment solutions in both clinical and research settings.
India CRISPR Gene Editing Therapeutics Market Insight
The India CRISPR gene editing therapeutics market accounted for a significant share in Asia Pacific in 2025, attributed to the country's expanding biotechnology sector, increasing healthcare awareness, and rising investment in genomic research. India stands as one of the emerging markets for advanced therapeutics, and CRISPR technologies are gaining traction in research and clinical applications. The push towards biotechnology innovation and the presence of a skilled research workforce, alongside supportive government initiatives, are key factors propelling the market in India
CRISPR Gene Editing Therapeutics Market Share
The CRISPR Gene Editing Therapeutics industry is primarily led by well-established companies, including:
- CRISPR Therapeutics AG (Switzerland)
- Intellia Therapeutics, Inc. (U.S.)
- Editas Medicine, Inc. (U.S.)
- Beam Therapeutics Inc. (U.S.)
- Caribou Biosciences, Inc. (U.S.)
- Verve Therapeutics, Inc. (U.S.)
- Mammoth Biosciences, Inc. (U.S.)
- ERS Genomics Limited (Ireland)
- Precision BioSciences, Inc. (U.S.)
- Sangamo Therapeutics, Inc. (U.S.)
- Cellectis S.A. (France)
- Lonza Group Ltd (Switzerland)
- Takara Bio Inc. (Japan)
- Thermo Fisher Scientific Inc. (U.S.)
- Merck KGaA (Germany)
- Agilent Technologies, Inc. (U.S.)
- GenScript Biotech Corporation (China)
- Horizon Discovery Group plc (U.K.)
- Synthego Corporation (U.S.)
- Inscripta, Inc. (U.S.)
What are the Recent Developments in Global CRISPR Gene Editing Therapeutics Market?
- In January 2025, CRISPR Therapeutics announced progress in its in vivo gene editing programs, including Phase 1 clinical trials for cardiovascular diseases using lipid nanoparticle delivery systems. The company reported significant reductions in triglycerides and LDL levels, highlighting the growing potential of CRISPR therapies beyond rare diseases into more common conditions. This development underscores the expansion of CRISPR applications into broader therapeutic areas
- In April 2024, Regeneron Pharmaceuticals partnered with Mammoth Biosciences to develop CRISPR-based gene editing therapies for multiple diseases. The collaboration combines Mammoth’s CRISPR platform with Regeneron’s delivery technologies to enable treatment across different tissues. This partnership reflects the increasing strategic alliances accelerating innovation and commercialization in the CRISPR therapeutics space
- In February 2024, the European Medicines Agency approved the CRISPR-based therapy CASGEVY for the treatment of sickle cell disease and beta-thalassemia following earlier approvals. This marked a significant milestone in expanding access to gene-editing therapies across Europe. The approval reinforced the global regulatory acceptance of CRISPR-based therapeutics and their transition into mainstream clinical use
- In December 2023, U.S. Food and Drug Administration approved CASGEVY, developed by Vertex Pharmaceuticals Incorporated and CRISPR Therapeutics, marking the world’s first CRISPR/Cas9-based gene editing therapy for sickle cell disease. This historic approval demonstrated the clinical viability of CRISPR technology and opened the door for future gene-editing therapeutics. It represents a major breakthrough in the treatment of genetic disorder
- In November 2023, Medicines and Healthcare products Regulatory Agency granted the first-ever regulatory authorization for a CRISPR gene-edited therapy, CASGEVY, in the United Kingdom. This approval positioned the UK as the first country to authorize a CRISPR-based therapeutic, accelerating global momentum for gene editing technologies. The decision marked a pivotal step toward commercialization of CRISPR therapies worldwide
SKU-
Get online access to the report on the World's First Market Intelligence Cloud
- Interactive Data Analysis Dashboard
- Company Analysis Dashboard for high growth potential opportunities
- Research Analyst Access for customization & queries
- Competitor Analysis with Interactive dashboard
- Latest News, Updates & Trend analysis
- Harness the Power of Benchmark Analysis for Comprehensive Competitor Tracking
Research Methodology
Data collection and base year analysis are done using data collection modules with large sample sizes. The stage includes obtaining market information or related data through various sources and strategies. It includes examining and planning all the data acquired from the past in advance. It likewise envelops the examination of information inconsistencies seen across different information sources. The market data is analysed and estimated using market statistical and coherent models. Also, market share analysis and key trend analysis are the major success factors in the market report. To know more, please request an analyst call or drop down your inquiry.
The key research methodology used by DBMR research team is data triangulation which involves data mining, analysis of the impact of data variables on the market and primary (industry expert) validation. Data models include Vendor Positioning Grid, Market Time Line Analysis, Market Overview and Guide, Company Positioning Grid, Patent Analysis, Pricing Analysis, Company Market Share Analysis, Standards of Measurement, Global versus Regional and Vendor Share Analysis. To know more about the research methodology, drop in an inquiry to speak to our industry experts.
Customization Available
Data Bridge Market Research is a leader in advanced formative research. We take pride in servicing our existing and new customers with data and analysis that match and suits their goal. The report can be customized to include price trend analysis of target brands understanding the market for additional countries (ask for the list of countries), clinical trial results data, literature review, refurbished market and product base analysis. Market analysis of target competitors can be analyzed from technology-based analysis to market portfolio strategies. We can add as many competitors that you require data about in the format and data style you are looking for. Our team of analysts can also provide you data in crude raw excel files pivot tables (Fact book) or can assist you in creating presentations from the data sets available in the report.