Global In Vivo Crispr Delivery Platforms Market
Market Size in USD Billion
CAGR :
% 
 USD
1.73 Billion
USD
8.49 Billion
2025
2033
USD
1.73 Billion
USD
8.49 Billion
2025
2033
| 2026 - 2033 | |
| USD 1.73 Billion | |
| USD 8.49 Billion | |
|
|
|
|
In Vivo CRISPR Delivery Platforms Market Size
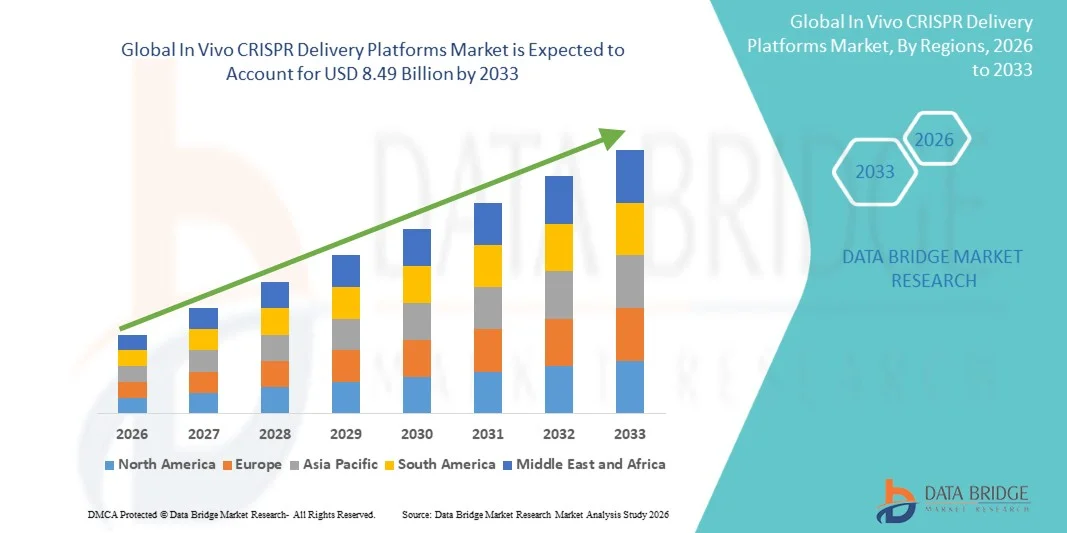
- The global In Vivo CRISPR Delivery Platforms market size was valued at USD 1.73 billion in 2025 and is expected to reach USD 8.49 billion by 2033, at a CAGR of 22.00% during the forecast period
- The market growth is largely fueled by the increasing advancements in gene editing technologies and the growing adoption of CRISPR-based therapeutics, leading to rising research activities in genetic engineering and precision medicine across biotechnology and pharmaceutical sectors
- Furthermore, increasing investments in genetic disorder treatment, oncology research, and next-generation gene therapy platforms are establishing in vivo CRISPR delivery platforms as critical tools for efficient and targeted gene editing. These converging factors are accelerating the uptake of In Vivo CRISPR Delivery Platforms solutions, thereby significantly boosting the industry's growth
In Vivo CRISPR Delivery Platforms Market Analysis
- In vivo CRISPR delivery platforms, enabling the direct delivery of gene-editing components into living cells and tissues, are becoming critical tools in modern gene therapy and precision medicine due to their ability to enable targeted genetic modifications for the treatment of complex diseases
- The escalating demand for in vivo CRISPR delivery platforms is primarily fueled by the rapid advancement of gene editing technologies, increasing research in genetic disorder treatment, and the growing focus on developing next-generation gene therapies for diseases such as cancer, rare genetic disorders, and inherited conditions
- North America dominated the In Vivo CRISPR Delivery Platforms market with the largest revenue share of approximately 43.1% in 2025, supported by strong biotechnology research infrastructure, high investments in gene therapy development, and the presence of leading biotechnology and pharmaceutical companies. The U.S. is experiencing substantial growth in the adoption of in vivo CRISPR delivery technologies across biotechnology companies and research institutions due to increasing clinical research activities and funding for advanced gene-editing therapeutics
- Asia-Pacific is expected to be the fastest-growing region in the In Vivo CRISPR Delivery Platforms market during the forecast period, registering a CAGR of approximately 10.2%, driven by expanding biotechnology research capabilities, rising investments in genomic medicine, and increasing focus on advanced gene therapy development across countries such as China, Japan, and India
- The Genetic Disorders segment dominated the largest market revenue share of around 53.1% in 2025, driven by the rising prevalence of monogenic and rare hereditary diseases
Report Scope and In Vivo CRISPR Delivery Platforms Market Segmentation
|
Attributes |
In Vivo CRISPR Delivery Platforms Key Market Insights |
|
Segments Covered |
|
|
Countries Covered |
North America
Europe
Asia-Pacific
Middle East and Africa
South America
|
|
Key Market Players |
• Editas Medicine (U.S.) |
|
Market Opportunities |
|
|
Value Added Data Infosets |
In addition to the insights on market scenarios such as market value, growth rate, segmentation, geographical coverage, and major players, the market reports curated by the Data Bridge Market Research also include in-depth expert analysis, patient epidemiology, pipeline analysis, pricing analysis, and regulatory framework. |
In Vivo CRISPR Delivery Platforms Market Trends
“Advancements in Targeted Gene Editing and Therapeutics”
- A significant trend in the global in vivo CRISPR delivery platforms market is the shift toward highly precise and targeted gene editing methods. Researchers and biotech companies are increasingly developing delivery vectors capable of targeting specific tissues or cell types, minimizing off-target effects and enhancing therapeutic efficacy
- Novel viral and non-viral delivery systems, including adeno-associated viruses (AAV), lipid nanoparticles (LNPs), and electroporation-based methods, are being optimized for higher efficiency, safety, and reproducibility in clinical application
- For instance, Editas Medicine has advanced its AAV-mediated CRISPR therapy for Leber congenital amaurosis into early clinical trials, and Beam Therapeutics is using LNP-based delivery for base editing in sickle cell disease models
- There is also a growing integration of CRISPR platforms with base editors and prime editing technologies, enabling precise single-nucleotide modifications for treating monogenic disorders, such as sickle cell disease, Duchenne muscular dystrophy, and inherited retinal diseases
- The market is further witnessing collaborations between academic institutions, biotech startups, and pharmaceutical giants to expand in vivo gene editing pipelines for both rare genetic diseases and complex disorders like cancer and cardiovascular diseases
In Vivo CRISPR Delivery Platforms Market Dynamics
Driver
“Rising Demand for Gene Therapy and Precision Medicine”
- The increasing prevalence of genetic disorders, chronic diseases, and cancer is driving the adoption of in vivo CRISPR delivery platforms as a cornerstone of precision medicine. Governments and private investors are significantly funding research in gene therapy to develop curative solutions, particularly for conditions with limited treatment option
- The adoption of in vivo CRISPR platforms is further fueled by the demand for scalable and clinically translatable delivery methods that can reach systemic or tissue-specific targets.
- For instance, Intellia Therapeutics reported successful preclinical systemic delivery of CRISPR-Cas9 via lipid nanoparticles to treat transthyretin amyloidosis in mouse models, demonstrating potential for human application
- Advancements in regulatory approvals for gene therapies and supportive frameworks for clinical research in North America, Europe, and parts of Asia-Pacific are boosting the market. This includes the accelerated review of innovative gene-editing therapies under programs such as the U.S. FDA’s Breakthrough Therapy Designation and EMA’s PRIME (PRIority MEdicines) initiative
- Furthermore, the rise in biotechnology R&D investments, coupled with increasing collaborations between academic labs and pharmaceutical companies, is driving pipeline expansion and adoption of advanced in vivo CRISPR delivery platforms globally
Restraint/Challenge
“Safety Concerns, Off-Target Effects, and Regulatory Challenges”
- Despite rapid technological progress, safety concerns remain a key restraint for market growth. Off-target gene editing, immune responses against delivery vectors, and long-term safety of in vivo CRISPR therapies pose significant hurdles for clinical translation
- High development costs and complex manufacturing processes for viral and non-viral delivery systems limit accessibility and scalability, particularly in emerging markets
- For instance, viral vector-based therapies such as Spark Therapeutics’ Luxturna face manufacturing bottlenecks and high per-patient costs, impacting broader adoption
- Regulatory challenges also hinder market expansion, as global regulatory frameworks for gene-editing therapeutics are still evolving. Different regions have varying requirements for preclinical and clinical studies, making harmonization and global adoption difficult
- Intellectual property (IP) disputes related to CRISPR technologies can also impact licensing agreements, collaborations, and overall market entry strategies for new players
- Limited patient awareness and clinician familiarity with in vivo CRISPR therapies can slow adoption, as education and training on the safe and effective use of these platforms are still in early stages
- Supply chain constraints, including availability of high-quality reagents, specialized viral vectors, and GMP-grade manufacturing facilities, can delay research and commercial-scale production
- Ethical concerns surrounding germline editing, potential unintended genetic modifications, and societal acceptance of gene editing may pose long-term barriers to market penetration
- Reimbursement and pricing challenges in both developed and developing countries can restrict patient access, as advanced CRISPR therapies may not be covered by insurance or public health programs initially
In Vivo CRISPR Delivery Platforms Market Scope
The In Vivo CRISPR Delivery Platforms market is segmented on the basis of delivery method and application.
• By Delivery Method
On the basis of delivery method, the market is segmented into Viral Delivery Systems and Non-Viral Delivery Systems. The Viral Delivery Systems segment dominated the largest market revenue share of approximately 47.5% in 2025, driven by its high efficiency in delivering CRISPR components into target cells and tissues. Viral vectors such as adenoviruses, lentiviruses, and adeno-associated viruses (AAVs) offer stable gene expression and are widely used in preclinical and clinical studies. The segment benefits from increasing research in gene therapy, rising prevalence of genetic disorders, and expanding clinical trials targeting monogenic and complex diseases. Hospitals, research institutes, and biotechnology companies prefer viral systems for their predictable transduction efficiency and versatility across tissue types. Technological advancements in viral vector engineering have enhanced safety, reduced immunogenicity, and improved delivery precision. Regulatory approvals supporting viral delivery in gene therapy applications further reinforce the segment’s dominance. The growing adoption of CRISPR-based therapeutics in oncology, rare genetic diseases, and regenerative medicine also contributes to revenue. Increased investment by biopharmaceutical companies in vector optimization and scalable production techniques ensures recurring demand. Training programs and academic collaborations are expanding the use of viral systems in translational research. Geographically, North America and Europe lead in adoption due to established healthcare infrastructure and early regulatory approvals. Overall, viral delivery systems remain the preferred choice due to their efficiency, reliability, and broad application scope.
The Non-Viral Delivery Systems segment is expected to witness the fastest CAGR of 11.8% from 2026 to 2033, fueled by the growing demand for safer, less immunogenic delivery methods in gene editing therapies. Non-viral systems, including lipid nanoparticles, polymer-based carriers, and electroporation methods, provide transient CRISPR delivery without integrating into the host genome. The segment is gaining traction in oncology, genetic disorder therapies, and personalized medicine applications due to reduced risk of insertional mutagenesis. Technological advancements in nanocarriers and delivery vehicles have improved cellular uptake, tissue targeting, and in vivo stability. Pharmaceutical and biotechnology companies are increasingly adopting non-viral approaches to meet regulatory and safety requirements for clinical trials. Rising patient preference for minimally invasive therapies and expanding research on targeted gene editing further accelerate growth. The segment also benefits from cost-effective production, scalability, and compatibility with multiple CRISPR modalities, making it suitable for large-scale therapeutic development. Increasing collaborations between academic institutions and industry players drive innovation and application of non-viral delivery platforms globally. Emerging markets are witnessing faster adoption due to lower infrastructural requirements and flexibility in therapeutic development.
• By Application
On the basis of application, the market is segmented into Genetic Disorders and Oncology. The Genetic Disorders segment dominated the largest market revenue share of around 53.1% in 2025, driven by the rising prevalence of monogenic and rare hereditary diseases. CRISPR-based in vivo delivery enables targeted gene correction, offering potential curative outcomes for diseases such as cystic fibrosis, sickle cell anemia, and Duchenne muscular dystrophy. Hospitals, research institutes, and biopharmaceutical companies are increasingly adopting in vivo CRISPR platforms for preclinical and clinical development. Regulatory support for gene therapy trials and increased funding for rare disease research further bolster adoption. Technological advancements in delivery vectors and genome editing efficiency enhance treatment efficacy and patient safety. The segment also benefits from expanding awareness among clinicians and patients regarding gene-editing therapies. Recurrent use in ongoing research programs and therapeutic pipelines ensures a stable revenue stream. North America and Europe dominate due to well-established healthcare infrastructure and early clinical adoption. Strategic partnerships between academia and biotech firms further support the segment’s growth.
The Oncology segment is expected to witness the fastest CAGR of 12.3% from 2026 to 2033, fueled by increasing research into CRISPR-based cancer immunotherapies and targeted gene editing. In vivo delivery platforms enable precise editing of oncogenes, tumor suppressor genes, and immune checkpoints, offering new therapeutic avenues. Rising cancer prevalence and unmet treatment needs drive adoption in hospitals, research labs, and biotech companies. Technological innovations in viral and non-viral delivery improve targeting specificity, reduce off-target effects, and enhance treatment safety. Growing clinical trials for hematological and solid tumors, along with increased funding in oncology research, accelerate segment growth. The segment also benefits from collaborations between pharmaceutical companies and academic centers to develop next-generation CRISPR therapeutics. Expanding awareness of precision oncology and personalized medicine contributes to faster adoption. Emerging markets are seeing increasing clinical research infrastructure, supporting segment expansion. Overall, oncology represents a high-growth application area due to its therapeutic potential, innovation, and clinical demand.
In Vivo CRISPR Delivery Platforms Market Regional Analysis
- North America dominated the In Vivo CRISPR Delivery Platforms market with the largest revenue share of approximately 43.1% in 2025, supported by strong biotechnology research infrastructure, high investments in gene therapy development, and the presence of leading biotechnology and pharmaceutical companies
- The U.S. market is experiencing substantial growth in the adoption of in vivo CRISPR delivery technologies across biotechnology companies, research institutions, and academic centers. For instance, clinical trials conducted by Editas Medicine and CRISPR Therapeutics are driving practical applications of these delivery platforms for treating genetic disorders
- Canada is also contributing to regional growth, with increasing funding for genomic research and expanding gene therapy clinical programs in specialized research hospitals and academic institutes, strengthening the adoption of advanced in vivo CRISPR technologies
U.S. In Vivo CRISPR Delivery Platforms Market Insight
The U.S. In Vivo CRISPR Delivery Platforms market captured the largest revenue share of the North American region in 2025, driven by robust clinical research activities, a strong biotech ecosystem, and rising government and private funding for gene-editing therapeutics. Institutions such as the Broad Institute and University of California are actively conducting in vivo CRISPR-based studies for rare genetic diseases, oncology, and immunotherapy applications. The growing collaboration between biotech startups and established pharmaceutical companies further accelerates the adoption of advanced in vivo CRISPR delivery systems.
Europe In Vivo CRISPR Delivery Platforms Market Insight
The Europe In Vivo CRISPR Delivery Platforms market is projected to expand at a substantial CAGR during the forecast period, fueled by increasing investments in genomic medicine and stringent regulations supporting gene therapy development. Germany, France, and the U.K. are actively investing in research infrastructure and clinical trials for in vivo CRISPR applications, particularly in rare disease therapy and hematology. Collaborative European projects, such as Horizon Europe funding initiatives, are also contributing to the growth of CRISPR delivery platform adoption across the region.
U.K. In Vivo CRISPR Delivery Platforms Market Insight
The U.K. In Vivo CRISPR Delivery Platforms market is expected to grow steadily due to the presence of advanced research institutions, increasing government support for genomic medicine, and growing investment in biopharmaceutical startups focused on in vivo CRISPR therapeutics. Partnerships between universities and biotech companies for developing innovative delivery vectors are further strengthening market growth.
Germany In Vivo CRISPR Delivery Platforms Market Insight
Germany In Vivo CRISPR Delivery Platforms market is witnessing strong growth in the market due to a well-developed biotechnology sector, investments in gene therapy research, and government-backed initiatives supporting clinical trials. Hospitals and specialized research centers are actively adopting in vivo CRISPR delivery platforms for experimental therapies targeting genetic disorders, oncology, and immunology.
Asia-Pacific In Vivo CRISPR Delivery Platforms Market Insight
The Asia-Pacific market is expected to be the fastest-growing region, registering a CAGR of approximately 10.2% during the forecast period. Growth is driven by expanding biotechnology research capabilities, rising investments in genomic medicine, and increasing focus on advanced gene therapy development across countries such as China, Japan, and India. In China, companies such as Beijing Genomics Institute (BGI) and major hospitals are implementing in vivo CRISPR platforms for clinical trials targeting hematological disorders and cancer, significantly boosting regional adoption.
Japan In Vivo CRISPR Delivery Platforms Market Insight
Japan In Vivo CRISPR Delivery Platforms market is gaining momentum due to high technological adoption, strong government funding for genomic medicine, and active clinical trial pipelines in gene therapy. Universities and biotech companies are collaborating to develop innovative in vivo CRISPR vectors for inherited disorders and oncology applications.
China In Vivo CRISPR Delivery Platforms Market Insight
China In Vivo CRISPR Delivery Platforms market accounted for the largest market revenue share in Asia-Pacific in 2025, driven by rapid biotechnology research expansion, increasing clinical trials, and strong domestic pharmaceutical manufacturers. The government’s push for precision medicine and genomic innovation, alongside increased healthcare funding, is propelling widespread adoption of in vivo CRISPR delivery platforms.
In Vivo CRISPR Delivery Platforms Market Share
The In Vivo CRISPR Delivery Platforms industry is primarily led by well-established companies, including:
• Editas Medicine (U.S.)
• Intellia Therapeutics (U.S.)
• CRISPR Therapeutics (Switzerland)
• Caribou Biosciences (U.S.)
• Beam Therapeutics (U.S.)
• Sangamo Therapeutics (U.S.)
• Precision BioSciences (U.S.)
• Verve Therapeutics (U.S.)
• Allogene Therapeutics (U.S.)
• GenScript Biotech (China)
• Synthego (U.S.)
• Cellectis (France)
• Poseida Therapeutics (U.S.)
• Intrexon (U.S.)
• Origene Technologies (China)
• Takara Bio (Japan)
• Horizon Discovery (U.K.)
• eGenesis (U.S.)
• Prime Medicine (U.S.)
• Twist Bioscience (U.S.)
Latest Developments in Global In Vivo CRISPR Delivery Platforms Market
- In October 2021, Intellia Therapeutics reported successful in vivo CRISPR gene editing in a human subject using its proprietary lipid nanoparticle (LNP) delivery system for transthyretin amyloidosis (ATTR), demonstrating durable reductions in disease‑causing protein levels following a single IV dose and marking a major milestone in non‑viral in vivo CRISPR delivery technologies
- In March 2023, CRISPR Therapeutics announced the advancement of its proprietary liver‑targeted LNP delivery platform and ongoing clinical trials for two in vivo CRISPR programs—CTX310 targeting ANGPTL3 and CTX320 targeting elevated lipoprotein(a)—which are being developed to treat cardiovascular diseases through direct in vivo gene editing in liver cells
- In July 2025, Verve Therapeutics’ in vivo CRISPR base‑editing therapy VERVE‑102, designed to reduce LDL cholesterol by editing the PCSK9 gene, showed promising early clinical results and received continued investment interest; its development and potential Phase II trials positioned the therapy on track to transform cardiovascular disease management through single‑dose gene editing
- In August 2025, CRISPR‑Gels, a nanogel‑based CRISPR delivery platform capable of encapsulating CRISPR‑Cas9 ribonucleoproteins (RNPs), was highlighted in preclinical studies for efficient in vivo release and gene editing activity, representing a new area of non‑viral CRISPR delivery research with potential for improved safety profiles over viral vectors
- In February 2025, CRISPR Therapeutics highlighted multiple anticipated 2025 milestones for its in vivo CRISPR delivery programs, including dose escalation updates for CTX310 and CTX320 and progression of next‑generation candidates such as CTX321, underscoring ongoing clinical development of lipid nanoparticle‑based in vivo gene editing therapies
SKU-
Get online access to the report on the World's First Market Intelligence Cloud
- Interactive Data Analysis Dashboard
- Company Analysis Dashboard for high growth potential opportunities
- Research Analyst Access for customization & queries
- Competitor Analysis with Interactive dashboard
- Latest News, Updates & Trend analysis
- Harness the Power of Benchmark Analysis for Comprehensive Competitor Tracking
Research Methodology
Data collection and base year analysis are done using data collection modules with large sample sizes. The stage includes obtaining market information or related data through various sources and strategies. It includes examining and planning all the data acquired from the past in advance. It likewise envelops the examination of information inconsistencies seen across different information sources. The market data is analysed and estimated using market statistical and coherent models. Also, market share analysis and key trend analysis are the major success factors in the market report. To know more, please request an analyst call or drop down your inquiry.
The key research methodology used by DBMR research team is data triangulation which involves data mining, analysis of the impact of data variables on the market and primary (industry expert) validation. Data models include Vendor Positioning Grid, Market Time Line Analysis, Market Overview and Guide, Company Positioning Grid, Patent Analysis, Pricing Analysis, Company Market Share Analysis, Standards of Measurement, Global versus Regional and Vendor Share Analysis. To know more about the research methodology, drop in an inquiry to speak to our industry experts.
Customization Available
Data Bridge Market Research is a leader in advanced formative research. We take pride in servicing our existing and new customers with data and analysis that match and suits their goal. The report can be customized to include price trend analysis of target brands understanding the market for additional countries (ask for the list of countries), clinical trial results data, literature review, refurbished market and product base analysis. Market analysis of target competitors can be analyzed from technology-based analysis to market portfolio strategies. We can add as many competitors that you require data about in the format and data style you are looking for. Our team of analysts can also provide you data in crude raw excel files pivot tables (Fact book) or can assist you in creating presentations from the data sets available in the report.