Global Intravenous Immunoglobulin Market
Market Size in USD Million
CAGR :
% 
 USD
11.97 Million
USD
21.83 Million
2022
2030
USD
11.97 Million
USD
21.83 Million
2022
2030
| 2023 –2030 | |
| USD 11.97 Million | |
| USD 21.83 Million | |
|
|
|
|
Intravenous Immunoglobulin Market Analysis and Size
Immunoglobulin are manufactured from a huge pool of human plasma that is collected from at least 100 donors or more. The intramuscular route of administration is a conventional method and is not preferred currently, as it is painful. Intravenous immunoglobulin has developed as a boon in the last few years for patients with compromised immune systems. The increasing burden of target diseases such as primary immunodeficiency diseases is projected to boost the growth of the market.
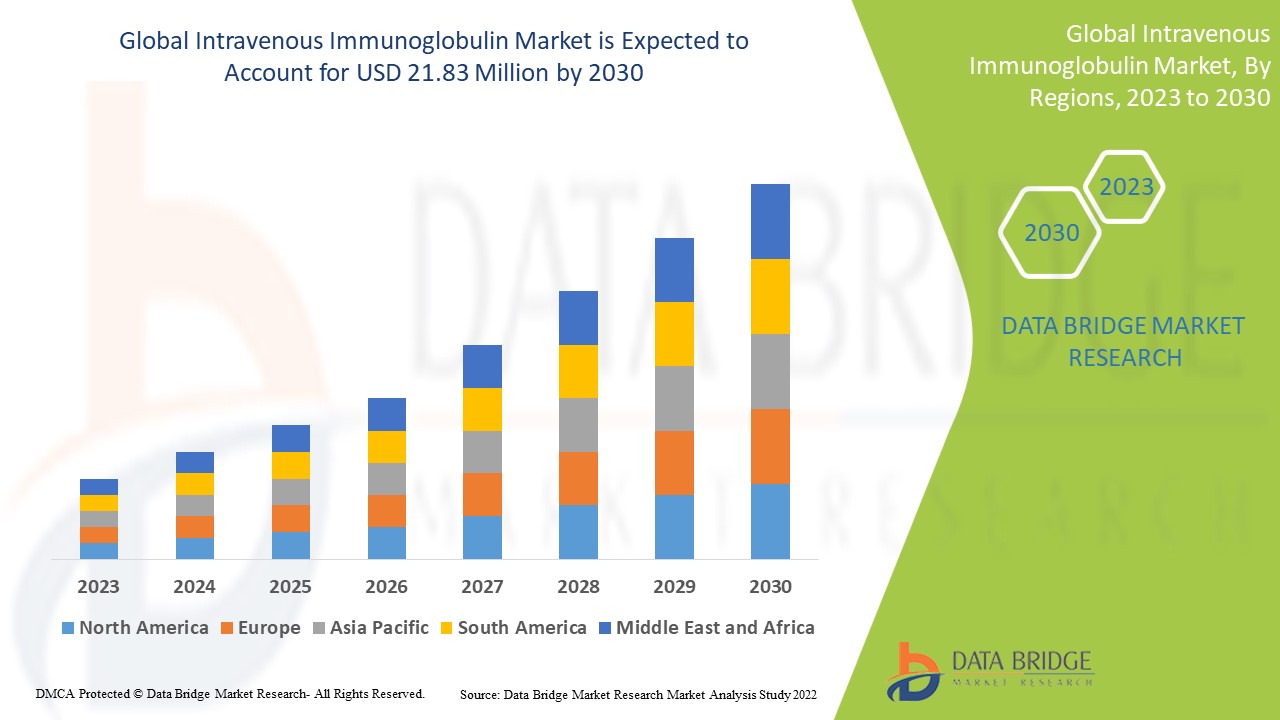
Data Bridge Market Research analyses a growth rate in the intravenous immunoglobulin market in the forecast period 2023-2030. The expected CAGR of intravenous immunoglobulin market is tend to be around 7.80% in the mentioned forecast period. The market was valued at USD 11.97 million in 2022, and it would grow upto USD 21.83 million by 2030. In addition to the market insights such as market value, growth rate, market segments, geographical coverage, market players, and market scenario, the market report curated by the Data Bridge Market Research team also includes in-depth expert analysis, patient epidemiology, pipeline analysis, pricing analysis, and regulatory framework.
Intravenous Immunoglobulin Market Scope and Segmentation
|
Report Metric |
Details |
|
Forecast Period |
2023 to 2030 |
|
Base Year |
2022 |
|
Historic Years |
2021 (Customizable to 2015 - 2020) |
|
Quantitative Units |
Revenue in USD Million, Volumes in Units, Pricing in USD |
|
Segments Covered |
Application (Hypogammaglobulinemia, CIDP, Congential AIDS), Route of Administration (Intravenous and Subcutaneous), End-Users (Hospitals, Homecare, Specialty Clinics, Others), Distribution Channel (Hospital Pharmacy, Retail Pharmacy, online Pharmacies and Others) |
|
Countries Covered |
U.S., Canada and Mexico in North America, Germany, France, U.K., Netherlands, Switzerland, Belgium, Russia, Italy, Spain, Turkey, Rest of Europe in Europe, China, Japan, India, South Korea, Singapore, Malaysia, Australia, Thailand, Indonesia, Philippines, Rest of Asia-Pacific (APAC) in the Asia-Pacific (APAC), Saudi Arabia, U.A.E, South Africa, Egypt, Israel, Rest of Middle East and Africa (MEA) as a part of Middle East and Africa (MEA), Brazil, Argentina and Rest of South America as part of South America |
|
Market Players Covered |
Baxter (U.S.), Bayer AG (Germany), Merck & Co., Inc. (U.S.), Biotest AG (Germany), Top Bio Group Co., Ltd. (China), CSL Limited (Australia), Octapharma AG (Switzerland), Grifols, S.A (Spain), Kedrion S.p.A (Italy) |
|
Market Opportunities |
|
Market Definition
Immunoglobulin is the product breakdown of blood plasma that contains antibody. Immunoglobulin is a blood product which is generally administered through intravenous route. It consists of polyvalent IgG antibodies extracted from blood donors' plasma. Many patients are suffering from disorders such Wiskott Aldrich Syndrome, X-linked agammaglobulinemia who are unable to produce enough antibodies. Thus they benefit from intravenous immunoglobulin to maintain the sufficient level of antibodies in the body.
Global Intravenous Immunoglobulin Market Dynamics
Drivers
- Growing Incidences of Primary Immune Deficiencies (PIDs)
As per W.H.O, at present, there are more than 50 different Primary Immune Deficiencies (PIDs) such as specific antibody deficiency and X-lined hypo-gamma-globulinemia. This deficiences include 176 different types of rare hereditary disorders. As per the U.S. National Library of Medicine and National Institute of Health, around 6 million patients are suffering from PID globally. Increasing occurrences of these diseases are anticipated to boost the demand for immunoglobulin therapies during the forecast period.
- Growing Demand For Hospitals For These Patients
The hospitals segment dominated the market due to increase in the number of patients opting for hospital treatment rather than standalone clinics. Furthermore, patients receiving IVIG infusions at home have improved the quality of life, as they get more comfortable in the daily activities. For instance, IVIG infusions at home care have minimized the scheduling clashes for multiple patients at a time.
Opportunities
- Increasing Healthcare Expenditure For Immunoglobulins
There has been increasing funding for several immunoglobulins that are helping to expand the market's growth. For instance, The National Institute of Allergy and Infectious Diseases (NIAID), part of the National Institutes of Health, sponsored and funded the phase III trial, known as Inpatient Treatment with Anti-Coronavirus Immunoglobulin, or ITAC. The antibody solution which was tested in the ITAC trial is anti-coronavirus hyperimmune intravenous immunoglobulin or hIVIG.
- Increasing Strategic Partnerships and Acquisitions between Organizations
Several market players are adopting numerous strategies such as mergers and acquisitions, distribution, and other collaborative agreements to gain a huge share by growing their market penetration. For instance, on 25 April 2022, Grifols S.A purchased Biotest, which is a significant and transformative deal that will boost growth and innovation. This deal will allow to accelerate and extend its product range, increase patient access to plasma medicines, operate the biggest private European network of plasma facilities, and increase revenue growth and margin development.
Restraints/Challenges
- High Cost of Treatment
The cost for diagnosis and treatment of demyelinating disease is very high. The increasing cost of IVIG therapy is estimated to be a major barrier to market expansion. Immunoglobulin infusions are typically given once every 3 to 4 weeks, and the therapy is ongoing about 12-16 sessions annually. The projected cost of IVIG is USD 73.89 per gram, with a total cost of USD 10,000, depending on the severity of the patient condition.
This intravenous immunoglobulin market report provides details of new recent developments, trade regulations, import-export analysis, production analysis, value chain optimization, market share, impact of domestic and localized market players, analyses opportunities in terms of emerging revenue pockets, changes in market regulations, strategic market growth analysis, market size, category market growths, application niches and dominance, product approvals, product launches, geographic expansions, technological innovations in the market. To gain more info on the intravenous immunoglobulin market contact Data Bridge Market Research for an Analyst Brief, our team will help you take an informed market decision to achieve market growth.
COVID-19 Impact on Global Intravenous Immunoglobulin Market
COVID-19 has caused significant issues for patients with mental and neurological disorders because of their increased susceptibility to infection. MIS-C (multi-system inflammatory syndrome) has been witnessed in several patients, especially among the younger population. Several medical professionals have been estimating the efficacy of IVIG injection for COVID-19 patients. For instance, in April 2021, Grifols, S.A. which is a key provider of plasma-derived medicines, contributed to a research study in collaboration with both the NIH and NIAID, to analyze an intravenous anti-SARS-CoV-2 hyperimmune globulin among outpatients, which is projected to boost the market growth during the forecast period.
Recent Development:
- In August 2021, Emergent BioSolutions, Inc., which is a multinational specialty biopharmaceutical company based in the U.S., stated the initiation of phase III clinical trial to assess its investigational SARS-CoV-2 Immune Globulin Intravenous (COVID-HIG) plasma-derived therapy as a capable outpatient treatment for patients with COVID-19 who are at higher risk of progression to severe disease. Therefore, the factors mentioned above are estimated to boost the growth of the global intravenous immunoglobulin market during the forecast period.
Global Intravenous Immunoglobulin Market Scope
The intravenous immunoglobulin market is segmented on the basis of application, route of administration, end-user, distribution channel. The growth amongst these segments will help you analyze meagre growth segments in the industries and provide the users with a valuable market overview and market insights to help them make strategic decisions for identifying core market applications.
Application
- Hypogammaglobulinemia
- CIDP
- Congential AIDS
Route of Administration
- Intravenous
- Subcutaneous
End-Users
- Hospitals
- Homecare
- Specialty Clinics
- Others
Distribution Channel
- Hospital Pharmacy
- Retail Pharmacy
- Online Pharmacies
- Others
Intravenous Immunoglobulin Market Regional Analysis/Insights
The intravenous immunoglobulin market is analyzed and market size insights and trends are provided by application, route of administration,end-user, distribution channel as referenced above.
The major countries covered in the intravenous immunoglobulin market report are the U.S., Canada and Mexico in North America, Germany, France, U.K., Netherlands, Switzerland, Belgium, Russia, Italy, Spain, Turkey, Rest of Europe in Europe, China, Japan, India, South Korea, Singapore, Malaysia, Australia, Thailand, Indonesia, Philippines, Rest of Asia-Pacific (APAC) in the Asia-Pacific (APAC), Saudi Arabia, U.A.E, South Africa, Egypt, Israel, Rest of Middle East and Africa (MEA) as a part of Middle East and Africa (MEA), Brazil, Argentina and Rest of South America as part of South America.
North America is considered to have the highest lucrative growth in the forecast period due to the presence of key manufacturers of the product is high and, increasing research and development activities, healthcare expenditure.
Asia-Pacific is dominating the market due to the growing awareness and potential opportunities for adopting immunoglobulin-based therapies for treating patients with primary immune deficiencies, along with a growing geriatric population.
The country section of the report also provides individual market impacting factors and changes in regulation in the market domestically that impacts the current and future trends of the market. Also, the presence and availability of global brands and their challenges faced due to large or scarce competition from local and domestic brands, impact of domestic tariffs and trade routes are considered while providing forecast analysis of the country data.
Competitive Landscape and Global Intravenous Immunoglobulin Market Share Analysis
The intravenous immunoglobulin market competitive landscape provides details by competitor. Details included are company overview, company financials, revenue generated, market potential, investment in research and development, new market initiatives, global presence, production sites and facilities, production capacities, company strengths and weaknesses, product launch, product width and breadth, application dominance. The above data points provided are only related to the companies' focus related to intravenous immunoglobulin market.
Key players operating in the intravenous immunoglobulin market include:
- Baxter (U.S.)
- Bayer AG (Germany)
- Merck & Co., Inc. (U.S.)
- Biotest AG (Germany)
- Top Bio Group Co., Ltd.(China)
- CSL Limited (Australia)
- Octapharma AG (Switzerland)
- Grifols, S.A (Spain)
- Kedrion S.p.A (Italy)
SKU-
Get online access to the report on the World's First Market Intelligence Cloud
- Interactive Data Analysis Dashboard
- Company Analysis Dashboard for high growth potential opportunities
- Research Analyst Access for customization & queries
- Competitor Analysis with Interactive dashboard
- Latest News, Updates & Trend analysis
- Harness the Power of Benchmark Analysis for Comprehensive Competitor Tracking
Table of Content
1 INTRODUCTION
1.1 OBJECTIVES OF THE STUDY
1.2 MARKET DEFINITION
1.3 OVERVIEW OF GLOBAL INTRAVENOUS IMMUNOGLOBULIN MARKET
1.4 CURRENCY AND PRICING
1.5 LIMITATION
1.6 MARKETS COVERED
2 MARKET SEGMENTATION
2.1 KEY TAKEAWAYS
2.2 ARRIVING AT THE GLOBAL INTRAVENOUS IMMUNOGLOBULIN MARKET SIZE
2.2.1 VENDOR POSITIONING GRID
2.2.2 TECHNOLOGY LIFE LINE CURVE
2.2.3 TRIPOD DATA VALIDATION MODEL
2.2.4 MARKET GUIDE
2.2.5 MULTIVARIATE MODELLING
2.2.6 TOP TO BOTTOM ANALYSIS
2.2.7 CHALLENGE MATRIX
2.2.8 APPLICATION COVERAGE GRID
2.2.9 STANDARDS OF MEASUREMENT
2.2.10 VENDOR SHARE ANALYSIS
2.2.11 EPIDEMIOLOGY MODELING
2.2.12 DATA POINTS FROM KEY PRIMARY INTERVIEWS
2.2.13 DATA POINTS FROM KEY SECONDARY DATABASES
2.3 GLOBAL INTRAVENOUS IMMUNOGLOBULIN MARKET: RESEARCH SNAPSHOT
2.4 ASSUMPTIONS
3 EXECUTIVE SUMMARY
4 PREMIUM INSIGHTS
4.1 PORTER'S 5 FORCES
4.2 PESTEL ANALYSIS
4.3 ANALYSIS OF TRADITIONAL AND NOVEL PRODUCTS
5 INDUSTRY INSIGHTS
5.1 PATENT ANALYSIS
5.1.1 PATENT LANDSCAPE
5.1.2 USPTO NUMBER
5.1.3 PATENT EXPIRY
5.1.4 EPIO NUMBER
5.1.5 PATENT STRENGTH AND QUALITY
5.1.6 PATENT CLAIMS
5.1.7 PATENT CITATIONS
5.1.8 PATENT LITIGATION AND LICENSING
5.1.9 FILE OF PATENT
5.1.10 PATENT RECEIVED CONTRIES
5.1.11 TECHNOLOGY BACKGROUND
5.2 DRUG TREATMENT RATE BY MATURED MARKETS
5.3 DEMOGRAPHIC TRENDS: IMPACTS ON ALL INCIDENCE RATES
5.4 PATIENT FLOW DIAGRAM
5.5 KEY PRICING STRATEGIES
5.6 KEY PATIENT ENROLLMENT STRATEGIES
5.7 INTERVIEWS WITH SPECIALIST
5.8 OTHER KOL SNAPSHOTS
6 EPIDEMOLOGY
6.1 INCIDENCE RATE
6.2 TREATMENT RATE
6.3 MORTALITY RATE
6.4 DRUG ADHERENCE AND THERAPY SWITCH MODEL
6.5 PATIENT TREATMENT SUCCESS RATES
7 MERGERS AND ACQUISITION
7.1 LICENSING
7.2 COMMERCIALIZATION AGREEMENTS
8 REGULATORY FRAMEWORK
8.1 REGULATORY APPROVAL PROCESS
8.2 GEOGRAPHIES' EASE OF REGULATORY APPROVAL
8.3 REGULATORY APPROVAL PATHWAYS
8.4 LICENSING AND REGISTRATION
8.5 POST-MARKETING SURVEILLANCE
8.6 GOOD MANUFACTURING PRACTICES (GMPS) GUIDELINES
9 PIPELINE ANALYSIS
9.1 CLINICAL TRIALS AND PHASE ANALYSIS
9.2 DRUG THERAPY PIPELINE
9.3 PHASE III CANDIDATES
9.4 PHASE II CANDIDATES
9.5 PHASE I CANDIDATES
9.6 OTHERS (PRE-CLINICAL AND RESEARCH)
10 MARKETED DRUG ANALYSIS
10.1 DRUG
10.1.1 BRAND NAME
10.1.2 GENERICS NAME
10.2 THERAPEUTIC INDIACTION
10.3 PHARACOLOGICAL CLASS OD THE DRUG
10.4 DRUG PRIMARY INDICATION
10.5 MARKET STATUS
10.6 MEDICATION TYPE
10.7 DRUG DOSAGES FORM
10.8 DOSAGES AVAILABILITY
10.9 PACKAGING TYPE
10.1 DRUG ROUTE OF ADMINISTRATION
10.11 DOSING FREQUENCY
10.12 DRUG INSIGHT
10.13 AN OVERVIEW OF THE DRUG DEVELOPMENT ACTIVITIES SUCH AS REGULATORY MILSTONE, SAFETY DATA AND EFFICACY DATA, MARKET EXCLUSIVITY DATA.
10.13.1 FORECAST MARKET OUTLOOK
10.13.2 CROSS COMPETITION
10.13.3 THERAPEUTIC PORTFOLIO
10.14 CURRENT DEVELOPMENT SCENARIO
11 MARKET OVERVIEW
11.1 DRIVERS
11.2 RESTRAINS
11.3 OPPURTUNITY
11.4 CHALLENGES
12 MARKET ACCESS
12.1 10-YEAR MARKET FORECAST
12.2 CLINICAL TRIAL RECENT UPDATES
12.3 ANNUAL NEW FDA APPROVED DRUGS
12.4 DRUGS MANUFACTURER AND DEALS
12.5 MAJOR DRUG UPTAKE
12.6 CURRENT TREATMENT PRACTICES
12.7 IMPACT OF UPCOMING THERAPY
13 R & D ANALYSIS
13.1 COMPARATIVE ANALYSIS
13.2 DRUG DEVELOPMENTAL LANDSCAPE
13.3 IN-DEPTH INSIGHTS ON REGULATORY MILESTONES
13.4 THERAPEUTIC ASSESSMENT
13.5 ASSET-BASED COLLABORATIONS AND PARTNERSHIPS
14 GLOBAL INTRAVENOUS IMMUNOGLOBULIN MARKET, BY TYPE
(NOTE: MARKET VALUE, VOLUME AND ASP ANALYSIS WOULD BE PROVIDED FOR ALL SEGMENTS AND SUB-SEGMENTS OF PRODUCT)
14.1 OVERVIEW
14.2 IGG
14.2.1 IGG1
14.2.2 IGG2
14.2.3 IGG3
14.2.4 IGG4
14.3 IGA
14.3.1 IGA1
14.3.2 IGA2
14.4 IGM
14.5 IGE
14.6 IGD
15 GLOBAL INTRAVENOUS IMMUNOGLOBULIN MARKET, BY INDICATION
15.1 OVERVIEW
15.2 PRIMARY HUMORAL IMMUNODEFICIENCY DISEASE (PIDD)
15.2.1 COMMON VARIABLE IMMUNODEFICIENCY (CVID)
15.2.2 X-LINKED AGAMMAGLOBULINEMIA
15.2.3 CONGENITAL AGAMMAGLOBULINEMIA
15.2.4 WISKOTT-ALDRICH SYNDROME
15.2.5 SEVERE COMBINED IMMUNODEFICIENCIES (SCID)
15.3 IDIOPATHIC THROMBOCYTOPENIC PURPURA (ITP)
15.4 CHRONIC INFLAMMATORY DEMYELINATING POLYNEUROPATHY (CIDP)
15.5 KAWASAKI’S VASCULITIS
15.6 MYASTHENIA GRAVIS
15.6.1 GENERALIZED MYASTHENIA GRAVIS
15.6.2 OCULAR MYASTHENIA GRAVIS
15.6.3 TRANSIENT NEONATAL MYASTHENIA GRAVIS
15.7 LUPUS
15.8 MYOSITIS
15.9 MULTIFOCAL MOTOR NEUROPATHY
15.1 GUILLAIN BARRÉ SYNDROME
15.11 HYPOPLASMINOGENEMIA
15.11.1 HEREDITARY
15.11.2 NONFAMILIAL
15.12 ACQUIRED IMMUNODIFICIENCY DISORDERS
15.13 CANCER
15.13.1 CHRONIC LYMPHOCYTIC LYMPHOMA
15.13.2 MULTIPLE MYELOMA
15.14 OTHER INDICATIONS
16 GLOBAL INTRAVENOUS IMMUNOGLOBULIN MARKET, BY CONCENTRATION
16.1 OVERVIEW
16.2 IVIG 5%
16.3 IVIG 10%
17 GLOBAL INTRAVENOUS IMMUNOGLOBULIN MARKET, BY AGE GROUP
17.1 OVERVIEW
17.2 PEDIATRIC
17.3 ADULT
17.4 GERIATRIC
18 GLOBAL INTRAVENOUS IMMUNOGLOBULIN MARKET, BY DRUG TYPE
18.1 OVERVIEW
18.2 BRANDED DRUGS
18.2.1 GAMUNEX
18.2.2 FLEBOGAMMA DIF
18.2.3 GAMMAGARD S/D
18.2.4 GAMMAGARD LIQUID
18.2.5 GAMMAKED
18.2.6 GAMMAPLEX
18.2.7 GAMUNEX-C
18.2.8 OCTAGAM
18.2.9 PRIVIGEN
18.3 GENERIC
19 GLOBAL INTRAVENOUS IMMUNOGLOBULIN MARKET, BY END USER
19.1 OVERVIEW
19.2 HOSPITALS
19.3 SPECIALTY CLINIC
19.4 HOMECARE
19.5 OTHERS
20 GLOBAL INTRAVENOUS IMMUNOGLOBULIN MARKET, BY DISTRIBUTION CHANNEL
20.1 OVERVIEW
20.2 HOSPITAL PHARMACY
20.3 RETAIL PHARMACY
20.4 OTHERS
21 GLOBAL INTRAVENOUS IMMUNOGLOBULIN MARKET, SWOT AND DBMR ANALYSIS
22 GLOBAL INTRAVENOUS IMMUNOGLOBULIN MARKET, COMPANY LANDSCAPE
22.1 COMPANY SHARE ANALYSIS: NORTH AMERICA
22.2 COMPANY SHARE ANALYSIS: EUROPE
22.3 COMPANY SHARE ANALYSIS: ASIA-PACIFIC
22.4 MERGERS & ACQUISITIONS
22.5 NEW PRODUCT DEVELOPMENT & APPROVALS
22.6 EXPANSIONS
22.7 REGULATORY CHANGES
22.8 PARTNERSHIP AND OTHER STRATEGIC DEVELOPMENTS
23 GLOBAL INTRAVENOUS IMMUNOGLOBULIN MARKET, BY GEOGRAPHY
GLOBAL INTRAVENOUS IMMUNOGLOBULIN MARKET, (ALL SEGMENTATION PROVIDED ABOVE IS REPRESENTED IN THIS CHAPTER BY COUNTRY)
23.1 NORTH AMERICA
23.1.1 U.S.
23.1.1.1. U.S. INTRAVENOUS IMMUNOGLOBULIN MARKET, BY TYPE
23.1.1.2. U.S. INTRAVENOUS IMMUNOGLOBULIN MARKET, BY INDICATION
23.1.1.3. U.S. INTRAVENOUS IMMUNOGLOBULIN MARKET, BY CONCENTRATION
23.1.1.4. U.S. INTRAVENOUS IMMUNOGLOBULIN MARKET, BY AGE GROUP
23.1.1.5. U.S. INTRAVENOUS IMMUNOGLOBULIN MARKET, BY DRUG TYPE
23.1.1.6. U.S. INTRAVENOUS IMMUNOGLOBULIN MARKET, BY END USER
23.1.1.7. U.S. INTRAVENOUS IMMUNOGLOBULIN MARKET, BY DISTRUBUTION CHANNEL
23.1.2 CANADA
23.1.3 MEXICO
23.2 EUROPE
23.2.1 GERMANY
23.2.2 FRANCE
23.2.3 U.K.
23.2.4 HUNGARY
23.2.5 LITHUANIA
23.2.6 AUSTRIA
23.2.7 IRELAND
23.2.8 NORWAY
23.2.9 POLAND
23.2.10 ITALY
23.2.11 SPAIN
23.2.12 RUSSIA
23.2.13 TURKEY
23.2.14 NETHERLANDS
23.2.15 SWITZERLAND
23.2.16 REST OF EUROPE
23.3 ASIA-PACIFIC
23.3.1 JAPAN
23.3.2 CHINA
23.3.3 SOUTH KOREA
23.3.4 INDIA
23.3.5 AUSTRALIA
23.3.6 SINGAPORE
23.3.7 THAILAND
23.3.8 MALAYSIA
23.3.9 INDONESIA
23.3.10 PHILIPPINES
23.3.11 VIETNAM
23.3.12 REST OF ASIA-PACIFIC
23.4 SOUTH AMERICA
23.4.1 BRAZIL
23.4.2 ARGENTINA
23.4.3 PERU
23.4.4 COLOMBIA
23.4.5 VENEZUELA
23.4.6 REST OF SOUTH AMERICA
23.5 MIDDLE EAST AND AFRICA
23.5.1 SOUTH AFRICA
23.5.2 SAUDI ARABIA
23.5.3 UAE
23.5.4 EGYPT
23.5.5 KUWAIT
23.5.6 ISRAEL
23.5.7 REST OF MIDDLE EAST AND AFRICA
23.6 KEY PRIMARY INSIGHTS: BY MAJOR COUNTRIES
24 GLOBAL INTRAVENOUS IMMUNOGLOBULIN MARKET, COMPANY PROFILE
24.1 OCTAPHARMA AG
24.1.1 COMPANY OVERVIEW
24.1.2 REVENUE ANALYSIS
24.1.3 GEOGRAPHIC PRESENCE
24.1.4 PRODUCT PORTFOLIO
24.1.5 RECENT DEVELOPMENTS
24.2 GRIFOLS
24.2.1 COMPANY OVERVIEW
24.2.2 REVENUE ANALYSIS
24.2.3 GEOGRAPHIC PRESENCE
24.2.4 PRODUCT PORTFOLIO
24.2.5 RECENT DEVELOPMENTS
24.3 TAKEDA PHARMACEUTICALS U.S.A., INC.
24.3.1 COMPANY OVERVIEW
24.3.2 REVENUE ANALYSIS
24.3.3 GEOGRAPHIC PRESENCE
24.3.4 PRODUCT PORTFOLIO
24.3.5 RECENT DEVELOPMENTS
24.4 ADMA BIOLOGICS.
24.4.1 COMPANY OVERVIEW
24.4.2 REVENUE ANALYSIS
24.4.3 GEOGRAPHIC PRESENCE
24.4.4 PRODUCT PORTFOLIO
24.4.5 RECENT DEVELOPMENTS
24.5 BIOTEST UK.
24.5.1 COMPANY OVERVIEW
24.5.2 REVENUE ANALYSIS
24.5.3 GEOGRAPHIC PRESENCE
24.5.4 PRODUCT PORTFOLIO
24.5.5 RECENT DEVELOPMENTS
24.6 CSL
24.6.1 COMPANY OVERVIEW
24.6.2 REVENUE ANALYSIS
24.6.3 GEOGRAPHIC PRESENCE
24.6.4 PRODUCT PORTFOLIO
24.6.5 RECENT DEVELOPMENTS
24.7 KEDRION BIOPHARMA INC.
24.7.1 COMPANY OVERVIEW
24.7.2 REVENUE ANALYSIS
24.7.3 GEOGRAPHIC PRESENCE
24.7.4 PRODUCT PORTFOLIO
24.7.5 RECENT DEVELOPMENTS
24.8 PROTHYA BIOSOLUTION
24.8.1 COMPANY OVERVIEW
24.8.2 REVENUE ANALYSIS
24.8.3 GEOGRAPHIC PRESENCE
24.8.4 PRODUCT PORTFOLIO
24.8.5 RECENT DEVELOPMENTS
24.9 MERCK & CO., INC.
24.9.1 COMPANY OVERVIEW
24.9.2 REVENUE ANALYSIS
24.9.3 GEOGRAPHIC PRESENCE
24.9.4 PRODUCT PORTFOLIO
24.9.5 RECENT DEVELOPMENTS
24.1 KAMADA PHARMACEUTICALS.
24.10.1 COMPANY OVERVIEW
24.10.2 REVENUE ANALYSIS
24.10.3 GEOGRAPHIC PRESENCE
24.10.4 PRODUCT PORTFOLIO
24.10.5 RECENT DEVELOPMENTS
24.11 EMERGENT
24.11.1 COMPANY OVERVIEW
24.11.2 REVENUE ANALYSIS
24.11.3 GEOGRAPHIC PRESENCE
24.11.4 PRODUCT PORTFOLIO
24.11.5 RECENT DEVELOPMENTS
24.12 LFB ( LABORATOIRE FRANÇAIS DU FRACTIONNEMENT DES BIOTECHNOLOGIES )
24.12.1 COMPANY OVERVIEW
24.12.2 REVENUE ANALYSIS
24.12.3 GEOGRAPHIC PRESENCE
24.12.4 PRODUCT PORTFOLIO
24.12.5 RECENT DEVELOPMENTS
24.13 BIO PRODUCTS LABORATORY LTD.
24.13.1 COMPANY OVERVIEW
24.13.2 REVENUE ANALYSIS
24.13.3 GEOGRAPHIC PRESENCE
24.13.4 PRODUCT PORTFOLIO
24.13.5 RECENT DEVELOPMENTS
24.14 CHINA BIOLOGIC PRODUCTS HOLDINGS, INC.
24.14.1 COMPANY OVERVIEW
24.14.2 REVENUE ANALYSIS
24.14.3 GEOGRAPHIC PRESENCE
24.14.4 PRODUCT PORTFOLIO
24.14.5 RECENT DEVELOPMENTS
24.15 JOHNSON AND JOHNSON
24.15.1 COMPANY OVERVIEW
24.15.2 REVENUE ANALYSIS
24.15.3 GEOGRAPHIC PRESENCE
24.15.4 PRODUCT PORTFOLIO
24.15.5 RECENT DEVELOPMENTS
24.16 GC BIOPHARMA CORP.
24.16.1 COMPANY OVERVIEW
24.16.2 REVENUE ANALYSIS
24.16.3 GEOGRAPHIC PRESENCE
24.16.4 PRODUCT PORTFOLIO
24.16.5 RECENT DEVELOPMENTS
24.17 NOVARTIS AG
24.17.1 COMPANY OVERVIEW
24.17.2 REVENUE ANALYSIS
24.17.3 GEOGRAPHIC PRESENCE
24.17.4 PRODUCT PORTFOLIO
24.17.5 RECENT DEVELOPMENTS
24.18 BIOPHARMA PLASMA
24.18.1 COMPANY OVERVIEW
24.18.2 REVENUE ANALYSIS
24.18.3 GEOGRAPHIC PRESENCE
24.18.4 PRODUCT PORTFOLIO
24.18.5 RECENT DEVELOPMENTS
24.19 SYNERMORE BIOLOGICS
24.19.1 COMPANY OVERVIEW
24.19.2 REVENUE ANALYSIS
24.19.3 GEOGRAPHIC PRESENCE
24.19.4 PRODUCT PORTFOLIO
24.19.5 RECENT DEVELOPMENTS
24.2 RELIANCE LIFE SCIENCES
24.20.1 COMPANY OVERVIEW
24.20.2 REVENUE ANALYSIS
24.20.3 GEOGRAPHIC PRESENCE
24.20.4 PRODUCT PORTFOLIO
24.20.5 RECENT DEVELOPMENTS
24.21 TAJ PHARMA GROUP
24.21.1 COMPANY OVERVIEW
24.21.2 REVENUE ANALYSIS
24.21.3 GEOGRAPHIC PRESENCE
24.21.4 PRODUCT PORTFOLIO
24.21.5 RECENT DEVELOPMENTS
25 RELATED REPORTS
26 CONCLUSION
27 QUESTIONNAIRE
28 ABOUT DATA BRIDGE MARKET RESEARCH

Research Methodology
Data collection and base year analysis are done using data collection modules with large sample sizes. The stage includes obtaining market information or related data through various sources and strategies. It includes examining and planning all the data acquired from the past in advance. It likewise envelops the examination of information inconsistencies seen across different information sources. The market data is analysed and estimated using market statistical and coherent models. Also, market share analysis and key trend analysis are the major success factors in the market report. To know more, please request an analyst call or drop down your inquiry.
The key research methodology used by DBMR research team is data triangulation which involves data mining, analysis of the impact of data variables on the market and primary (industry expert) validation. Data models include Vendor Positioning Grid, Market Time Line Analysis, Market Overview and Guide, Company Positioning Grid, Patent Analysis, Pricing Analysis, Company Market Share Analysis, Standards of Measurement, Global versus Regional and Vendor Share Analysis. To know more about the research methodology, drop in an inquiry to speak to our industry experts.
Customization Available
Data Bridge Market Research is a leader in advanced formative research. We take pride in servicing our existing and new customers with data and analysis that match and suits their goal. The report can be customized to include price trend analysis of target brands understanding the market for additional countries (ask for the list of countries), clinical trial results data, literature review, refurbished market and product base analysis. Market analysis of target competitors can be analyzed from technology-based analysis to market portfolio strategies. We can add as many competitors that you require data about in the format and data style you are looking for. Our team of analysts can also provide you data in crude raw excel files pivot tables (Fact book) or can assist you in creating presentations from the data sets available in the report.













