Global Kras G12d Inhibitor Drug Market
Market Size in USD Million
CAGR :
% 
 USD
842.00 Million
USD
3,972.47 Million
2025
2033
USD
842.00 Million
USD
3,972.47 Million
2025
2033
| 2026 - 2033 | |
| USD 842.00 Million | |
| USD 3,972.47 Million | |
|
|
|
|
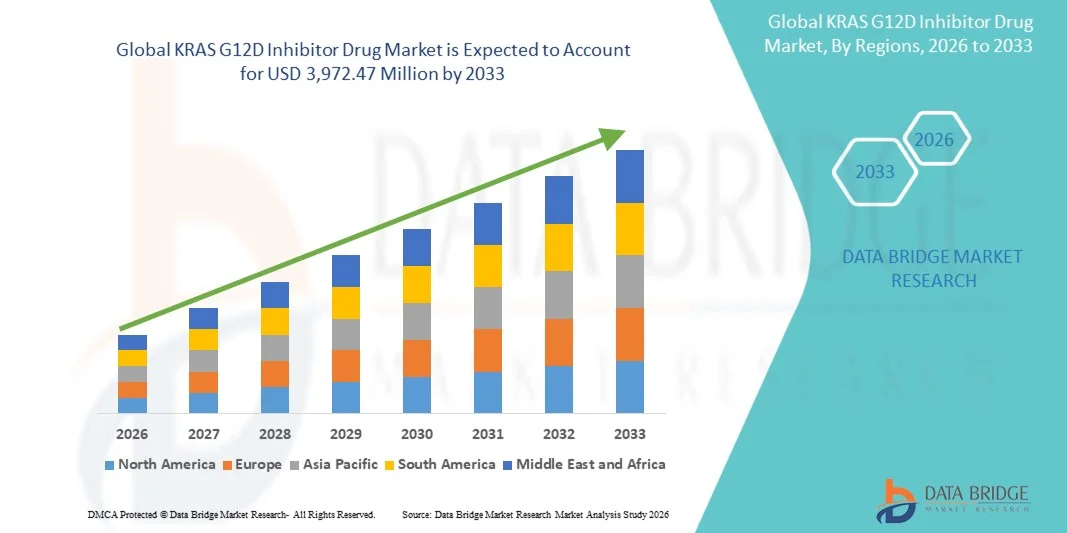
KRAS G12D Inhibitor Drug Market Size
- The global KRAS G12D inhibitor drug market size was valued at USD 842.00 million in 2025 and is expected to reach USD 3,972.47 million by 2033, at a CAGR of 21.40% during the forecast period
- The market growth is primarily driven by the rising prevalence of KRAS-mutated cancers, including pancreatic, colorectal, and non-small cell lung cancers, along with increasing demand for precision oncology and targeted therapies
- Furthermore, rapid advancements in oncology drug discovery, expansion of clinical pipelines for KRAS G12D-specific inhibitors, and growing investments from pharmaceutical and biotechnology companies are accelerating drug development, thereby significantly boosting the industry’s growth
KRAS G12D Inhibitor Drug Market Analysis
- KRAS G12D inhibitor drugs, designed to selectively target the KRAS G12D mutation responsible for driving tumor growth, are emerging as a key class of precision oncology therapeutics with strong potential in treating hard-to-treat solid tumors such as pancreatic, colorectal, and lung cancers due to their ability to block aberrant signaling pathways at the molecular level
- The growing demand for KRAS G12D inhibitor drugs is primarily driven by the rising burden of KRAS-mutated cancers, increasing focus on targeted cancer therapies, and rapid expansion of oncology R&D pipelines supported by strong clinical trial activity and breakthrough drug design technologies
- North America dominated the KRAS G12D inhibitor drug market with the largest revenue share of 42.6% in 2025, supported by advanced oncology infrastructure, high clinical trial density, strong funding from biotechnology firms, and early adoption of novel targeted therapies, particularly in the U.S. where precision medicine initiatives are highly developed
- Asia-Pacific is expected to be the fastest growing region in the KRAS G12D inhibitor drug market during the forecast period, driven by increasing cancer incidence, expanding biotechnology investments, improving clinical research capabilities, and growing participation in global oncology trials across countries such as China, Japan, and India
- Small molecule inhibitors segment dominated the KRAS G12D inhibitor drug market with the largest share of 58.3% in 2025, owing to their high specificity, oral bioavailability potential, and strong clinical advancement compared to other emerging modalities such as biologics and RNA-based therapies
Report Scope and KRAS G12D Inhibitor Drug Market Segmentation
|
Attributes |
KRAS G12D Inhibitor Drug Key Market Insights |
|
Segments Covered |
|
|
Countries Covered |
North America
Europe
Asia-Pacific
Middle East and Africa
South America
|
|
Key Market Players |
|
|
Market Opportunities |
|
|
Value Added Data Infosets |
In addition to the insights on market scenarios such as market value, growth rate, segmentation, geographical coverage, and major players, the market reports curated by the Data Bridge Market Research also include in-depth expert analysis, patient epidemiology, pipeline analysis, pricing analysis, and regulatory framework |
KRAS G12D Inhibitor Drug Market Trends
“Expansion of Precision Oncology and Mutation-Specific Drug Design”
- A significant and accelerating trend in the global KRAS G12D inhibitor drug market is the increasing focus on mutation-specific precision oncology therapies designed to selectively inhibit KRAS G12D-driven tumor signaling pathways in hard-to-treat solid cancers
- For instance, KRAS G12D-selective small molecule programs such as MRTX1133 by Mirati Therapeutics (Bristol Myers Squibb) are advancing in preclinical and early clinical studies targeting pancreatic and colorectal cancers
- The development of next-generation covalent and non-covalent inhibitors is enabling improved binding specificity and overcoming limitations of earlier KRAS-targeting approaches, thereby enhancing therapeutic potential and durability of response
- The integration of biomarker-driven patient selection and companion diagnostics is supporting more precise clinical trial enrollment and improving success rates in late-stage oncology development programs
- Increasing collaboration between biotechnology firms and academic research institutes is accelerating early-stage discovery of novel KRAS G12D inhibitory compounds with improved efficacy and safety profiles
- The rising use of AI-driven drug discovery platforms is enabling faster identification of KRAS G12D binding candidates and optimizing lead compound selection in oncology pipelines
KRAS G12D Inhibitor Drug Market Dynamics
Driver
“Rising Cancer Burden and Strong Oncology R&D Investments”
- The increasing prevalence of KRAS G12D-mutated cancers, particularly in pancreatic, colorectal, and lung cancer patients, is a major driver accelerating demand for targeted therapeutic options
- For instance, large pharmaceutical companies such as Amgen, Eli Lilly, and Bristol Myers Squibb are actively expanding oncology pipelines with significant investments in KRAS-targeted drug discovery programs
- Growing adoption of precision medicine and molecular diagnostics is enabling earlier identification of KRAS mutations, supporting higher uptake of targeted treatment approaches in clinical settings
- Furthermore, expanding clinical trial activity across North America, Europe, and Asia-Pacific is accelerating drug development timelines and improving commercialization prospects for KRAS G12D inhibitors
- Increasing healthcare funding and government support for oncology innovation are further strengthening the ecosystem for advanced targeted therapy development
- Rising collaborations between biotech startups and big pharma companies are accelerating licensing agreements and co-development of KRAS G12D inhibitor candidates
- Expanding patient access programs and orphan drug incentives are further encouraging investment in rare mutation-specific oncology drug development
Restraint/Challenge
“High Drug Development Complexity and Resistance Mechanisms”
- The significant challenges related to drug design complexity and limited structural targeting options for KRAS G12D mutation are restraining rapid development and commercialization of effective therapies
- For instance, unlike KRAS G12C, the G12D mutation lacks a highly reactive binding pocket, making it more difficult for small molecule inhibitors to achieve strong and sustained inhibition
- Emerging resistance mechanisms in tumor cells, including pathway reactivation and compensatory signaling through MAPK and PI3K pathways, further reduce long-term treatment efficacy
- Additionally, high clinical trial failure rates and long development timelines increase R&D costs and delay potential market entry of promising drug candidates
- Limited availability of validated biomarkers for patient stratification reduces treatment precision and slows down regulatory approval pathways
- Strict regulatory requirements for oncology drugs, including extensive safety and efficacy validation, further extend approval timelines and increase development uncertain
KRAS G12D Inhibitor Drug Market Scope
The market is segmented on the basis of drug type, therapy type, application, and end user.
- By Drug Type
On the basis of drug type, the KRAS G12D inhibitor drug market is segmented into small molecule inhibitors, biologics, and others. The small molecule inhibitors segment dominated the market with the largest market revenue share of 58.3% in 2025, driven by their ability to directly target intracellular KRAS signaling pathways and strong advancement in early-stage oncology pipelines. These inhibitors are preferred due to their oral bioavailability potential and relatively faster clinical development compared to complex biologic modalities. Pharmaceutical companies are heavily investing in small molecule platforms as they offer scalable manufacturing and better tumor penetration. The segment is also supported by strong clinical momentum of lead candidates such as MRTX1133-type molecules in preclinical and Phase I studies. Growing focus on mutation-specific targeting further strengthens dominance of this segment in precision oncology.
The biologics segment is anticipated to witness the fastest growth rate of 24.6% from 2026 to 2033, fueled by advancements in engineered antibody formats and protein-based targeting approaches for KRAS-driven cancers. Biologics offer high specificity and reduced off-target toxicity, making them attractive for combination cancer therapies. Increasing research in antibody-drug conjugates and engineered protein inhibitors is expanding their application scope. Strong biopharmaceutical R&D investments and academic collaborations are accelerating innovation in this segment. Additionally, biologics are gaining traction in combination regimens with immunotherapy to overcome tumor resistance mechanisms.
- By Therapy Type
On the basis of therapy type, the KRAS G12D inhibitor drug market is segmented into monotherapy and combination therapy. The monotherapy segment dominated the market with the largest revenue share of 61.4% in 2025, driven by its use in early clinical trials and initial regulatory evaluation of KRAS G12D-targeting drugs. Monotherapy approaches allow clearer assessment of drug efficacy and safety profiles in mutation-specific patient populations. This segment benefits from streamlined clinical trial design and faster regulatory pathways for breakthrough oncology drugs. Pharmaceutical developers initially prioritize monotherapy to establish proof-of-concept in KRAS-driven tumors. Its dominance is also supported by strong pipeline activity in first-generation KRAS G12D selective inhibitors.
The combination therapy segment is expected to witness the fastest growth rate of 26.1% from 2026 to 2033, driven by the need to overcome adaptive resistance mechanisms in KRAS-mutated cancers. Combination regimens with immunotherapy, SHP2 inhibitors, and chemotherapy are showing improved clinical outcomes in solid tumors. Increasing evidence of tumor pathway reactivation is encouraging multi-targeted treatment strategies. Pharmaceutical companies are actively designing trials that combine KRAS G12D inhibitors with checkpoint inhibitors to enhance immune response. This segment is further supported by strong clinical research focus on durable response improvement and survival extension.
- By Application
On the basis of application, the KRAS G12D inhibitor drug market is segmented into pancreatic cancer (PDAC), colorectal cancer (CRC), non-small cell lung cancer (NSCLC), and other solid tumors. The pancreatic cancer (PDAC) segment dominated the market with the largest revenue share of 39.8% in 2025, driven by the high prevalence of KRAS mutations in pancreatic tumors and extremely limited existing treatment options. PDAC remains one of the most aggressive cancers with poor survival rates, creating strong demand for targeted therapeutic innovation. KRAS G12D mutation is particularly common in pancreatic adenocarcinoma, making it a key focus area for drug developers. High unmet clinical need is accelerating clinical trial prioritization in this indication. Strong research funding and orphan drug incentives further support dominance of this segment.
The colorectal cancer (CRC) segment is expected to witness the fastest growth rate of 23.9% from 2026 to 2033, driven by rising incidence of KRAS-mutated colorectal tumors and increasing adoption of precision oncology diagnostics. Improved molecular screening is enabling better identification of KRAS G12D mutations in CRC patients. Pharmaceutical companies are expanding clinical trials targeting CRC due to its large patient pool and commercial potential. Combination therapy approaches are showing promising efficacy in refractory colorectal cancer cases. Growing awareness of genetic profiling in oncology is further accelerating adoption in this segment.
- By End User
On the basis of end user, the KRAS G12D inhibitor drug market is segmented into hospitals, oncology centers, cancer research institutes, academic institutions, and specialty clinics. The hospitals segment dominated the market with the largest revenue share of 46.7% in 2025, driven by their central role in cancer diagnosis, treatment administration, and clinical trial participation. Hospitals serve as primary sites for administering advanced targeted therapies under oncologist supervision. Availability of specialized oncology departments supports strong adoption of KRAS-targeted treatments. High patient inflow and integrated diagnostic capabilities further reinforce hospital dominance. Hospitals also act as key hubs for early access programs and innovative oncology drugs.
The oncology centers segment is expected to witness the fastest growth rate of 25.3% from 2026 to 2033, driven by increasing specialization in cancer care and expansion of dedicated oncology treatment networks. These centers are increasingly involved in advanced clinical trials for precision oncology drugs. Focused infrastructure for targeted therapy administration enhances treatment outcomes and patient monitoring. Rising demand for personalized cancer treatment is accelerating adoption in these facilities. Strong collaboration with pharmaceutical companies is further boosting growth of this segment.
KRAS G12D Inhibitor Drug Market Regional Analysis
- North America dominated the KRAS G12D inhibitor drug market with the largest revenue share of 42.6% in 2025, supported by advanced oncology infrastructure, high clinical trial density, strong funding from biotechnology firms, and early adoption of novel targeted therapies
- Pharmaceutical companies and biotechnology firms in the region are highly focused on KRAS-targeted innovation, supported by extensive clinical trial activity and rapid adoption of breakthrough oncology treatments in specialized cancer centers
- This dominance is further supported by strong funding from government and private investors, well-established regulatory pathways for orphan oncology drugs, and increasing use of biomarker-based patient selection, establishing KRAS G12D inhibitors as a key focus in precision medicine development
U.S. KRAS G12D Inhibitor Drug Market Insight
The U.S. KRAS G12D inhibitor drug market captured the largest revenue share of 81% in 2025 within North America, fueled by strong adoption of precision oncology and advanced cancer research infrastructure. Patients and healthcare providers are increasingly prioritizing targeted therapies for hard-to-treat cancers through mutation-specific treatment approaches. The growing focus on KRAS-driven tumor management, combined with strong clinical trial activity and rapid drug development pipelines, further propels the market. Moreover, the increasing integration of biomarker-based diagnostics and advanced oncology platforms in the U.S. is significantly contributing to the market’s expansion.
Europe KRAS G12D Inhibitor Drug Market Insight
The Europe KRAS G12D inhibitor drug market is projected to expand at a substantial CAGR throughout the forecast period, primarily driven by rising cancer burden and increasing demand for advanced precision oncology treatments. The increase in clinical research collaborations between pharmaceutical companies and academic institutes is fostering the development of KRAS-targeted therapies. European healthcare systems are also increasingly adopting molecular diagnostic testing to support targeted treatment decisions. Additionally, strong regulatory support for innovative oncology drugs is significantly contributing to market expansion.
U.K. KRAS G12D Inhibitor Drug Market Insight
The U.K. KRAS G12D inhibitor drug market is anticipated to grow at a noteworthy CAGR during the forecast period, driven by increasing focus on precision medicine and rising incidence of KRAS-mutated cancers. The growing participation of the U.K. in global oncology clinical trials is strengthening access to innovative drug candidates. Additionally, increasing awareness of targeted cancer therapies among clinicians is supporting adoption. The country’s strong research ecosystem and healthcare infrastructure are further stimulating market growth.
Germany KRAS G12D Inhibitor Drug Market Insight
The Germany KRAS G12D inhibitor drug market is expected to expand at a considerable CAGR during the forecast period, fueled by strong emphasis on innovation and advanced oncology treatment development. Germany’s well-established healthcare infrastructure supports rapid adoption of precision oncology drugs in hospital and research settings. Increasing integration of molecular diagnostics into routine cancer care is further driving demand. Moreover, strong investment in biotechnology research is promoting development of next-generation KRAS-targeted therapies.
Asia-Pacific KRAS G12D Inhibitor Drug Market Insight
The Asia-Pacific KRAS G12D inhibitor drug market is poised to grow at the fastest CAGR of 24.3% during the forecast period of 2026 to 2033, driven by rising cancer incidence and expanding access to advanced oncology treatments. Increasing participation in global clinical trials and growing investment in biotechnology research are strengthening market growth. Governments in the region are also supporting precision medicine initiatives and pharmaceutical innovation. Furthermore, improving healthcare infrastructure and rising awareness of targeted cancer therapies are accelerating market adoption.
Japan KRAS G12D Inhibitor Drug Market Insight
The Japan KRAS G12D inhibitor drug market is gaining momentum due to the country’s advanced healthcare system and strong focus on high-tech oncology solutions. The increasing number of cancer patients and growing demand for personalized medicine are supporting adoption of targeted therapies. Japan’s active participation in oncology research and clinical trials is further driving market growth. Additionally, integration of precision diagnostics into cancer care is improving treatment outcomes and supporting drug uptake.
India KRAS G12D Inhibitor Drug Market Insight
The India KRAS G12D inhibitor drug market accounted for the largest market revenue share in Asia Pacific in 2025, attributed to the rising cancer burden and rapid expansion of healthcare infrastructure. Increasing awareness of precision oncology and improving access to advanced diagnostic testing are driving demand for targeted therapies. India’s growing participation in global clinical trials is further supporting market development. Additionally, rising healthcare investment and government support for biotechnology innovation are key factors propelling the market.
KRAS G12D Inhibitor Drug Market Share
The KRAS G12D Inhibitor Drug industry is primarily led by well-established companies, including:
- Mirati Therapeutics Inc. (U.S.)
- Bristol-Myers Squibb Company (U.S.)
- Bayer AG (Germany)
- Kumquat Biosciences Inc. (U.S.)
- Revolution Medicines, Inc. (U.S.)
- Eli Lilly and Company (U.S.)
- Incyte Corporation (U.S.)
- Amgen Inc. (U.S.)
- Pfizer Inc. (U.S.)
- Novartis AG (Switzerland)
- AstraZeneca PLC (U.K.)
- Merck & Co., Inc. (U.S.)
- Sanofi (France)
- Takeda Pharmaceutical Company Limited (Japan)
- BeiGene Ltd. (China)
- BridgeBio Pharma, Inc. (U.S.)
- Verastem Oncology (U.S.)
- Black Diamond Therapeutics, Inc. (U.S.)
What are the Recent Developments in Global KRAS G12D Inhibitor Drug Market?
- In October 2025, oncology research reports highlighted MRTX1133 as the first direct KRAS G12D inhibitor entering clinical development, emphasizing its role as a breakthrough molecule targeting one of the most common mutations in pancreatic ductal adenocarcinoma and colorectal cancer
- In April 2025, Mirati Therapeutics (Bristol Myers Squibb) reported ongoing clinical evaluation updates for MRTX1133, the first selective KRAS G12D inhibitor, through its Phase 1/2 trial in advanced solid tumors including pancreatic and colorectal cancers. The study continued patient enrollment and dose optimization to evaluate safety, pharmacokinetics, and early anti-tumor activity in KRAS G12D-mutated cancers
- In August 2025, Bayer entered a global exclusive licensing agreement with Kumquat Biosciences to develop and commercialize a novel KRAS G12D inhibitor, marking a significant industry collaboration aimed at advancing early-stage oncology pipelines for KRAS-driven cancers
- In March 2025, the Phase 1/2 clinical trial of MRTX1133 was reported to have completed its primary Phase 1 stage, with the study later being terminated before Phase 2 due to pharmacokinetic variability and suboptimal exposure results, highlighting key development challenges in KRAS G12D targeting therapies
- In January 2023, the U.S. FDA cleared Mirati Therapeutics to initiate clinical trials for MRTX1133, enabling the first human testing of a selective KRAS G12D inhibitor for patients with colorectal, pancreatic, and non-small cell lung cancers
SKU-
Get online access to the report on the World's First Market Intelligence Cloud
- Interactive Data Analysis Dashboard
- Company Analysis Dashboard for high growth potential opportunities
- Research Analyst Access for customization & queries
- Competitor Analysis with Interactive dashboard
- Latest News, Updates & Trend analysis
- Harness the Power of Benchmark Analysis for Comprehensive Competitor Tracking
Research Methodology
Data collection and base year analysis are done using data collection modules with large sample sizes. The stage includes obtaining market information or related data through various sources and strategies. It includes examining and planning all the data acquired from the past in advance. It likewise envelops the examination of information inconsistencies seen across different information sources. The market data is analysed and estimated using market statistical and coherent models. Also, market share analysis and key trend analysis are the major success factors in the market report. To know more, please request an analyst call or drop down your inquiry.
The key research methodology used by DBMR research team is data triangulation which involves data mining, analysis of the impact of data variables on the market and primary (industry expert) validation. Data models include Vendor Positioning Grid, Market Time Line Analysis, Market Overview and Guide, Company Positioning Grid, Patent Analysis, Pricing Analysis, Company Market Share Analysis, Standards of Measurement, Global versus Regional and Vendor Share Analysis. To know more about the research methodology, drop in an inquiry to speak to our industry experts.
Customization Available
Data Bridge Market Research is a leader in advanced formative research. We take pride in servicing our existing and new customers with data and analysis that match and suits their goal. The report can be customized to include price trend analysis of target brands understanding the market for additional countries (ask for the list of countries), clinical trial results data, literature review, refurbished market and product base analysis. Market analysis of target competitors can be analyzed from technology-based analysis to market portfolio strategies. We can add as many competitors that you require data about in the format and data style you are looking for. Our team of analysts can also provide you data in crude raw excel files pivot tables (Fact book) or can assist you in creating presentations from the data sets available in the report.