Global Met Inhibitor Drug Market
Market Size in USD Billion
CAGR :
% 
 USD
1.89 Billion
USD
5.94 Billion
2025
2033
USD
1.89 Billion
USD
5.94 Billion
2025
2033
| 2026 - 2033 | |
| USD 1.89 Billion | |
| USD 5.94 Billion | |
|
|
|
|
MET Inhibitor Drug Market Size
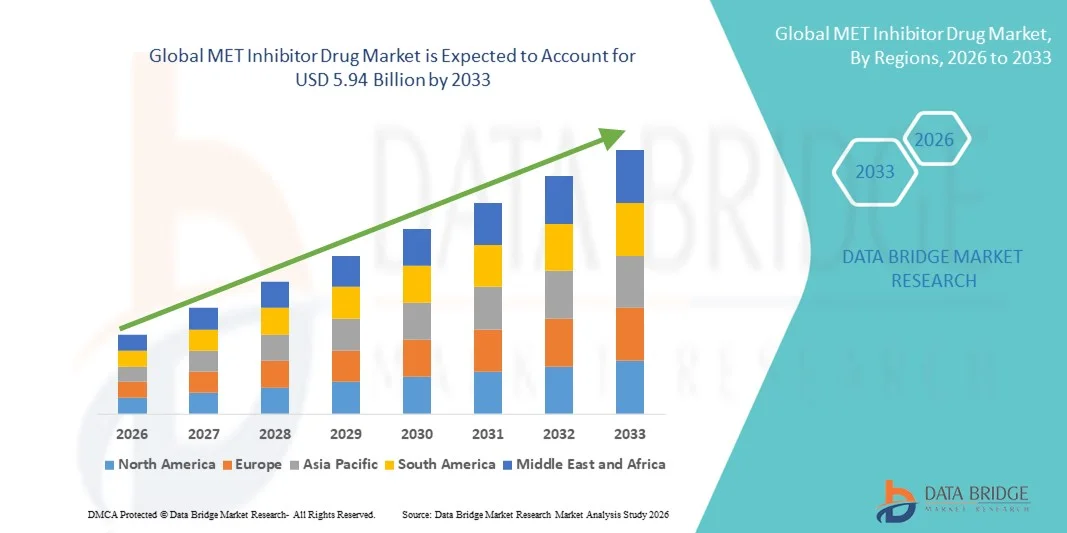
- The global MET Inhibitor Drug market size was valued at USD 1.89 billion in 2025 and is expected to reach USD 5.94 billion by 2033, at a CAGR of 15.40% during the forecast period
- The market growth is largely fueled by increasing prevalence of MET-driven cancers, rising adoption of targeted cancer therapies, and growing awareness among clinicians and patients about precision oncology treatments
- Furthermore, advancements in drug development, such as small molecule inhibitors and monoclonal antibodies targeting the MET pathway, alongside expanding clinical trial pipelines and favorable regulatory approvals, are accelerating the uptake of MET Inhibitor Drug solutions, thereby significantly boosting the industry's growth
MET Inhibitor Drug Market Analysis
- MET Inhibitor Drugs, offering targeted therapeutic options for MET-driven cancers, are increasingly vital components of modern oncology treatment protocols in both hospitals and specialty cancer centers due to their high efficacy, precision, and ability to complement existing therapies
- The escalating demand for MET Inhibitor Drugs is primarily fueled by the rising prevalence of MET-driven cancers, growing awareness among clinicians and patients, and increasing investment in oncology research and personalized medicine
- North America dominated the MET inhibitor drug market with the largest revenue share of 42.7% in 2025, characterized by advanced healthcare infrastructure, high adoption of targeted therapies, and a strong presence of key pharmaceutical players, with the U.S. experiencing substantial growth in MET Inhibitor Drug usage across oncology centers, hospitals, and research institutes, driven by innovations in small molecule inhibitors and monoclonal antibodies
- Asia-Pacific is expected to be the fastest-growing region in the MET inhibitor drug market during the forecast period, expanding at a CAGR of 11.2% from 2025 to 2032, due to increasing cancer incidence, rising healthcare spending, expanding access to targeted therapies, and supportive government initiatives promoting advanced oncology treatments in countries such as China, India, and Japan
- The small molecule inhibitors segment dominated the largest market revenue share of 44.5% in 2025, driven by their oral bioavailability, ease of administration, and established clinical efficacy
Report Scope and MET Inhibitor Drug Market Segmentation
|
Attributes |
MET Inhibitor Drug Key Market Insights |
|
Segments Covered |
|
|
Countries Covered |
North America
Europe
Asia-Pacific
Middle East and Africa
South America
|
|
Key Market Players |
|
|
Market Opportunities |
|
|
Value Added Data Infosets |
In addition to the insights on market scenarios such as market value, growth rate, segmentation, geographical coverage, and major players, the market reports curated by the Data Bridge Market Research also include in-depth expert analysis, patient epidemiology, pipeline analysis, pricing analysis, and regulatory framework. |
MET Inhibitor Drug Market Trends
Enhanced Market Developments in MET Inhibitor Drugs
- A significant and accelerating trend in the global MET Inhibitor Drug market is the increasing focus on precision oncology and biomarker-driven therapies, which aim to target cancer cells more effectively while minimizing adverse effects on healthy tissues
- This trend reflects a shift in oncology from broad-spectrum chemotherapy to highly personalized treatment regimens tailored to individual genetic profiles
- For instance, Capmatinib (Novartis) and Tepotinib (Merck) have been approved for NSCLC patients with MET exon 14 skipping mutations, highlighting the move toward genetically guided therapy and demonstrating how molecular profiling is influencing clinical decision-making
- Combination therapies involving MET inhibitors with other targeted drugs, such as EGFR inhibitors or immune checkpoint blockers, are gaining attention in clinical trials to overcome acquired resistance and improve long-term patient survival
- The increasing adoption of companion diagnostic tests and next-generation sequencing technologies is enabling earlier detection of MET alterations, allowing physicians to identify suitable candidates for MET-targeted therapy more effectively and streamline treatment protocols
MET Inhibitor Drug Market Dynamics
Driver
Growing Adoption Due to Rising Cancer Prevalence and Personalized Medicine
- The rising incidence of cancers linked to MET gene alterations—including non-small cell lung cancer (NSCLC), gastric cancer, and certain sarcomas—is fueling demand for MET inhibitor therapies
- These cancers often exhibit aggressive behavior and limited response to conventional treatments, making targeted therapy a critical option
- For instances, MET exon 14 skipping mutations are observed in approximately 3–4% of all NSCLC cases globally, prompting oncologists to increasingly prescribe targeted therapies like Tepotinib and Capmatinib. This demonstrates how specific genetic profiling is directly driving drug utilization in clinical settings
- Substantial R&D investment by leading pharmaceutical companies, coupled with ongoing clinical trials exploring novel MET inhibitors and combination therapies, is expanding the therapeutic pipeline and attracting healthcare stakeholders to the market
- Supportive regulatory frameworks, improved reimbursement policies in developed countries, and rising awareness among oncologists about the clinical benefits of precision medicine are further accelerating the adoption of MET inhibitor drugs
Restraint/Challenge
High Costs, Limited Accessibility, and Resistance Concerns
- The high cost of MET inhibitor therapies and limited access in emerging and low-income countries continue to constrain market growth. Even in developed regions, the affordability of long-term treatment remains a concern for patients without comprehensive insurance coverage
- For instance, the annual treatment cost of Capmatinib can reach tens of thousands of dollars, which can be prohibitive for patients in low- and middle-income countries, despite its proven efficacy in MET-driven NSCLC
- Drug resistance due to secondary mutations or tumor heterogeneity, as well as potential adverse effects, may limit patient adherence to therapy and reduce long-term treatment effectiveness
- Complex biomarker testing requirements, limited infrastructure for genetic profiling in certain regions, and insufficient physician awareness in emerging markets may also hinder broad adoption of MET inhibitors
- Addressing these challenges through expanded patient assistance programs, broader insurance coverage, investment in diagnostic infrastructure, and ongoing physician education will be essential to sustain growth in the global MET inhibitor drug market
MET Inhibitor Drug Market Scope
The market is segmented on the basis of drug type and application.
- By Drug Type
On the basis of drug type, the MET Inhibitor Drug market is segmented into small molecule inhibitors, monoclonal antibodies, and combination therapies. The small molecule inhibitors segment dominated the largest market revenue share of 44.5% in 2025, driven by their oral bioavailability, ease of administration, and established clinical efficacy. These inhibitors effectively target MET receptor tyrosine kinases in various cancers, including NSCLC and gastric cancer. Favorable pharmacokinetics and safety profiles support broad adoption across oncology centers. The growing prevalence of MET-driven tumors enhances demand. Continuous clinical research and successful phase II/III trials strengthen confidence in small molecule therapies. Strong physician preference for oral targeted therapies further reinforces market dominance. Robust regulatory approvals in developed regions accelerate market penetration. Adoption in combination regimens with chemotherapy and immunotherapy expands therapeutic applications. Increasing patient awareness regarding targeted therapies contributes to uptake. Favorable reimbursement frameworks in key markets support adoption. Collaborative partnerships between pharmaceutical companies and research institutes drive innovation. Expansion in emerging markets broadens patient access. High efficacy in metastatic and resistant cancer cases strengthens competitive positioning.
The monoclonal antibodies segment is expected to witness the fastest CAGR of 13.8% from 2026 to 2033, fueled by growing interest in targeted biologics with reduced systemic toxicity. Monoclonal antibodies against MET show promising efficacy in both hematologic and solid tumors. Rising investment in biologic R&D accelerates pipeline development. Increasing approvals in NSCLC, HCC, and gastric cancer further stimulate growth. Combination strategies with immune checkpoint inhibitors enhance therapeutic outcomes. Adoption in specialty oncology centers is rising due to precise targeting and manageable side effect profiles. Patient preference for antibody therapies with fewer off-target effects boosts demand. Expansion of clinical trials globally supports faster market adoption. Technological advances in antibody engineering improve specificity and half-life. Government initiatives to support innovative cancer therapies strengthen market access. Strategic collaborations between biotech firms and hospitals enhance distribution and clinical adoption. Rising awareness of personalized medicine drives higher uptake rates. Continuous monitoring of biomarker expression aids in patient selection and therapy optimization.
- By Application
On the basis of application, the MET Inhibitor Drug market is segmented into non-small cell lung cancer (NSCLC), gastric cancer, hepatocellular carcinoma (HCC), and other solid tumors. The NSCLC segment accounted for the largest market revenue share of 38.9% in 2025, driven by the high global incidence of lung cancer and identification of MET exon 14 skipping mutations in a significant patient subset. Targeted MET therapies improve progression-free survival and overall outcomes, encouraging physician adoption. Comprehensive screening programs and molecular diagnostics support early identification of eligible patients. Rising research on combination therapies and resistance mechanisms enhances clinical utility. Strong reimbursement policies in key regions promote access. Increasing availability of MET-targeted drugs in hospital oncology units expands reach. Continuous awareness campaigns on precision oncology increase patient demand. Favorable regulatory approvals for first- and second-line treatments further consolidate segment dominance. Multinational collaborations enable access to novel therapies in emerging markets. Expanded clinical guidelines recommend MET testing, supporting adoption. Growth in metastatic and advanced-stage patient populations drives demand.
The gastric cancer segment is expected to witness the fastest CAGR of 14.6% from 2026 to 2033, fueled by increasing prevalence of MET-amplified gastric tumors in APAC and Europe. Rising use of molecular profiling to guide therapy selection accelerates adoption. Combination regimens with chemotherapy and immunotherapy improve treatment outcomes. Expanding oncology infrastructure and diagnostic capabilities in emerging markets supports segment growth. Growing physician awareness regarding MET-targeted interventions drives clinical adoption. Patient advocacy and education programs enhance therapy uptake. Increasing funding for gastric cancer research accelerates pipeline development. Favorable government initiatives for precision oncology strengthen access. Rising adoption in specialized cancer centers ensures higher patient reach. Development of next-generation MET inhibitors improves efficacy and tolerability. Strategic collaborations among biotech and pharma companies enhance drug distribution and clinical trial availability. Continuous innovations in companion diagnostics facilitate patient stratification and personalized therapy.
MET Inhibitor Drug Market Regional Analysis
- North America dominated the MET inhibitor drug market with the largest revenue share of 42.7% in 2025, driven by advanced healthcare infrastructure, high adoption of targeted therapies, and a strong presence of key pharmaceutical players. The region has witnessed substantial growth in MET inhibitor usage across oncology centers, hospitals, and research institutes, reflecting innovations in small molecule inhibitors and monoclonal antibodies
- For instance, the U.S. has seen widespread adoption of Capmatinib and Tepotinib for NSCLC patients with MET exon 14 skipping mutations, while Canada has initiated early-access programs to facilitate treatment of rare MET-driven cancers
- The North American market benefits from robust clinical trial networks, favorable reimbursement policies, and a strong culture of precision oncology, supporting rapid introduction and adoption of new MET-targeted therapies
U.S. MET Inhibitor Drug Market Insight
The U.S. MET inhibitor drug market captured the largest revenue share within North America in 2025, accounting for 39.8% of the global market. Growth is driven by advanced oncology infrastructure, high adoption of targeted therapies, and strong presence of leading pharmaceutical manufacturers. MET inhibitor usage has increased significantly in hospitals, cancer research centers, and specialized oncology clinics, reflecting continuous innovation in small molecule inhibitors and monoclonal antibodies. High awareness among oncologists, favorable insurance coverage, and strong patient advocacy programs further bolster adoption of MET inhibitors in the U.S. oncology landscape.
Europe MET Inhibitor Drug Market Insight
The Europe MET inhibitor drug market is projected to expand at a substantial CAGR throughout the forecast period, driven by well-established healthcare systems, increasing cancer prevalence, and expanding access to targeted therapies. Government support for early cancer detection, investments in research facilities, and growing clinical trial participation are key factors boosting market adoption in Europe.
U.K. MET Inhibitor Drug Market Insight
The U.K. MET inhibitor drug market is anticipated to grow at a noteworthy CAGR, fueled by advanced healthcare infrastructure, rising adoption of precision oncology, and increasing awareness of targeted cancer therapies. Ongoing government initiatives to support access to innovative therapies and early intervention strategies are expected to sustain market growth.
Germany MET Inhibitor Drug Market Insight
The Germany MET inhibitor drug market is expected to expand at a considerable CAGR during the forecast period, supported by robust research networks, high awareness of targeted cancer therapies, and a strong pharmaceutical sector. Integration of precision oncology into clinical practice, coupled with favorable reimbursement schemes, promotes sustained market adoption.
Asia-Pacific MET Inhibitor Drug Market Insight
The Asia-Pacific MET Inhibitor Drug market is expected to be the fastest-growing region during the forecast period, expanding at a CAGR of 11.2% from 2025 to 2032. Growth is driven by rising cancer incidence, increasing healthcare spending, expanding access to targeted therapies, and government initiatives promoting advanced oncology treatments in countries such as China, India, and Japan. For instances, China has scaled up oncology centers offering MET inhibitor therapies, while India has introduced government-backed subsidy programs to expand access for MET-driven cancers. Rising awareness of personalized medicine, hospital modernization, and growing adoption of advanced oncology drugs are further accelerating market expansion across APAC.
Japan MET Inhibitor Drug Market Insight
The Japan MET inhibitor drug market is gaining momentum due to highly advanced healthcare systems, strong adoption of targeted therapies, and ongoing clinical research in oncology. For instance, Japanese cancer centers have integrated MET inhibitors into standard care for NSCLC patients, supported by robust post-marketing surveillance and real-world studies. Government initiatives promoting access to innovative oncology therapies and a focus on precision medicine are expected to sustain growth in both hospital and research sectors.
China MET Inhibitor Drug Market Insight
The China MET inhibitor drug market accounted for the largest market revenue share in Asia-Pacific in 2025, driven by rapid urbanization, expanding middle-class population, high technological adoption, and government support for advanced oncology treatments. Integration of MET-targeted therapy into hospital protocols, expansion of oncology infrastructure, and smart city initiatives continue to strengthen market growth in China.
MET Inhibitor Drug Market Share
The MET Inhibitor Drug industry is primarily led by well-established companies, including:
- Novartis (Switzerland)
- AbbVie (U.S.)
- Pfizer (U.S.)
- AstraZeneca (U.K.)
- Merck & Co. (U.S.)
- Johnson & Johnson (U.S.)
- Roche (Switzerland)
- Amgen (U.S.)
- Boehringer Ingelheim (Germany)
- Takeda (Japan)
- BeiGene (China)
- Hutchison China MediTech (China)
- Celgene (U.S.)
- Seattle Genetics (U.S.)
- MacroGenics (U.S.)
- Blueprint Medicines (U.S.)
- Exelixis (U.S.)
- Tesaro (U.S.)
- Genentech (U.S.)
- Incyte (U.S.)
Latest Developments in Global MET Inhibitor Drug Market
- In February 2021, the U.S. Food and Drug Administration (FDA) granted **accelerated approval to **Tepmetko (tepotinib), the first once‑daily oral MET inhibitor specifically indicated for adults with metastatic non‑small cell lung cancer (NSCLC) whose tumors harbor MET exon 14 skipping alterations. This approval marked a major regulatory milestone for MET‑targeted precision therapy and expanded treatment options for patients with this genetic alteration
- In March 2023, Savolitinib (ORPATHYS), a highly selective oral MET inhibitor marketed in China, was included in the National Reimbursement Drug List (NRDL) — improving patient access to MET‑targeted therapy for METex14 skipping NSCLC and reflecting broader reimbursement support in one of the largest oncology markets globally
- In June 2025, Savolitinib’s indication in China was expanded through regulatory approval to include treatment of adults with EGFRm MET‑amplified NSCLC (second‑line) in combination with Tagrisso(osimertinib). This approval highlights the growing importance of combination strategies targeting both MET amplification and EGFR pathways to address resistance mechanisms in advanced lung cancer
- In May 2025, the U.S. FDA granted accelerated approval to Emrelis™ (telisotuzumab vedotin) — the first c‑MET targeted antibody‑drug conjugate (ADC) — for adults with locally advanced or metastatic non‑squamous NSCLC with high c‑MET protein overexpression who have previously received systemic therapy. This approval reflects an important expansion of MET‑directed therapies beyond small‑molecule kinase inhibitors into antibody‑based targeted oncology
SKU-
Get online access to the report on the World's First Market Intelligence Cloud
- Interactive Data Analysis Dashboard
- Company Analysis Dashboard for high growth potential opportunities
- Research Analyst Access for customization & queries
- Competitor Analysis with Interactive dashboard
- Latest News, Updates & Trend analysis
- Harness the Power of Benchmark Analysis for Comprehensive Competitor Tracking
Research Methodology
Data collection and base year analysis are done using data collection modules with large sample sizes. The stage includes obtaining market information or related data through various sources and strategies. It includes examining and planning all the data acquired from the past in advance. It likewise envelops the examination of information inconsistencies seen across different information sources. The market data is analysed and estimated using market statistical and coherent models. Also, market share analysis and key trend analysis are the major success factors in the market report. To know more, please request an analyst call or drop down your inquiry.
The key research methodology used by DBMR research team is data triangulation which involves data mining, analysis of the impact of data variables on the market and primary (industry expert) validation. Data models include Vendor Positioning Grid, Market Time Line Analysis, Market Overview and Guide, Company Positioning Grid, Patent Analysis, Pricing Analysis, Company Market Share Analysis, Standards of Measurement, Global versus Regional and Vendor Share Analysis. To know more about the research methodology, drop in an inquiry to speak to our industry experts.
Customization Available
Data Bridge Market Research is a leader in advanced formative research. We take pride in servicing our existing and new customers with data and analysis that match and suits their goal. The report can be customized to include price trend analysis of target brands understanding the market for additional countries (ask for the list of countries), clinical trial results data, literature review, refurbished market and product base analysis. Market analysis of target competitors can be analyzed from technology-based analysis to market portfolio strategies. We can add as many competitors that you require data about in the format and data style you are looking for. Our team of analysts can also provide you data in crude raw excel files pivot tables (Fact book) or can assist you in creating presentations from the data sets available in the report.