Global Mitochondrial Disease Targeted Therapies Market
Market Size in USD Billion
CAGR :
% 
 USD
2.04 Billion
USD
9.31 Billion
2025
2033
USD
2.04 Billion
USD
9.31 Billion
2025
2033
| 2026 - 2033 | |
| USD 2.04 Billion | |
| USD 9.31 Billion | |
|
|
|
|
Mitochondrial Disease Targeted Therapies Market Size
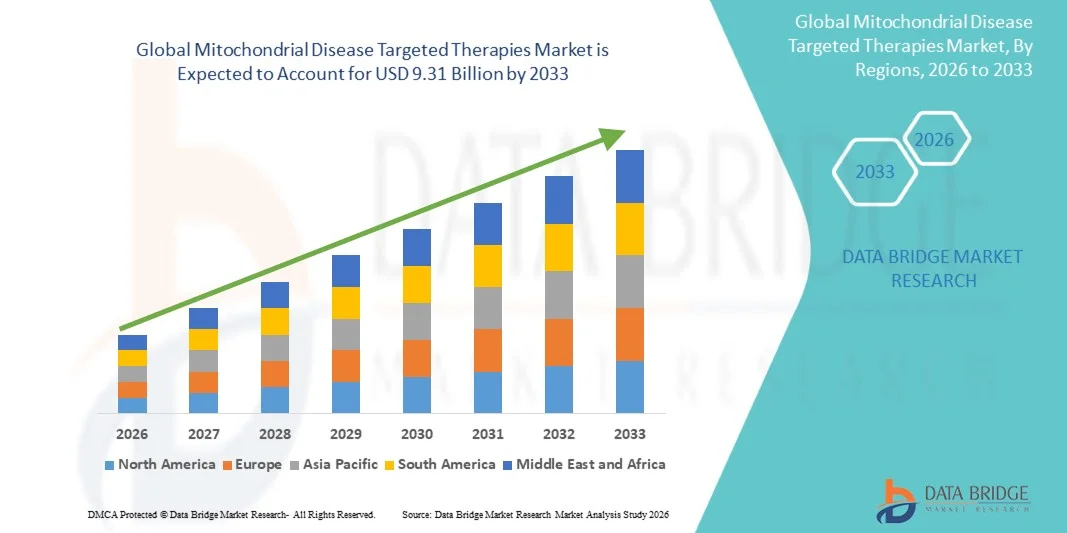
- The global mitochondrial disease targeted therapies market size was valued at USD 2.04 billion in 2025 and is expected to reach USD 9.31 billion by 2033, at a CAGR of 20.90% during the forecast period
- The market growth is largely fueled by the increasing prevalence of mitochondrial disorders, rising awareness among healthcare providers and patients, and advancements in targeted therapeutic solutions that address the underlying causes of these conditions
- Furthermore, growing investments in research and development, coupled with the emergence of innovative treatment modalities such as gene therapy, enzyme replacement therapy, and small molecule therapeutics, are accelerating the uptake of Mitochondrial Disease Targeted Therapies solutions, thereby significantly boosting the industry's growth
Mitochondrial Disease Targeted Therapies Market Analysis
- Mitochondrial Disease Targeted Therapies, including gene therapies, enzyme replacement therapies, and small molecule treatments, are increasingly vital in addressing the root causes of mitochondrial disorders and improving patient outcomes across various clinical settings
- The escalating demand for these therapies is primarily fueled by the rising prevalence of mitochondrial diseases, growing awareness among healthcare providers and patients, and advancements in precision medicine enabling personalized treatment approaches
- The North America dominated the mitochondrial disease targeted therapies market with the largest revenue share of 38.7% in 2025, driven by high adoption of advanced therapeutic solutions, strong R&D capabilities, and a robust presence of key industry players, with substantial growth in specialized treatment centers and clinical programs for mitochondrial disorders
- Asia Pacific is expected to be the fastest growing region in the mitochondrial disease targeted therapies market during the forecast period, supported by increasing healthcare investments, expansion of specialized treatment facilities, and rising awareness and diagnosis rates of mitochondrial disorders among patients and clinicians
- Gene Therapy dominated the market with a revenue share of 45.3% in 2025, driven by its potential to address underlying genetic defects and provide long-term therapeutic benefits
Report Scope and Mitochondrial Disease Targeted Therapies Market Segmentation
|
Attributes |
Mitochondrial Disease Targeted Therapies Key Market Insights |
|
Segments Covered |
|
|
Countries Covered |
North America
Europe
Asia-Pacific
Middle East and Africa
South America
|
|
Key Market Players |
• Stealth BioTherapeutics (U.S.) |
|
Market Opportunities |
|
|
Value Added Data Infosets |
In addition to the insights on market scenarios such as market value, growth rate, segmentation, geographical coverage, and major players, the market reports curated by the Data Bridge Market Research also include in-depth expert analysis, patient epidemiology, pipeline analysis, pricing analysis, and regulatory framework. |
Mitochondrial Disease Targeted Therapies Market Trends
“Advancements and Growing Adoption of Targeted Therapies”
- A significant and accelerating trend in the global Mitochondrial Disease Targeted Therapies market is the increasing focus on precision medicine and targeted therapeutic interventions
- This approach emphasizes tailoring treatments based on specific mitochondrial dysfunctions, genetic variations, and patient profiles, thereby enhancing treatment efficacy and reducing adverse effects
- For instance, the use of novel small-molecule therapies, gene therapies, and mitochondrial co-factor supplementation has gained traction in specialized clinics and research institutions worldwide, improving patient outcomes in conditions such as Leigh syndrome and mitochondrial myopathy
- The growing trend also includes the development of therapies that target specific cellular pathways, such as oxidative phosphorylation modulators and reactive oxygen species (ROS) regulators, enabling a more personalized approach to mitochondrial dysfunction management
- Clinical research and biotechnology innovations continue to propel the expansion of therapy options, with an increasing number of late-stage trials demonstrating safety and efficacy
- This trend reflects a broader shift toward individualized care, highlighting the importance of integrating molecular diagnostics, biomarker analysis, and patient monitoring systems in therapeutic strategies
Mitochondrial Disease Targeted Therapies Market Dynamics
Driver
“Rising Prevalence of Mitochondrial Disorders and Growing Healthcare Awareness”
- The increasing prevalence of mitochondrial disorders globally, coupled with growing awareness among clinicians and patients about the benefits of targeted interventions, is a key driver of market growth
- Early diagnosis and intervention are critical in managing progressive mitochondrial conditions, creating strong demand for advanced therapies
- For instance, in 2025, several biotech firms launched novel therapies targeting mitochondrial respiratory chain deficiencies, with promising results in pediatric and adult populations. Such initiatives are expected to expand market penetration and drive growth in the forecast period
- Healthcare providers and research institutions are actively promoting early testing and intervention programs, further boosting the adoption of targeted therapies
- Rising government and private funding for rare disease research, coupled with partnerships between pharmaceutical companies and academic centers, is enhancing the development pipeline for mitochondrial therapies
- The increasing patient preference for minimally invasive, efficacious, and personalized treatments further supports the growth of the Mitochondrial Disease Targeted Therapies market
Restraint/Challenge
“High Treatment Costs and Limited Accessibility”
- The relatively high cost of targeted mitochondrial therapies poses a significant challenge to market expansion, especially in developing regions where healthcare infrastructure and reimbursement systems may be limited
- For instance, novel gene therapies and enzyme replacement treatments can reach substantial price points, limiting accessibility for patients without comprehensive insurance coverage or government support
- In addition, the complexity of diagnosing mitochondrial disorders and the need for specialized clinical expertise can hinder early adoption of these therapies, affecting overall market growth
- Addressing these challenges requires the development of more cost-effective therapies, expanded reimbursement frameworks, and increased patient and clinician awareness about treatment options
- While ongoing research is gradually introducing more affordable small-molecule therapies and combination approaches, perceived treatment costs and limited availability in certain regions continue to restrict broader adoption
- Strategies such as expanding clinical trial networks, public-private partnerships, and global awareness campaigns will be vital to overcoming these barriers and sustaining long-term market growth
Mitochondrial Disease Targeted Therapies Market Scope
The market is segmented on the basis of therapy type, disease indication, and end user.
• By Therapy Type
On the basis of therapy type, the Mitochondrial Disease Targeted Therapies market is segmented into Gene Therapy, Small Molecule Therapy, Enzyme Replacement Therapy, and Nutritional Supplementation. Gene Therapy dominated the market with a revenue share of 45.3% in 2025, driven by its potential to address underlying genetic defects and provide long-term therapeutic benefits. Hospitals and specialized clinics are increasingly investing in advanced gene-editing platforms. Clinical trials demonstrating safety and efficacy encourage adoption. Regulatory approvals in key markets facilitate patient access. Personalized medicine approaches and precision diagnostics support the segment. Gene therapy adoption is higher in developed regions due to established healthcare infrastructure. Government funding and grants promote research and accessibility. Integration with hospital-based genomics programs improves outcomes. Awareness campaigns and patient advocacy accelerate patient enrollment. Advanced delivery vectors enhance therapeutic efficiency. Ongoing collaborations between biotech firms and hospitals support commercialization.
Small Molecule Therapy is expected to witness the fastest CAGR of 12.1% from 2026 to 2033, fueled by increasing research on mitochondrial-targeted drugs, growing patient pool, and wider availability in both hospital and home care settings. Its oral administration route and ease of integration into existing treatment regimens drive adoption. Emerging markets are expanding access to small molecule therapies. Research on combination therapies enhances efficacy. Cost-effectiveness compared to gene therapy makes it attractive for broader populations. Physician awareness and clinical guidelines promote usage. Pharmaceutical companies are launching novel mitochondrial modulators. Growing prevalence of Leigh syndrome, MELAS, and MERRF supports segment growth. Early diagnosis improves treatment uptake.
• By Disease Indication
On the basis of disease indication, the market is segmented into Leigh Syndrome, MELAS, MERRF, and Other Mitochondrial Disorders. Leigh Syndrome dominated the market with a revenue share of 36.7% in 2025, driven by high prevalence in pediatric populations and severe clinical outcomes. Specialized clinics prioritize early intervention using gene therapy and small molecule options. Hospitals and research laboratories adopt standardized treatment protocols. Awareness campaigns and newborn screening programs improve early diagnosis. Clinical research focusing on metabolic stabilization supports segment growth. Patient registries and advocacy groups enhance treatment access. Multidisciplinary care models ensure better outcomes. Government initiatives in rare disease management facilitate therapy adoption. Integration of advanced diagnostics supports precise treatment planning. Collaboration between biotech and healthcare providers accelerates therapy availability.
MELAS is expected to witness the fastest CAGR of 11.5% from 2026 to 2033, fueled by increasing recognition of stroke-like episodes and lactic acidosis in patients and the growing adoption of enzyme replacement and nutritional supplementation therapies. Emerging diagnostic tools enable early detection. Physician awareness programs support uptake. Hospitals and clinics are expanding specialized mitochondrial care programs. Clinical trials and research publications reinforce treatment efficacy. Improved patient monitoring systems support therapy adherence. Reimbursement policies in developed regions enhance affordability. Patient education on lifestyle and supplementation encourages compliance. Expansion of home care settings increases therapy accessibility. Innovative therapeutic pipelines drive market momentum.
• By End User
On the basis of end user, the market is segmented into Hospitals, Specialized Clinics, Research Laboratories, and Home Care Settings. Hospitals dominated the market with a revenue share of 48.5% in 2025, driven by high patient volume, advanced infrastructure, and multi-disciplinary care for mitochondrial disorders. Hospitals provide access to gene therapy, enzyme replacement, and nutritional supplementation under clinical supervision. Established patient referral networks and integration with pediatric and metabolic units support dominance. Hospitals maintain high-quality treatment protocols and monitoring. Government funding and grants facilitate access to advanced therapies. Clinical trials conducted at hospitals boost adoption. Experienced staff and specialized departments ensure effective therapy administration. Repeat visits and chronic disease management sustain revenue flow. Strategic collaborations with biotech firms enhance treatment access.
Home Care Settings are expected to witness the fastest CAGR of 10.8% from 2026 to 2033, driven by increasing demand for patient-centric care, chronic management, and convenience. Portable monitoring tools, telehealth integration, and at-home nutritional supplementation support therapy adherence. Patient preference for home-based care reduces hospital visits and associated costs. Adoption of small molecule therapies and nutritional interventions is higher in home care. Training programs for caregivers facilitate safe therapy administration. Awareness campaigns on rare mitochondrial disorders boost adoption. Expansion of home healthcare infrastructure in urban and semi-urban regions supports growth. Reimbursement and insurance coverage for home care services enhance affordability.
Mitochondrial Disease Targeted Therapies Market Regional Analysis
- North America dominated the mitochondrial disease targeted therapies market with the largest revenue share of 38.7% in 2025, driven by high adoption of advanced therapeutic solutions, strong R&D capabilities, and a robust presence of key industry players
- The region has witnessed substantial growth in specialized treatment centers, clinical programs, and precision medicine initiatives targeting mitochondrial disorders, contributing significantly to market expansion
- Consumers and healthcare providers in the region increasingly value innovative therapies, early diagnostic interventions, and comprehensive care programs, which support improved patient outcomes. The well-established healthcare infrastructure and high healthcare expenditure further reinforce North America’s leading position in the market
U.S. Mitochondrial Disease Targeted Therapies Market Insight
The U.S. mitochondrial disease targeted therapies market accounted for the largest share within North America in 2025, fueled by the rapid adoption of advanced therapeutic interventions and specialized clinical programs. Increased awareness among physicians and patients about mitochondrial disorders, coupled with supportive government policies for rare disease treatments, has accelerated the adoption of targeted therapies. Furthermore, the presence of leading biotech and pharmaceutical companies focusing on mitochondrial medicine enhances the country’s market dominance.
Europe Mitochondrial Disease Targeted Therapies Market Insight
The Europe mitochondrial disease targeted therapies market is projected to expand at a substantial CAGR during the forecast period, driven by growing awareness of mitochondrial disorders, well-established healthcare systems, and government initiatives promoting access to specialized therapies. The region is witnessing increased investment in clinical trials and research for mitochondrial-targeted interventions, while rising urbanization and improved healthcare access further stimulate market growth.
U.K. Mitochondrial Disease Targeted Therapies Market Insight
The U.K. mitochondrial disease targeted therapies market is anticipated to grow steadily due to the rising incidence of mitochondrial disorders and growing demand for advanced therapeutic care. Increased awareness campaigns, integration of specialized treatment programs in hospitals, and the expanding availability of diagnostic facilities are driving market adoption. Additionally, policy support for rare disease management enhances patient access to innovative therapies.
Germany Mitochondrial Disease Targeted Therapies Market Insight
Germany’s mitochondrial disease targeted therapies market is expected to expand at a notable CAGR, fueled by strong R&D investments, advanced healthcare infrastructure, and an emphasis on innovative treatment approaches. Rising awareness among clinicians and patients regarding mitochondrial disorders, combined with adoption of targeted therapies in hospitals and specialty clinics, supports the market growth. The focus on sustainability and healthcare quality ensures the continuous development of patient-centric treatment programs.
Asia-Pacific Mitochondrial Disease Targeted Therapies Market Insight
The Asia-Pacific mitochondrial disease targeted therapies market is poised to grow at the fastest CAGR during the forecast period, supported by increasing healthcare investments, expansion of specialized treatment facilities, and rising awareness and diagnosis rates of mitochondrial disorders among patients and clinicians. Rapid urbanization, growing disposable incomes, and improving healthcare infrastructure in countries such as China, Japan, and India further drive market adoption.
Japan Mitochondrial Disease Targeted Therapies Market Insight
The Japan mitochondrial disease targeted therapies market is gaining momentum due to high healthcare standards, increasing focus on rare and genetic disorders, and growing patient awareness. The adoption of targeted therapies is supported by government initiatives, robust clinical research networks, and a strong emphasis on personalized treatment approaches, particularly for aging populations susceptible to mitochondrial dysfunctions.
China Mitochondrial Disease Targeted Therapies Market Insight
China accounted for the largest share of the Asia-Pacific market in 2025, attributed to the country’s expanding middle class, rising healthcare spending, and rapid urbanization. Increased investments in diagnostic centers, growing awareness of mitochondrial disorders, and the development of specialized treatment facilities are driving adoption. Additionally, supportive government programs for rare diseases and strong local biotech manufacturing capabilities further bolster the market.
Mitochondrial Disease Targeted Therapies Market Share
The Mitochondrial Disease Targeted Therapies industry is primarily led by well-established companies, including:
• Stealth BioTherapeutics (U.S.)
• Santhera Pharmaceuticals (Switzerland)
• Roche (Switzerland)
• GenSight Biologics (France)
• Amicus Therapeutics (U.S.)
• Sarepta Therapeutics (U.S.)
• Biogen (U.S.)
• Ultragenyx Pharmaceutical (U.S.)
• Mitobridge (U.S.)
• Catalyst Pharmaceuticals (U.S.)
• Wave Life Sciences (U.S.)
• Reata Pharmaceuticals (U.S.)
• Pfizer (U.S.)
• Avrobio (U.S.)
• Orchard Therapeutics (U.K.)
• Astellas Pharma (Japan)
• Mitsubishi Tanabe Pharma (Japan)
• Sanofi (France)
• Bayer (Germany)
• Biocartis (Belgium)
Latest Developments in Global Mitochondrial Disease Targeted Therapies Market
- In September 2025, the U.S. Food and Drug Administration (FDA) granted accelerated approval to FORZINITY (elamipretide HCl) — developed by Stealth BioTherapeutics — as the first-ever targeted therapy for Barth syndrome, a rare mitochondrial disorder characterized by impaired energy production. This approval marks a historic milestone as the first mitochondria‑targeted therapy to reach the market, designed to restore mitochondrial structure and function by binding to cardiolipin within mitochondria
- In February 2025, Reneo Pharmaceuticals announced that its investigational therapy REN001 received Orphan Drug Designation from the U.S. FDA for the treatment of primary mitochondrial myopathies (PMM). REN001 is a selective PPARδ agonist aimed at improving energy metabolism in skeletal muscle, addressing core mitochondrial dysfunction symptoms such as exercise intolerance and fatigue
- In May 2025, Thiogenesis Therapeutics initiated the first patients in a Phase II clinical trial of TTI‑0102 for MELAS (Mitochondrial Encephalomyopathy with Lactic Acidosis and Stroke‑like episodes). The randomized, double‑blind, placebo‑controlled study is an important step toward developing new therapies for this severe mitochondrial disease, focusing on safety, efficacy, and biomarker endpoints
- In July 2025, the U.S. FDA granted Breakthrough Therapy Designation to LX2006 from Lexeo Therapeutics for the treatment of Friedreich’s ataxia, a mitochondrial‑linked neurodegenerative disease, based on interim Phase I/II data showing clinically meaningful improvements in relevant functional biomarkers
- In March 2025, the U.S. FDA accepted a New Drug Application (NDA) from UCB for a treatment targeting Thymidine Kinase 2 deficiency (TK2d) — a mitochondrial DNA depletion syndrome — under Priority Review, advancing potential targeted therapy options in mitochondrial disorders
- In June 2025, the U.S. FDA granted Fast Track designation to zagociguat (from Tisento Therapeutics) for the treatment of MELAS syndrome, recognizing the urgent need for effective therapies and facilitating accelerated development and review
SKU-
Get online access to the report on the World's First Market Intelligence Cloud
- Interactive Data Analysis Dashboard
- Company Analysis Dashboard for high growth potential opportunities
- Research Analyst Access for customization & queries
- Competitor Analysis with Interactive dashboard
- Latest News, Updates & Trend analysis
- Harness the Power of Benchmark Analysis for Comprehensive Competitor Tracking
Research Methodology
Data collection and base year analysis are done using data collection modules with large sample sizes. The stage includes obtaining market information or related data through various sources and strategies. It includes examining and planning all the data acquired from the past in advance. It likewise envelops the examination of information inconsistencies seen across different information sources. The market data is analysed and estimated using market statistical and coherent models. Also, market share analysis and key trend analysis are the major success factors in the market report. To know more, please request an analyst call or drop down your inquiry.
The key research methodology used by DBMR research team is data triangulation which involves data mining, analysis of the impact of data variables on the market and primary (industry expert) validation. Data models include Vendor Positioning Grid, Market Time Line Analysis, Market Overview and Guide, Company Positioning Grid, Patent Analysis, Pricing Analysis, Company Market Share Analysis, Standards of Measurement, Global versus Regional and Vendor Share Analysis. To know more about the research methodology, drop in an inquiry to speak to our industry experts.
Customization Available
Data Bridge Market Research is a leader in advanced formative research. We take pride in servicing our existing and new customers with data and analysis that match and suits their goal. The report can be customized to include price trend analysis of target brands understanding the market for additional countries (ask for the list of countries), clinical trial results data, literature review, refurbished market and product base analysis. Market analysis of target competitors can be analyzed from technology-based analysis to market portfolio strategies. We can add as many competitors that you require data about in the format and data style you are looking for. Our team of analysts can also provide you data in crude raw excel files pivot tables (Fact book) or can assist you in creating presentations from the data sets available in the report.