Global Nanotechnology Based Medical Device Market
Market Size in USD Billion
CAGR :
% 
 USD
4.12 Billion
USD
10.31 Billion
2025
2033
USD
4.12 Billion
USD
10.31 Billion
2025
2033
| 2026 –2033 | |
| USD 4.12 Billion | |
| USD 10.31 Billion | |
|
|
|
|
Nanotechnology Based Medical Device Market Size
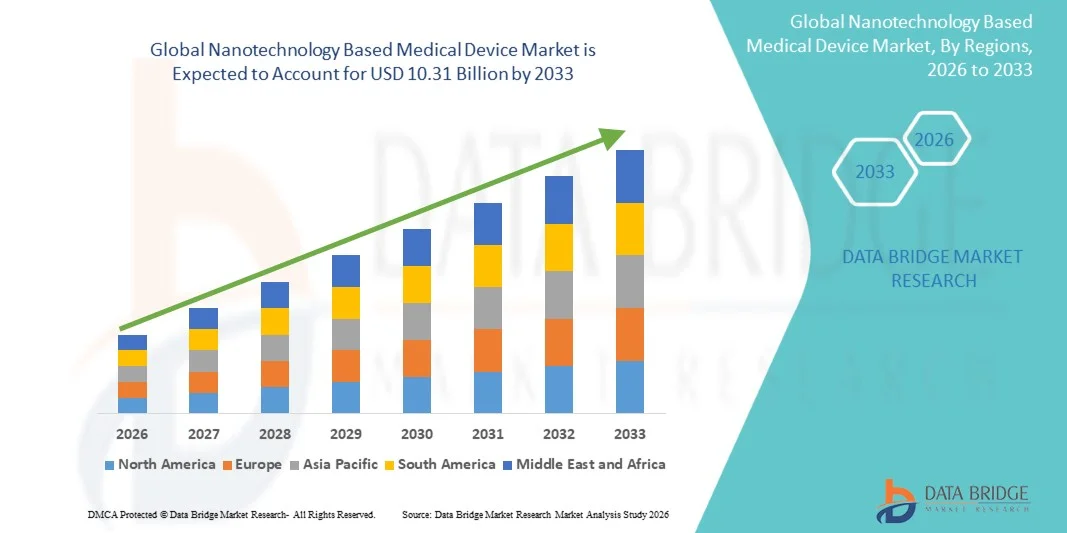
- The global nanotechnology based medical device market size was valued at USD 4.12 billion in 2025 and is expected to reach USD 10.31 billion by 2033, at a CAGR of 12.15% during the forecast period
- The market growth is largely fueled by the increasing integration of nanotechnology in medical devices and continuous advancements in precision medicine, leading to enhanced diagnostic accuracy and improved treatment outcomes across healthcare settings
- Furthermore, rising demand for minimally invasive procedures, targeted drug delivery systems, and advanced diagnostic tools is establishing nanotechnology-based medical devices as a crucial component of modern healthcare. These converging factors are accelerating the uptake of nanotechnology-based medical device solutions, thereby significantly boosting the industry's growth
Nanotechnology Based Medical Device Market Analysis
- Nanotechnology-based medical devices, incorporating nanoscale materials and components for enhanced diagnostics, drug delivery, and therapeutic applications, are increasingly vital in modern healthcare systems across hospitals, research institutions, and specialized care settings due to their superior precision, efficiency, and ability to operate at the molecular level
- The escalating demand for nanotechnology-based medical devices is primarily fueled by advancements in nanomedicine, rising prevalence of chronic diseases, growing need for early and accurate diagnosis, and increasing preference for minimally invasive and targeted treatment solutions
- North America dominated the nanotechnology based medical device market with the largest revenue share of 41.3% in 2025, characterized by strong research infrastructure, high healthcare expenditure, and the presence of leading market players, with the U.S. experiencing substantial growth in adoption, particularly in advanced diagnostics and targeted drug delivery systems driven by continuous innovations in nanotechnology and precision medicine
- Asia-Pacific is expected to be the fastest growing region in the nanotechnology based medical device market during the forecast period due to increasing healthcare investments, expanding research activities, rising awareness of advanced medical technologies, and improving healthcare infrastructure
- The therapeutic applications segment held the largest market revenue share of 52.1% in 2025, driven by the extensive use of nanotechnology in drug delivery systems, cancer treatment, and regenerative medicine
Report Scope and Nanotechnology Based Medical Device Market Segmentation
|
Attributes |
Nanotechnology Based Medical Device Key Market Insights |
|
Segments Covered |
|
|
Countries Covered |
North America
Europe
Asia-Pacific
Middle East and Africa
South America
|
|
Key Market Players |
• Abbott (U.S.) |
|
Market Opportunities |
|
|
Value Added Data Infosets |
In addition to the insights on market scenarios such as market value, growth rate, segmentation, geographical coverage, and major players, the market reports curated by the Data Bridge Market Research also include in-depth expert analysis, patient epidemiology, pipeline analysis, pricing analysis, and regulatory framework. |
Nanotechnology Based Medical Device Market Trends
“Advancements in Precision Medicine and Miniaturization Driving Innovation”
- A significant and accelerating trend in the global nanotechnology based medical device market is the rapid advancement in precision medicine supported by nanoscale technologies, enabling highly targeted diagnostics and therapeutic interventions
- These innovations are transforming how diseases are detected, monitored, and treated at the cellular and molecular levels
- For instance, nanotechnology-enabled drug delivery systems are being increasingly incorporated into medical devices to ensure targeted delivery of therapeutics, particularly in oncology and cardiovascular applications, thereby improving treatment efficacy while minimizing side effects
- The continuous miniaturization of medical devices using nanomaterials is allowing the development of highly sensitive diagnostic tools, implantable devices, and biosensors capable of real-time monitoring of physiological parameters
- In addition, the integration of nanotechnology in imaging devices is enhancing resolution and accuracy, enabling early disease detection and improved clinical decision-making
- Growing research and development activities, along with increasing investments in nanomedicine, are accelerating the commercialization of advanced medical devices across various healthcare segments
- This trend towards more precise, efficient, and minimally invasive medical solutions is reshaping healthcare delivery, with a strong focus on improving patient outcomes and reducing treatment complexity
- The demand for nanotechnology-based devices is expanding across applications such as diagnostics, drug delivery, imaging, and regenerative medicine, driven by the need for advanced and personalized healthcare solutions.
Nanotechnology Based Medical Device Market Dynamics
Driver
“Growing Demand Driven by Advancements in Healthcare Technology and Rising Chronic Diseases”
- The increasing prevalence of chronic diseases such as cancer, cardiovascular disorders, and neurological conditions is a major driver for the growth of the Nanotechnology Based Medical Device market, as there is a growing need for advanced diagnostic and therapeutic solutions
- For instance, nanotechnology-based drug delivery devices are being widely explored and adopted in cancer treatment to deliver chemotherapeutic agents directly to tumor cells, improving treatment precision and reducing systemic toxicity
- Rapid advancements in healthcare technology, including nanomaterials, biosensors, and nano-enabled imaging systems, are enabling the development of innovative medical devices with enhanced functionality and accuracy
- The growing demand for early disease detection and real-time monitoring is further driving the adoption of nanoscale diagnostic devices and wearable health monitoring system
- In addition, increasing investments from both public and private sectors in nanotechnology research are supporting the development and commercialization of advanced medical devices
- The shift toward minimally invasive procedures and personalized medicine is also contributing significantly to market growth, as nanotechnology enables targeted and efficient treatment approaches
- Expanding applications of nanotechnology in areas such as regenerative medicine, tissue engineering, and implantable devices are further strengthening market expansion
- Moreover, the rising adoption of advanced healthcare infrastructure in emerging economies is creating new growth opportunities for nanotechnology-based medical devices
Restraint/Challenge
“High Development Costs and Regulatory Complexities”
- The high cost associated with the development and commercialization of nanotechnology-based medical devices remains a significant challenge, as these technologies require extensive research, specialized materials, and advanced manufacturing processes
- For instance, the development of nano-enabled drug delivery systems and diagnostic devices often involves lengthy clinical trials and stringent validation processes, increasing overall time-to-market and financial investment
- Regulatory complexities and lack of standardized guidelines for nanotechnology in healthcare can create uncertainties for manufacturers, slowing down product approvals and market entry
- In addition, concerns regarding the long-term safety and biocompatibility of nanomaterials may limit adoption, as healthcare providers and patients require strong clinical evidence of safety and effectiveness
- Limited awareness and understanding of nanotechnology among healthcare professionals in certain regions can further hinder adoption
- The need for highly skilled professionals and specialized infrastructure for manufacturing and deployment adds to the overall cost burden
- Furthermore, challenges related to scalability and consistent quality control during mass production can impact market growth
- Ethical considerations and environmental concerns regarding the use and disposal of nanomaterials also pose potential barriers
- Addressing these challenges through regulatory harmonization, increased funding for research, improved safety validation, and cost optimization strategies will be crucial for the sustained growth of the nanotechnology based medical device market
Nanotechnology Based Medical Device Market Scope
The market is segmented on the basis of product and application.
• By Product
On the basis of product, the Nanotechnology Based Medical Device market is segmented into active implantable devices, biochips, implantable materials, medical textiles and wound dressings, and others. The active implantable devices segment dominated the largest market revenue share of 34.7% in 2025, driven by the increasing adoption of advanced implantable technologies such as pacemakers, cochlear implants, and neurostimulators integrated with nanotechnology. These devices offer enhanced functionality, improved biocompatibility, and better patient outcomes compared to conventional devices. The growing prevalence of chronic diseases such as cardiovascular disorders and neurological conditions is significantly boosting demand. In addition, continuous advancements in nanomaterials are enabling the development of smaller, more efficient, and longer-lasting implantable devices. Healthcare providers are increasingly adopting these technologies due to their precision and reliability. The rising aging population is another key factor driving demand. Moreover, increasing healthcare expenditure and improved access to advanced medical treatments are supporting segment growth. The integration of smart monitoring capabilities further enhances their appeal. Overall, active implantable devices remain dominant due to their critical role in life-saving treatments.
The biochips segment is expected to witness the fastest CAGR of 22.6% from 2026 to 2033, driven by the growing demand for rapid diagnostics and personalized medicine. Biochips enable high-throughput analysis of biological samples, making them essential for early disease detection and monitoring. The increasing adoption of lab-on-a-chip technologies is significantly boosting this segment. In addition, advancements in nanotechnology are improving the sensitivity and accuracy of biochips. The rising prevalence of infectious diseases and the need for point-of-care testing are key drivers. Pharmaceutical and biotechnology companies are also utilizing biochips for drug discovery and research applications. Moreover, the shift toward precision medicine is accelerating demand. Government initiatives supporting advanced diagnostic technologies further contribute to growth. The expansion of healthcare infrastructure in emerging markets is also a positive factor. Overall, this segment is growing rapidly due to its wide range of applications and technological advancements.
• By Application
On the basis of application, the Nanotechnology Based Medical Device market is segmented into therapeutic applications, diagnostic applications, and research applications. The therapeutic applications segment held the largest market revenue share of 52.1% in 2025, driven by the extensive use of nanotechnology in drug delivery systems, cancer treatment, and regenerative medicine. Nanotechnology-based therapeutic devices enable targeted drug delivery, reducing side effects and improving treatment efficacy. The increasing prevalence of chronic diseases such as cancer and cardiovascular disorders is a major factor supporting growth. In addition, advancements in nanomedicine are enabling innovative treatment approaches. Healthcare providers are increasingly adopting these solutions due to their improved outcomes. The rising demand for minimally invasive treatments is also contributing to segment growth. Moreover, increasing investment in healthcare research and development is driving innovation. The availability of advanced treatment options is enhancing patient care. The segment also benefits from growing awareness about nanotechnology in medicine. Overall, therapeutic applications dominate due to their significant impact on treatment effectiveness.
The diagnostic applications segment is expected to witness the fastest CAGR of 24.3% from 2026 to 2033, driven by the increasing need for early and accurate disease detection. Nanotechnology is enabling the development of highly sensitive diagnostic tools capable of detecting diseases at an early stage. The growing demand for point-of-care diagnostics is a key driver. In addition, advancements in imaging technologies and biosensors are enhancing diagnostic capabilities. The rising prevalence of infectious diseases and the need for rapid testing solutions are further supporting growth. Healthcare systems are increasingly focusing on preventive care, boosting demand for advanced diagnostics. Moreover, the integration of AI and data analytics with diagnostic devices is improving efficiency. Government initiatives promoting early diagnosis are also contributing to growth. The expansion of diagnostic laboratories and healthcare infrastructure is another key factor. Overall, this segment is expected to grow rapidly due to its critical role in improving healthcare outcomes.
Nanotechnology Based Medical Device Market Regional Analysis
- North America dominated the nanotechnology based medical device market with the largest revenue share of 41.3% in 2025, characterized by strong research infrastructure, high healthcare expenditure, and the presence of leading market players. The region benefits from advanced healthcare systems and extensive adoption of innovative technologies in diagnostics, therapeutics, and patient monitoring
- Healthcare providers and research institutions in the region are increasingly leveraging nanotechnology to enhance precision in diagnostics and targeted drug delivery systems. For instance, the growing use of nanoparticle-based imaging agents and nano-enabled drug delivery platforms in oncology and cardiovascular treatments is significantly improving clinical outcomes and driving adoption
- This widespread adoption is further supported by strong government funding for research, well-established regulatory frameworks, and continuous advancements in precision medicine, establishing nanotechnology-based medical devices as a critical component of next-generation healthcare solutions
U.S. Nanotechnology Based Medical Device Market Insight
The U.S. nanotechnology based medical device market captured the largest revenue share in 2025 within North America, driven by substantial investments in research and development and rapid adoption of advanced medical technologies. The market is experiencing significant growth in areas such as advanced diagnostics, targeted drug delivery, and minimally invasive treatments. Continuous innovation in nanotechnology and precision medicine, along with the presence of leading biotechnology and medical device companies, is accelerating market expansion. In addition, increasing demand for early disease detection and personalized treatment approaches is further propelling the adoption of nanotechnology-based devices across healthcare facilities.
Europe Nanotechnology Based Medical Device Market Insight
The Europe nanotechnology based medical device market is projected to expand at a substantial CAGR throughout the forecast period, primarily driven by strong government support for research and innovation in nanomedicine and advanced healthcare technologies. The region is witnessing increasing adoption of nano-enabled diagnostic tools and therapeutic devices aimed at improving treatment accuracy and efficiency. In addition, stringent regulatory standards and a focus on patient safety are encouraging the development of high-quality, reliable nanotechnology-based medical devices. The growing aging population and rising prevalence of chronic diseases are further supporting market growth.
U.K. Nanotechnology Based Medical Device Market Insight
The U.K. nanotechnology based medical device market is anticipated to grow at a noteworthy CAGR during the forecast period, driven by increasing investments in healthcare innovation and a strong focus on advanced diagnostic and therapeutic solutions. The adoption of nanotechnology in medical devices is being supported by research collaborations between academic institutions and healthcare providers. In addition, the rising demand for early disease detection, improved patient outcomes, and efficient healthcare delivery systems is contributing to market expansion in the country.
Germany Nanotechnology Based Medical Device Market Insight
The Germany nanotechnology based medical device market is expected to expand at a considerable CAGR during the forecast period, fueled by the country’s strong engineering expertise, advanced healthcare infrastructure, and emphasis on technological innovation. Germany is witnessing increased adoption of nanotechnology in medical imaging, diagnostics, and implantable devices. The growing focus on precision medicine and high-quality healthcare solutions, along with strong industrial and research capabilities, is driving the development and deployment of advanced nanotechnology-based medical devices.
Asia-Pacific Nanotechnology Based Medical Device Market Insight
The Asia-Pacific nanotechnology based medical device market is poised to grow at the fastest CAGR during the forecast period, driven by increasing healthcare investments, expanding research activities, rising awareness of advanced medical technologies, and improving healthcare infrastructure. Rapid economic development and government initiatives supporting innovation in healthcare are accelerating the adoption of nanotechnology-based solutions. In addition, the growing burden of chronic diseases and demand for efficient diagnostic and treatment options are further fueling market growth across the region.
Japan Nanotechnology Based Medical Device Market Insight
The Japan nanotechnology based medical device market is gaining momentum due to the country’s strong focus on technological advancement, well-established healthcare system, and increasing demand for high-precision medical solutions. The integration of nanotechnology in diagnostics, imaging, and therapeutic applications is enabling improved accuracy and treatment efficiency. In addition, Japan’s aging population is driving demand for advanced medical devices that support early diagnosis and effective disease management.
China Nanotechnology Based Medical Device Market Insight
The China nanotechnology based medical device market accounted for the largest market revenue share in Asia-Pacific in 2025, attributed to increasing investments in healthcare infrastructure, expanding research capabilities, and strong government support for nanotechnology development. The rising prevalence of chronic diseases and growing demand for advanced diagnostic and therapeutic solutions are significantly driving market growth. Furthermore, the presence of domestic manufacturers, increasing focus on innovation, and improving accessibility to advanced medical technologies are strengthening the adoption of nanotechnology-based medical devices in China.
Nanotechnology Based Medical Device Market Share
The Nanotechnology Based Medical Device industry is primarily led by well-established companies, including:
• Abbott (U.S.)
• Medtronic (Ireland)
• Johnson & Johnson (U.S.)
• Boston Scientific Corporation (U.S.)
• GE HealthCare (U.S.)
• Siemens Healthineers (Germany)
• Stryker Corporation (U.S.)
• 3M (U.S.)
• Smith & Nephew (U.K.)
• Baxter International (U.S.)
• Thermo Fisher Scientific (U.S.)
• Nanosphere Inc. (U.S.)
• Nanobiotix (France)
• Arrowhead Pharmaceuticals (U.S.)
• Invitae Corporation (U.S.)
• Agilent Technologies (U.S.)
• PerkinElmer (U.S.)
• Bruker Corporation (U.S.)
• Oxford Instruments (U.K.)
• Carl Zeiss Meditec (Germany)
Latest Developments in Global Nanotechnology Based Medical Device Market
- In May 2021, Nanowear Inc., a U.S.-based nanotechnology healthcare company, announced a commercial distribution partnership with a leading aesthetic and reconstructive surgical device manufacturer to enhance post-surgical recovery monitoring. The collaboration leverages Nanowear’s cloth-based nanosensor platform to enable real-time patient monitoring, improving surgical outcomes and recovery management. This development highlights the growing integration of nanotechnology with digital health and remote patient monitoring solutions
- In September 2021, Nanowear Inc. announced that it received its third U.S. FDA 510(k) clearance for its nanotechnology-enabled diagnostic platform, expanding its capabilities in remote monitoring and digital diagnostics. The platform utilizes nanosensors embedded in wearable fabrics to continuously monitor multiple physiological parameters. This milestone reflects increasing regulatory acceptance of nanotechnology-enabled medical devices and their role in next-generation healthcare delivery
- In January 2024, Nanowear Inc. received U.S. FDA 510(k) clearance for its SimpleSense™-BP system, an AI-enabled nanotechnology-based wearable designed for continuous blood pressure monitoring and hypertension diagnosis. The system integrates nanosensors with AI-driven analytics to provide real-time cardiovascular insights. This development underscores the rising demand for non-invasive, continuous monitoring solutions powered by nanotechnology
- In March 2024, Boston Scientific received U.S. FDA approval for its AGENT drug-coated balloon catheter, a device designed to treat coronary in-stent restenosis. The device delivers a targeted drug coating to prevent artery re-narrowing, representing a significant advancement in minimally invasive cardiovascular treatment. This approval highlights the growing role of nanoscale drug-delivery technologies in improving therapeutic outcomes in interventional cardiology
- In June 2024, Moon Surgical announced U.S. FDA clearance for its Maestro System, an advanced surgical robotics platform incorporating nanoscale sensing and AI-enabled capabilities to enhance intraoperative precision. The system has been used in over 1,100 procedures across multiple surgical specialties. This development reflects the convergence of nanotechnology, robotics, and artificial intelligence in modern surgical devices.
- In March 2025, industry reports highlighted increasing commercialization of nanotechnology-enabled medical devices, driven by collaborations and partnerships among healthcare technology companies. These developments are accelerating the transition of nanotechnology innovations from research to clinical practice, particularly in diagnostics, drug delivery, and minimally invasive devices. The trend underscores strong industry momentum toward precision medicine and advanced healthcare solutions
- In June 2025, ongoing clinical advancements in nanorobotics and nanosensor-based medical devices were reported, including trials of AI-powered nano-robots capable of targeted drug delivery for brain tumors. These devices are designed to respond to cellular conditions and release drugs at optimal times, representing a breakthrough in personalized medicine. This development highlights the future potential of autonomous nanotechnology-based medical devices in transforming treatment approaches
- In November 2025, regulatory and industry updates indicated that multiple nanotechnology-enabled medical devices had gained FDA approvals, signaling increasing acceptance and commercialization of nanoscale innovations in healthcare. These devices include advanced diagnostic tools, biosensors, and drug-delivery systems. The development reflects a growing pipeline of nanotechnology-based solutions entering the global medical device market
SKU-
Get online access to the report on the World's First Market Intelligence Cloud
- Interactive Data Analysis Dashboard
- Company Analysis Dashboard for high growth potential opportunities
- Research Analyst Access for customization & queries
- Competitor Analysis with Interactive dashboard
- Latest News, Updates & Trend analysis
- Harness the Power of Benchmark Analysis for Comprehensive Competitor Tracking
Research Methodology
Data collection and base year analysis are done using data collection modules with large sample sizes. The stage includes obtaining market information or related data through various sources and strategies. It includes examining and planning all the data acquired from the past in advance. It likewise envelops the examination of information inconsistencies seen across different information sources. The market data is analysed and estimated using market statistical and coherent models. Also, market share analysis and key trend analysis are the major success factors in the market report. To know more, please request an analyst call or drop down your inquiry.
The key research methodology used by DBMR research team is data triangulation which involves data mining, analysis of the impact of data variables on the market and primary (industry expert) validation. Data models include Vendor Positioning Grid, Market Time Line Analysis, Market Overview and Guide, Company Positioning Grid, Patent Analysis, Pricing Analysis, Company Market Share Analysis, Standards of Measurement, Global versus Regional and Vendor Share Analysis. To know more about the research methodology, drop in an inquiry to speak to our industry experts.
Customization Available
Data Bridge Market Research is a leader in advanced formative research. We take pride in servicing our existing and new customers with data and analysis that match and suits their goal. The report can be customized to include price trend analysis of target brands understanding the market for additional countries (ask for the list of countries), clinical trial results data, literature review, refurbished market and product base analysis. Market analysis of target competitors can be analyzed from technology-based analysis to market portfolio strategies. We can add as many competitors that you require data about in the format and data style you are looking for. Our team of analysts can also provide you data in crude raw excel files pivot tables (Fact book) or can assist you in creating presentations from the data sets available in the report.