Global Parenteral Nutrition Associated Cholestasis Treatment Market
Market Size in USD Billion
CAGR :
% 
 USD
7.38 Billion
USD
11.24 Billion
2024
2032
USD
7.38 Billion
USD
11.24 Billion
2024
2032
| 2025 - 2032 | |
| USD 7.38 Billion | |
| USD 11.24 Billion | |
|
|
|
|
Parenteral Nutrition-Associated Cholestasis Treatment Market Size
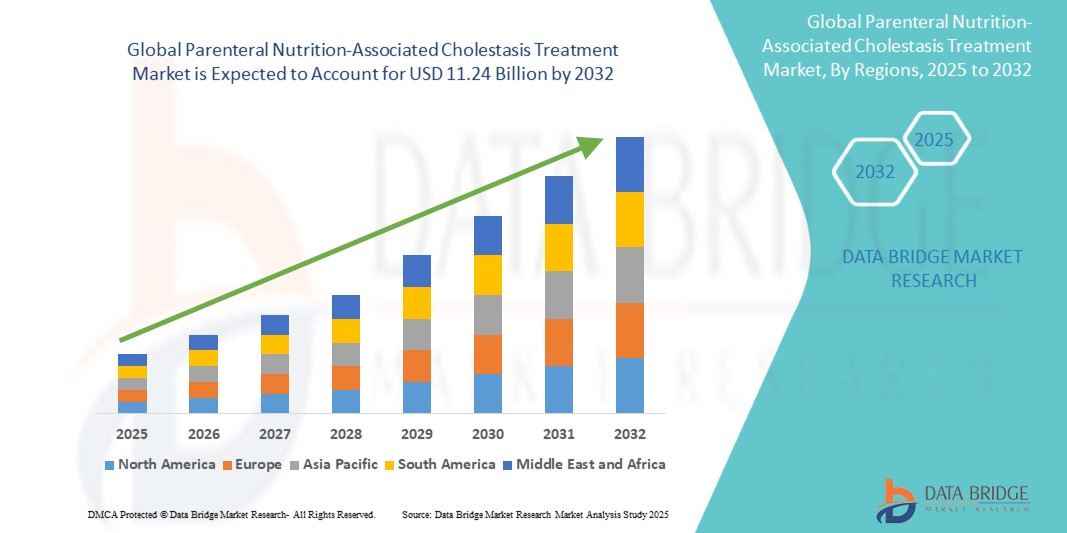
- The Global Parenteral Nutrition-Associated Cholestasis Treatment Market size was valued at USD 7.38 billion in 2024 and is expected to reach USD 11.24 billion by 2032, at a CAGR of 5.4% during the forecast period
- This growth is driven by factors such as the rising incidence of parenteral nutrition-associated cholestasis, increased awareness among clinicians, growing demand for safer lipid formulations, and advancements in diagnostic and monitoring technologies
Parenteral Nutrition-Associated Cholestasis Treatment Market Analysis
- Parenteral nutrition-associated cholestasis (PNAC) refers to a serious liver complication in preterm infants who are being administrated prolonged intravenous nutrition. Parenteral nutrition is defined as lifesaving intervention for many preterm infant’s other neonates suffering from severe illness
- The demand for Parenteral Nutrition-Associated Cholestasis Treatment is significantly driven by the increasing incidence of PNAC in neonates and patients requiring prolonged parenteral nutrition, heightened awareness among healthcare providers, advancements in lipid emulsions, and improved diagnostic and monitoring technologies
- North America is expected to dominate the Global Parenteral Nutrition-Associated Cholestasis Treatment market with a share of 36.4%, owing to its advanced neonatal and pediatric care infrastructure, high awareness among healthcare professionals, and strong presence of leading pharmaceutical and nutrition companies
- Asia-Pacific is projected to be the fastest-growing region in the Global Parenteral Nutrition-Associated Cholestasis Treatment market during the forecast period, driven by expanding healthcare access, increasing investments in neonatal care, and rising awareness of parenteral nutrition-related complications
- The Lipid Injectable Emulsion segment is anticipated to lead the market with a share of 34.5%, due to its essential role in minimizing cholestasis risk during long-term parenteral nutrition therapy. While emerging lipid formulations and novel treatment strategies are under development, traditional lipid emulsions remain the standard of care due to their proven efficacy, cost-effectiveness, and familiarity among clinicians
Report Scope and Parenteral Nutrition-Associated Cholestasis Treatment Market Segmentation
|
Attributes |
Parenteral Nutrition-Associated Cholestasis Treatment Key Market Insights |
|
Segments Covered |
|
|
Countries Covered |
North America
Europe
Asia-Pacific
Middle East and Africa
South America
|
|
Key Market Players |
|
|
Market Opportunities |
|
|
Value Added Data Infosets |
In addition to the insights on market scenarios such as market value, growth rate, segmentation, geographical coverage, and major players, the market reports curated by the Data Bridge Market Research also include import export analysis, production capacity overview, production consumption analysis, price trend analysis, climate change scenario, supply chain analysis, value chain analysis, raw material/consumables overview, vendor selection criteria, PESTLE Analysis, Porter Analysis, and regulatory framework. |
Parenteral Nutrition-Associated Cholestasis Treatment Market Trends
“Shift Toward Fish Oil-Based Lipid Emulsions and Personalized Nutrition Strategies to Mitigate PNAC Risk”
- One prominent trend in Parenteral Nutrition-Associated Cholestasis (PNAC) Treatment is the shift toward fish oil-based lipid emulsions and personalized nutrition strategies
- This shift is driven by increasing clinical evidence linking traditional soybean oil-based lipid emulsions to the development of PNAC in neonates and critically ill patients. Fish oil-based alternatives, rich in omega-3 fatty acids, have demonstrated anti-inflammatory properties and a lower incidence of liver complications
- For instance, healthcare providers are increasingly adopting products like Omegaven by Fresenius Kabi, a fish oil-based intravenous lipid emulsion, which has shown effectiveness in reducing cholestasis in pediatric patients on long-term parenteral nutrition
- This trend is significantly transforming the clinical management of PNAC by reducing liver-related complications, improving patient outcomes, and supporting a more tailored approach to nutrition therapy based on individual metabolic needs and risk profiles
- The PNAC Treatment market is poised for long-term growth, fueled by rising demand for safer lipid alternatives, advancements in nutritional science, and a broader move toward evidence-based, personalized medical nutrition strategies in neonatal and critical care settings
Parenteral Nutrition-Associated Cholestasis Treatment Market Dynamics
Driver
“Rising Incidence of Premature Births and Critically Ill Patients Fuel Demand for Parenteral Nutrition Solutions”
- The increasing technological advancements in Parenteral Nutrition-Associated Cholestasis Treatment are significantly driving the demand for more accurate and efficient diagnostic and surgical tools
- With continuous innovations such as magnetic tracers, radar reflectors, and wireless localization systems, healthcare providers can now offer more precise, non-invasive, and radiation-free alternatives to traditional wire-based localization methods
- As new technologies emerge, the ability to detect and localize small, non-palpable lesions has improved, enhancing the effectiveness of early-stage breast cancer treatment. These advancements enable clinicians to perform more targeted surgeries, minimizing the risk of complications and reducing recovery times for patients
- New technologies also allow for greater flexibility in scheduling procedures, reducing the stress and inconvenience associated with traditional methods. This is particularly important as hospitals and breast care centers aim to streamline operations and improve patient outcomes
- With advancements in imaging and localization technologies, the need for accurate, reliable, and minimally invasive solutions is increasing, ensuring better diagnosis and treatment planning for breast cancer patients. As a result, there is an ongoing shift toward adopting these advanced localization methods in clinical practice
For instance,
- Endomag's Magseed system is a leading magnetic seed localization product that has gained significant traction in the breast cancer surgery space. The product offers a radiation-free, wire-free alternative to traditional methods, providing increased surgical precision and improved patient comfort during procedures
- The rise in technological advancements is expected to continue as more healthcare facilities prioritize the use of cutting-edge tools for better patient outcomes and reduced surgical risks
Opportunity
“High Strategic Partnerships”
- The rising incidence of premature births and critically ill patients is significantly driving the demand for parenteral nutrition solutions
- As the number of premature births and critically ill patients requiring long-term parenteral nutrition increases, there is a greater need for specialized treatment options to prevent complications like Parenteral Nutrition-Associated Cholestasis (PNAC
- This growing patient population is placing a demand on healthcare systems to provide safe, effective, and customized nutrition therapies, particularly for neonates and those with gastrointestinal or liver disorders. Addressing PNAC through targeted lipid emulsions and personalized care is essential to improving the quality of care for these vulnerable patients
For instance,
- Hospitals in the U.S. are increasingly adopting Omegaven®, a fish oil-based lipid emulsion shown to reduce the incidence of PNAC in preterm infants who are dependent on parenteral nutrition for extended periods
- The rising prevalence of premature births and critically ill patients is expected to drive continued growth in the demand for specialized parenteral nutrition therapies, creating a critical need for solutions that can minimize the risks of PNAC while improving patient outcomes
- As healthcare providers focus on enhancing neonatal care and reducing complications in critically ill patients, there will be an increased emphasis on advancing parenteral nutrition strategies to meet the specific nutritional and medical needs of these patients
Restraint/Challenge
“High Costs and Limited Accessibility of Advanced Parenteral Nutrition Therapies Hinder Market Growth”
- The high cost of advanced parenteral nutrition therapies presents a significant challenge for the market, especially in developing regions
- Parenteral nutrition solutions, including specialized lipid emulsions and other treatment modalities to prevent or manage PNAC, can be prohibitively expensive. These costs limit access to advanced treatments, especially in lower-income or resource-constrained areas
- The price of these treatments, coupled with the need for specialized monitoring equipment and skilled medical professionals, can create financial barriers that prevent healthcare systems in developing countries from providing optimal care for at-risk patients
For instance,
- in many low- and middle-income countries, hospitals often struggle to afford high-quality, fish oil-based lipid emulsions like Omegaven, which are essential for reducing the risk of PNAC in premature infants. As a result, healthcare providers in these regions may resort to more affordable, but less effective, soybean oil-based emulsions, which may increase the risk of PNAC
- The financial burden associated with advanced therapies and the need for specialized medical infrastructure can create significant disparities in the availability of effective PNAC treatments, limiting access to care and hindering the growth of the market in underserved regions
Parenteral Nutrition-Associated Cholestasis Treatment Market Scope
The market is segmented on the basis of drug, treatment, mode of administration, distribution channel and end user.
|
Segmentation |
Sub-Segmentation |
|
By Drug |
|
|
By Treatment |
|
|
By Mode of Administration |
|
|
By Distribution Channel |
|
|
By End User |
|
In 2025, the lipid injectable emulsion is projected to dominate the market with a largest share in drug segment
The lipid injectable emulsion segment is expected to dominate the Parenteral Nutrition-Associated Cholestasis Treatment market with the largest share of 34.5% in 2025 due to its widespread clinical adoption, cost-effectiveness, and proven efficacy in managing PNAC in critically ill patients. Despite the development of alternative treatments and newer lipid formulations, traditional lipid emulsions, such as soybean oil-based emulsions, remain a standard option in many healthcare settings. These treatments are well-established, relatively simple to administer, and are familiar to healthcare providers. Their long-standing use in preventing PNAC and managing nutritional support in neonates and patients on extended parenteral nutrition continues to sustain their dominant position in the market.
The Surgery is expected to account for the largest share during the forecast period in treatment segment
In 2025, the Surgery segment is expected to dominate the market due to its established efficacy, cost-effectiveness, and broad clinical adoption. These emulsions, including fish oil-based and soybean oil-based formulations, play a critical role in managing PNAC in patients requiring long-term parenteral nutrition. Despite the emergence of new lipid formulations and therapies, traditional lipid emulsions continue to be the preferred treatment option due to their proven track record in reducing liver complications associated with PNAC. Their widespread use in neonatal and pediatric care, combined with the simplicity of administration and cost-effectiveness, contributes to their market dominance. Furthermore, ongoing clinical research and positive regulatory outcomes continue to support the sustained growth of this segment in the market.
Parenteral Nutrition-Associated Cholestasis Treatment Market Regional Analysis
“North America Holds the Largest Share in the Parenteral Nutrition-Associated Cholestasis Treatment Market”
- North America dominates the Parenteral Nutrition-Associated Cholestasis Treatment market with a share of 36.4%, driven by its advanced healthcare infrastructure, high adoption of innovative lipid emulsions, and the presence of key market players specializing in parenteral nutrition solutions
- The U.S. holds a significant share of 78.3%, primarily due to the increasing demand for advanced lipid emulsions, such as fish oil-based intravenous lipid formulations, to manage PNAC in preterm infants and critically ill patients. The high prevalence of conditions requiring long-term parenteral nutrition further fuels the market’s growth in this region
- The presence of leading companies like Fresenius Kabi, B. Braun, and Omegaven® producers, along with continuous advancements in lipid formulation technologies, continues to drive innovation in PNAC treatment. Significant investment in research and development (R&D) further supports the continuous improvement of nutritional therapies to prevent and manage PNAC
- The increasing prevalence of premature births, along with greater awareness of PNAC risks, supports the market’s growth. As healthcare standards improve and the understanding of PNAC expands, the demand for more effective, safer, and patient-specific lipid emulsions is expected to continue rising, solidifying North America’s dominant position in the market
“Asia-Pacific is Projected to Register the Highest CAGR in the Parenteral Nutrition-Associated Cholestasis Treatment Market”
- The Asia-Pacific region is expected to witness the highest growth rate in the Parenteral Nutrition-Associated Cholestasis Treatment market, driven by rapid healthcare infrastructure development, increasing awareness of neonatal and pediatric nutrition, and rising adoption of parenteral nutrition in clinical settings
- Countries such as China, India, and Japan are emerging as key markets for PNAC treatment due to the growing incidence of premature births and gastrointestinal disorders requiring long-term parenteral nutrition. Increased awareness of PNAC and government-supported neonatal care initiatives are further accelerating demand for effective treatment options
- Japan, with its advanced healthcare infrastructure and emphasis on pediatric care, remains a significant market for PNAC therapies. The country is increasingly adopting improved lipid emulsions and clinical protocols to reduce PNAC incidence and enhance patient outcomes in neonatal intensive care units (NICUs)
- The rising focus on expanding access to critical care and improving nutrition management for vulnerable populations is expected to drive the demand for advanced lipid-based therapies across the APAC region, making it the fastest-growing regional market for PNAC treatment during the forecast period
Parenteral Nutrition-Associated Cholestasis Treatment Market Share
The market competitive landscape provides details by competitor. Details included are company overview, company financials, revenue generated, market potential, investment in research and development, new market initiatives, global presence, production sites and facilities, production capacities, company strengths and weaknesses, product launch, product width and breadth, application dominance. The above data points provided are only related to the companies' focus related to market.
The Major Market Leaders Operating in the Market Are:
- Teva Pharmaceutical Industries Ltd (Israel)
- Zydus Cadila (India
- ALLERGAN (U.S.)
- Endo Pharmaceuticals Inc. (U.S.)
- ANI Pharmaceuticals, Inc. (U.S.)
- Mylan N.V. (U.S.) (formerly Netherlands, now part of Viatris headquartered in the U.S.)
- Glenmark Pharmaceuticals Ltd (India)
- Amneal Pharmaceuticals LLC (U.S.)
- Epic Pharma, LLC (U.S.)
- Lannett (U.S.)
- Fresenius Kabi AG (Germany)
SKU-
Get online access to the report on the World's First Market Intelligence Cloud
- Interactive Data Analysis Dashboard
- Company Analysis Dashboard for high growth potential opportunities
- Research Analyst Access for customization & queries
- Competitor Analysis with Interactive dashboard
- Latest News, Updates & Trend analysis
- Harness the Power of Benchmark Analysis for Comprehensive Competitor Tracking
Research Methodology
Data collection and base year analysis are done using data collection modules with large sample sizes. The stage includes obtaining market information or related data through various sources and strategies. It includes examining and planning all the data acquired from the past in advance. It likewise envelops the examination of information inconsistencies seen across different information sources. The market data is analysed and estimated using market statistical and coherent models. Also, market share analysis and key trend analysis are the major success factors in the market report. To know more, please request an analyst call or drop down your inquiry.
The key research methodology used by DBMR research team is data triangulation which involves data mining, analysis of the impact of data variables on the market and primary (industry expert) validation. Data models include Vendor Positioning Grid, Market Time Line Analysis, Market Overview and Guide, Company Positioning Grid, Patent Analysis, Pricing Analysis, Company Market Share Analysis, Standards of Measurement, Global versus Regional and Vendor Share Analysis. To know more about the research methodology, drop in an inquiry to speak to our industry experts.
Customization Available
Data Bridge Market Research is a leader in advanced formative research. We take pride in servicing our existing and new customers with data and analysis that match and suits their goal. The report can be customized to include price trend analysis of target brands understanding the market for additional countries (ask for the list of countries), clinical trial results data, literature review, refurbished market and product base analysis. Market analysis of target competitors can be analyzed from technology-based analysis to market portfolio strategies. We can add as many competitors that you require data about in the format and data style you are looking for. Our team of analysts can also provide you data in crude raw excel files pivot tables (Fact book) or can assist you in creating presentations from the data sets available in the report.