Global Point Of Care Molecular Imaging Devices Market
Market Size in USD Billion
CAGR :
% 
 USD
1.04 Billion
USD
4.56 Billion
2025
2033
USD
1.04 Billion
USD
4.56 Billion
2025
2033
| 2026 - 2033 | |
| USD 1.04 Billion | |
| USD 4.56 Billion | |
|
|
|
|
Point-of-Care Molecular Imaging Devices Market Size
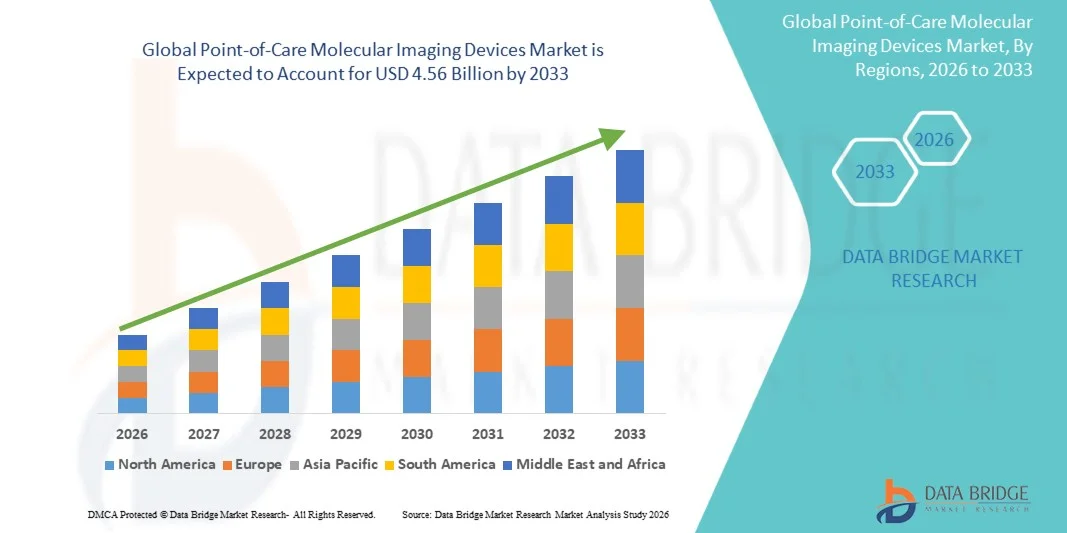
- The global point-of-care molecular imaging devices market size was valued at USD 1.04 billion in 2025 and is expected to reach USD 4.56 billion by 2033, at a CAGR of 20.30% during the forecast period
- The market growth is largely fueled by the increasing demand for rapid and accurate diagnostic solutions at the point of care, along with continuous technological advancements in molecular diagnostics and portable imaging platforms, enabling faster disease detection and improved clinical decision-making across healthcare settings
- Furthermore, the rising prevalence of infectious diseases and cancer, coupled with growing adoption of decentralized healthcare and personalized medicine, is establishing point-of-care molecular imaging devices as essential tools for timely diagnosis and monitoring. These converging factors are accelerating the adoption of advanced diagnostic systems, thereby significantly boosting the industry's growth
Point-of-Care Molecular Imaging Devices Market Analysis
- Point-of-care molecular imaging devices, designed to deliver rapid and highly sensitive diagnostic results directly at or near the patient care site, are becoming essential components of modern healthcare systems due to their ability to support early disease detection, real-time clinical decision-making, and decentralized diagnostic workflows across hospitals, clinics, and emergency settings
- The escalating demand for point-of-care molecular imaging devices is primarily fueled by the increasing prevalence of infectious diseases and cancer, growing demand for rapid diagnostic solutions, and continuous technological advancements in molecular diagnostics and portable imaging platforms
- North America dominated the point-of-care molecular imaging devices market with the largest revenue share of 39.8% in 2025, characterized by advanced healthcare infrastructure, strong adoption of innovative diagnostic technologies, and the presence of major biotechnology and medical device companies, with the U.S. experiencing substantial growth in point-of-care molecular testing across hospitals and diagnostic centers driven by rising investments in precision medicine and rapid diagnostic technologies
- Asia-Pacific is expected to be the fastest growing region in the point-of-care molecular imaging devices market during the forecast period due to expanding healthcare infrastructure, increasing government initiatives for early disease detection, and growing demand for rapid diagnostics in emerging economies
- The Polymerase Chain Reaction segment dominated the point-of-care molecular imaging devices market with a market share of 42.6% in 2025, driven by its high sensitivity, accuracy in pathogen detection, and widespread use in rapid molecular diagnostic platforms
Report Scope and Point-of-Care Molecular Imaging Devices Market Segmentation
|
Attributes |
Point-of-Care Molecular Imaging Devices Key Market Insights |
|
Segments Covered |
|
|
Countries Covered |
North America
Europe
Asia-Pacific
Middle East and Africa
South America
|
|
Key Market Players |
|
|
Market Opportunities |
|
|
Value Added Data Infosets |
In addition to the insights on market scenarios such as market value, growth rate, segmentation, geographical coverage, and major players, the market reports curated by the Data Bridge Market Research also include in-depth expert analysis, patient epidemiology, pipeline analysis, pricing analysis, and regulatory framework |
Point-of-Care Molecular Imaging Devices Market Trends
“Advancement of Rapid Molecular Diagnostics and Portable Testing Platforms”
- A significant and accelerating trend in the global point-of-care molecular imaging devices market is the increasing development of compact, rapid diagnostic platforms capable of delivering highly sensitive molecular test results directly at the patient care site. This technological progress is significantly improving the speed and efficiency of clinical diagnostics
- For instance, the Abbott Laboratories ID NOW molecular platform enables healthcare professionals to detect infectious diseases within minutes at the point of care. Similarly, the Cepheid GeneXpert system allows rapid molecular testing for conditions such as tuberculosis and respiratory infections in decentralized healthcare settings
- Integration of advanced molecular technologies in portable diagnostic devices enables features such as automated sample preparation, real-time nucleic acid amplification, and rapid pathogen identification. For instance, some modern molecular diagnostic systems incorporate cartridge-based testing that minimizes manual handling and improves testing accuracy. Furthermore, portable testing platforms provide clinicians with the ability to perform diagnostic tests directly in emergency rooms, outpatient clinics, and remote healthcare facilities
- The seamless integration of point-of-care molecular imaging devices with digital healthcare systems facilitates efficient data management and faster clinical decision-making. Through connected diagnostic platforms, healthcare providers can access test results, patient records, and disease surveillance data within a single system, improving patient care coordination and diagnostic workflows
- This trend toward faster, portable, and highly accurate molecular diagnostic systems is fundamentally transforming the way diseases are detected and managed in healthcare environments. Consequently, companies such as Roche Diagnostics are developing advanced molecular testing platforms capable of delivering rapid and reliable results directly at the point of care
- The demand for point-of-care molecular imaging devices that offer rapid diagnostics and portable testing capabilities is growing rapidly across hospitals, diagnostic centers, and community healthcare settings, as healthcare providers increasingly prioritize early disease detection and efficient clinical decision-making
- In addition, growing investment in next-generation molecular diagnostic technologies and miniaturized testing systems is enabling manufacturers to develop faster, more compact, and cost-efficient point-of-care molecular imaging devices for widespread clinical use
Point-of-Care Molecular Imaging Devices Market Dynamics
Driver
“Rising Demand for Rapid and Decentralized Diagnostic Solutions”
- The increasing need for rapid disease detection and decentralized diagnostic services, combined with the growing burden of infectious diseases and chronic conditions, is a significant driver for the heightened demand for point-of-care molecular imaging devices
- For instance, in March 2024, Roche announced the expansion of its molecular diagnostic portfolio to support rapid infectious disease detection in decentralized healthcare environments. Such strategies by key companies are expected to drive the point-of-care molecular imaging devices industry growth in the forecast period
- As healthcare systems focus more on early diagnosis and faster treatment decisions, point-of-care molecular imaging devices provide clinicians with the ability to obtain highly accurate diagnostic results within a short timeframe compared to traditional laboratory testing methods
- Furthermore, the increasing adoption of precision medicine and personalized healthcare approaches is making rapid molecular diagnostics an essential component of modern healthcare systems, enabling healthcare providers to tailor treatments based on accurate and timely diagnostic information
- The ability to conduct molecular testing directly at hospitals, clinics, and emergency departments significantly improves patient outcomes by reducing diagnostic turnaround times and enabling quicker therapeutic interventions. The increasing adoption of rapid molecular testing solutions in both developed and emerging healthcare markets further contributes to market growth
- Growing government initiatives supporting early disease detection and improved diagnostic infrastructure are also encouraging healthcare institutions to invest in advanced point-of-care molecular testing technologies
- In addition, increasing global focus on pandemic preparedness and infectious disease surveillance is boosting the adoption of rapid molecular diagnostic systems capable of delivering accurate results within minutes
Restraint/Challenge
“High Equipment Costs and Complex Regulatory Approval Processes”
- Concerns surrounding the high cost of advanced molecular diagnostic equipment and stringent regulatory approval requirements pose a significant challenge to broader market penetration. As point-of-care molecular imaging devices require sophisticated technologies and validation procedures, their adoption may be limited in resource-constrained healthcare environments
- For instance, strict regulatory approval pathways from agencies such as the U.S. Food and Drug Administration and the European Medicines Agency require extensive clinical validation to ensure safety and diagnostic accuracy
- Addressing these regulatory challenges through streamlined approval processes and robust clinical validation studies is essential for accelerating the availability of new point-of-care diagnostic technologies. Companies such as bioMérieux emphasize rigorous testing and quality assurance in the development of their molecular diagnostic platforms to meet global regulatory standards
- In addition, the relatively high initial cost of some advanced molecular diagnostic systems compared to conventional laboratory testing can be a barrier to adoption for smaller healthcare facilities, particularly in developing regions with limited healthcare budgets
- While technological advancements are gradually reducing the cost of molecular diagnostic devices, the perceived high investment required for installing and maintaining these systems can still hinder widespread adoption in certain healthcare settings
- Limited availability of skilled healthcare professionals trained to operate advanced molecular diagnostic equipment may further restrict the deployment of point-of-care molecular imaging devices in some healthcare facilities
- Furthermore, challenges related to device standardization, quality control, and integration with existing hospital laboratory systems can complicate the implementation of these diagnostic technologies across healthcare networks
Point-of-Care Molecular Imaging Devices Market Scope
The market is segmented on the basis of product, technology, application, and end user.
- By Product
On the basis of product, the point-of-care molecular imaging devices market is segmented into instruments, analyzers, assays & kits, and software & services. The assays & kits segment dominated the market with the largest market revenue share in 2025, driven by the high and recurring demand for diagnostic reagents used in molecular testing. These consumables are essential for detecting pathogens, genetic markers, and disease biomarkers during molecular diagnostic procedures. Healthcare providers rely heavily on assay kits for rapid and accurate detection of infectious diseases and cancer-related markers. The frequent need to replenish these kits during routine diagnostic testing further contributes to their dominant market share. Moreover, the increasing number of diagnostic tests performed globally and the expansion of molecular testing programs are supporting the strong demand for assays and kits across healthcare facilities.
The software & services segment is anticipated to witness the fastest growth rate during the forecast period from 2026 to 2033, fueled by the growing need for data management, digital integration, and automated diagnostic workflows. Advanced software platforms help healthcare providers interpret molecular diagnostic data quickly and improve clinical decision-making. Increasing adoption of connected diagnostic devices and cloud-based healthcare systems is further boosting demand for integrated software solutions. In addition, service offerings such as maintenance, calibration, and technical support are becoming critical as healthcare facilities adopt advanced molecular diagnostic systems. The expansion of digital healthcare infrastructure and AI-assisted diagnostics is expected to further accelerate growth in this segment.
- By Technology
On the basis of technology, the point-of-care molecular imaging devices market is segmented into polymerase chain reaction (PCR), isothermal nucleic acid amplification technology (INAAT), genetic sequencing-based, hybridization-based, microarray-based, and others. The polymerase chain reaction (PCR) segment dominated the market with the largest revenue share of 42.6% in 2025 due to its high sensitivity, accuracy, and reliability in detecting genetic material from pathogens and disease markers. PCR technology is widely used in the diagnosis of infectious diseases such as COVID-19, tuberculosis, and influenza. Its ability to amplify small amounts of DNA or RNA makes it a highly effective tool for early disease detection. The widespread availability of PCR-based testing platforms and strong regulatory approval for PCR diagnostics also contribute to its dominance. In addition, many point-of-care diagnostic platforms are designed around PCR technology, further strengthening its leading position in the market.
The isothermal nucleic acid amplification technology (INAAT) segment is expected to witness the fastest growth rate from 2026 to 2033, driven by its ability to perform rapid molecular testing without the need for complex thermal cycling equipment. INAAT technologies enable faster test results compared to conventional PCR methods, making them ideal for point-of-care settings. These technologies are increasingly being adopted in portable diagnostic devices designed for emergency rooms, clinics, and remote healthcare facilities. The simplicity of operation and reduced equipment requirements make INAAT-based systems highly suitable for decentralized healthcare environments. Growing research investments and technological advancements in rapid molecular testing platforms are expected to further accelerate growth in this segment.
- By Application
On the basis of application, the point-of-care molecular imaging devices market is segmented into infectious diseases, oncology, respiratory diseases, sexually transmitted diseases, hepatitis, gastrointestinal disorders, and others. The infectious diseases segment dominated the market with the largest revenue share in 2025 due to the rising global burden of infectious diseases and the need for rapid diagnosis to prevent disease transmission. Point-of-care molecular diagnostic devices allow healthcare professionals to quickly detect pathogens such as viruses, bacteria, and parasites. Early and accurate diagnosis is essential for initiating timely treatment and controlling outbreaks. The growing prevalence of diseases such as influenza, tuberculosis, and emerging viral infections has significantly increased demand for rapid molecular diagnostic testing. In addition, government initiatives focused on strengthening infectious disease surveillance programs are supporting the growth of this segment.
The oncology segment is anticipated to witness the fastest growth rate during the forecast period from 2026 to 2033, driven by increasing adoption of molecular diagnostics in cancer detection and monitoring. Point-of-care molecular imaging devices are increasingly used to identify cancer-related genetic mutations and biomarkers. Early detection of cancer significantly improves treatment outcomes and survival rates, which is encouraging healthcare providers to adopt advanced diagnostic technologies. The rising prevalence of cancer worldwide and growing awareness about early screening programs are also supporting this segment’s growth. Furthermore, advancements in precision medicine and personalized oncology treatments are expected to further increase the demand for rapid molecular cancer diagnostics.
- By End User
On the basis of end user, the point-of-care molecular imaging devices market is segmented into hospitals & ICUs, physician offices / clinics, diagnostic laboratories, research institutes, and others. The hospitals & ICUs segment dominated the market with the largest market revenue share in 2025, primarily due to the high patient volume and the need for immediate diagnostic results in critical care settings. Hospitals frequently rely on rapid molecular diagnostic systems to support emergency decision-making and patient management. The presence of advanced healthcare infrastructure and trained medical professionals also supports the adoption of sophisticated diagnostic technologies in hospitals. In addition, hospitals conduct a large number of diagnostic tests daily, contributing to the high utilization of point-of-care molecular diagnostic devices. Increasing investments in hospital diagnostic capabilities are further strengthening this segment’s dominance.
The physician offices / clinics segment is expected to witness the fastest growth rate from 2026 to 2033, driven by the growing shift toward decentralized healthcare and near-patient testing. Clinics are increasingly adopting portable molecular diagnostic devices to provide faster test results without relying on centralized laboratories. This trend improves patient convenience and reduces diagnostic turnaround times. The availability of compact and user-friendly diagnostic systems is making it easier for clinics to perform molecular testing on-site. Furthermore, the increasing demand for outpatient healthcare services and early disease detection is expected to significantly boost adoption of point-of-care molecular imaging devices in physician offices and clinics.
Point-of-Care Molecular Imaging Devices Market Regional Analysis
- North America dominated the point-of-care molecular imaging devices market with the largest revenue share of 39.8% in 2025, characterized by advanced healthcare infrastructure, strong adoption of innovative diagnostic technologies, and the presence of major biotechnology and medical device companies
- Healthcare providers in the region highly prioritize rapid and accurate diagnostic technologies, along with the integration of advanced molecular testing platforms that enable early disease detection and improved clinical decision-making in hospitals and diagnostic centers
- This widespread adoption is further supported by strong healthcare spending, a well-established diagnostic ecosystem, and growing investments in molecular diagnostics and precision medicine, establishing point-of-care molecular imaging devices as a critical component of modern healthcare systems across the region
U.S. Point-of-Care Molecular Imaging Devices Market Insight
The U.S. point-of-care molecular imaging devices market captured the largest revenue share of 79% in 2025 within North America, fueled by the strong adoption of advanced diagnostic technologies and the increasing demand for rapid disease detection solutions. Healthcare providers are increasingly prioritizing faster and more accurate diagnostic tools that can deliver results directly at the point of care. The growing prevalence of infectious diseases and cancer, combined with rising healthcare spending, further propels the market growth. Moreover, the expanding adoption of precision medicine and molecular diagnostics in hospitals and diagnostic laboratories is significantly contributing to the market's expansion.
Europe Point-of-Care Molecular Imaging Devices Market Insight
The Europe point-of-care molecular imaging devices market is projected to expand at a substantial CAGR throughout the forecast period, primarily driven by increasing demand for early disease detection and the strong presence of advanced healthcare systems across the region. The growing burden of chronic and infectious diseases, coupled with the demand for rapid diagnostic technologies, is fostering the adoption of point-of-care molecular diagnostic devices. European healthcare providers are also increasingly adopting decentralized diagnostic solutions to improve patient outcomes and reduce diagnostic turnaround times. The region is experiencing significant growth across hospitals, diagnostic laboratories, and outpatient clinics, with advanced molecular diagnostic devices being incorporated into both routine clinical testing and disease screening programs.
U.K. Point-of-Care Molecular Imaging Devices Market Insight
The U.K. point-of-care molecular imaging devices market is anticipated to grow at a noteworthy CAGR during the forecast period, driven by the increasing demand for rapid diagnostic technologies and the rising focus on early disease detection. In addition, the growing prevalence of infectious diseases and the need for efficient patient management are encouraging healthcare providers to adopt advanced molecular diagnostic solutions. The country’s strong healthcare infrastructure and increasing investment in innovative diagnostic technologies are expected to further stimulate market growth.
Germany Point-of-Care Molecular Imaging Devices Market Insight
The Germany point-of-care molecular imaging devices market is expected to expand at a considerable CAGR during the forecast period, fueled by increasing awareness of advanced molecular diagnostics and the demand for high-precision diagnostic technologies. Germany’s well-developed healthcare infrastructure, combined with strong research and development capabilities, promotes the adoption of point-of-care molecular diagnostic devices. The integration of rapid molecular testing technologies into hospital diagnostic systems is also becoming increasingly prevalent, with a strong focus on improving early disease detection and clinical decision-making efficiency.
Asia-Pacific Point-of-Care Molecular Imaging Devices Market Insight
The Asia-Pacific point-of-care molecular imaging devices market is poised to grow at the fastest CAGR of 23.4% during the forecast period of 2026 to 2033, driven by expanding healthcare infrastructure, rising healthcare expenditures, and increasing demand for rapid diagnostic technologies in emerging economies such as China, Japan, and India. The region’s growing focus on early disease detection and infectious disease surveillance is driving the adoption of point-of-care molecular diagnostic devices. Furthermore, increasing government initiatives supporting healthcare modernization and diagnostic accessibility are expanding the availability of advanced diagnostic technologies across the region.
Japan Point-of-Care Molecular Imaging Devices Market Insight
The Japan point-of-care molecular imaging devices market is gaining momentum due to the country’s strong healthcare infrastructure, advanced diagnostic capabilities, and growing demand for rapid molecular testing solutions. The Japanese healthcare system places a strong emphasis on early disease diagnosis and preventive healthcare strategies. The adoption of point-of-care molecular diagnostic devices is driven by increasing investments in medical technologies and the rising prevalence of chronic diseases. Moreover, the integration of advanced molecular diagnostic platforms into hospitals and research institutions is fueling growth across the country.
India Point-of-Care Molecular Imaging Devices Market Insight
The India point-of-care molecular imaging devices market accounted for the largest market revenue share in Asia Pacific in 2025, attributed to the country’s rapidly expanding healthcare infrastructure and increasing demand for affordable diagnostic solutions. India is emerging as a key market for molecular diagnostics due to the rising burden of infectious diseases and growing awareness regarding early disease detection. The expansion of diagnostic laboratories and healthcare facilities across urban and semi-urban regions is supporting market growth. In addition, government initiatives promoting healthcare accessibility and disease screening programs are further driving the adoption of point-of-care molecular diagnostic devices in India.
Point-of-Care Molecular Imaging Devices Market Share
The Point-of-Care Molecular Imaging Devices industry is primarily led by well-established companies, including:
- Abbott (U.S.)
- F. Hoffmann-La Roche Ltd (Switzerland)
- BIOMÉRIEUX (France)
- BD (U.S.)
- Thermo Fisher Scientific Inc. (U.S.)
- Siemens Healthineers AG (Germany)
- Hologic, Inc. (U.S.)
- Bio-Rad Laboratories, Inc. (U.S.)
- QIAGEN N.V. (Netherlands)
- QuidelOrtho Corporation (U.S.)
- DiaSorin S.p.A. (Italy)
- Visby Medical, Inc. (U.S.)
- Cue Health Inc. (U.S.)
- Binx Health, Inc. (U.S.)
- Molbio Diagnostics Pvt. Ltd. (India)
- Oxford Nanopore Technologies plc (U.K.)
- SD Biosensor, Inc. (South Korea)
- Seegene Inc. (South Korea)
- Novacyt S.A. (France)
- Genedrive plc (U.K.)
What are the Recent Developments in Global Point-of-Care Molecular Imaging Devices Market?
- In December 2025, Roche announced that the U.S. Food and Drug Administration (FDA) granted 510(k) clearance and CLIA waiver for its cobas® liat molecular point-of-care test for detecting Bordetella infections, including whooping cough. The test delivers PCR-accurate results in about 15 minutes, enabling physicians to diagnose patients directly during clinical visits and start treatment immediately
- In January 2025, Roche received FDA 510(k) clearance and CLIA waiver for its cobas® liat multiplex assay panels designed to diagnose sexually transmitted infections such as chlamydia, gonorrhea, and Mycoplasma genitalium at the point of care. The PCR-based test provides results in about 20 minutes, allowing clinicians to diagnose and treat patients in the same visit
- In March 2024, Cepheid announced that the U.S. FDA cleared its Xpert® Xpress GBS test, a molecular diagnostic assay used for detecting Group B Streptococcus in pregnant women. The test runs on the company’s GeneXpert® system, enabling rapid molecular detection to help prevent newborn infections during childbirth. The launch enhances the company’s molecular point-of-care testing portfolio and supports improved maternal and neonatal healthcare outcomes
- In January 2024, Cepheid received FDA clearance with CLIA waiver for its Xpert® Xpress MVP multiplex vaginal panel, which can diagnose bacterial vaginosis, vulvovaginal candidiasis, and trichomoniasis from a single sample. The test can be performed in near-patient environments and provides results in about 60 minutes, expanding molecular testing access in clinics and community healthcare settings
- In March 2022, Roche Diagnostics launched the cobas® 5800 System, a molecular diagnostic platform designed to expand testing capacity and improve laboratory efficiency. The system supports growing demand for molecular diagnostics by enabling automated and high-throughput testing workflows. This development reflects the increasing adoption of advanced molecular diagnostic systems aimed at improving disease detection and laboratory productivity
SKU-
Get online access to the report on the World's First Market Intelligence Cloud
- Interactive Data Analysis Dashboard
- Company Analysis Dashboard for high growth potential opportunities
- Research Analyst Access for customization & queries
- Competitor Analysis with Interactive dashboard
- Latest News, Updates & Trend analysis
- Harness the Power of Benchmark Analysis for Comprehensive Competitor Tracking
Research Methodology
Data collection and base year analysis are done using data collection modules with large sample sizes. The stage includes obtaining market information or related data through various sources and strategies. It includes examining and planning all the data acquired from the past in advance. It likewise envelops the examination of information inconsistencies seen across different information sources. The market data is analysed and estimated using market statistical and coherent models. Also, market share analysis and key trend analysis are the major success factors in the market report. To know more, please request an analyst call or drop down your inquiry.
The key research methodology used by DBMR research team is data triangulation which involves data mining, analysis of the impact of data variables on the market and primary (industry expert) validation. Data models include Vendor Positioning Grid, Market Time Line Analysis, Market Overview and Guide, Company Positioning Grid, Patent Analysis, Pricing Analysis, Company Market Share Analysis, Standards of Measurement, Global versus Regional and Vendor Share Analysis. To know more about the research methodology, drop in an inquiry to speak to our industry experts.
Customization Available
Data Bridge Market Research is a leader in advanced formative research. We take pride in servicing our existing and new customers with data and analysis that match and suits their goal. The report can be customized to include price trend analysis of target brands understanding the market for additional countries (ask for the list of countries), clinical trial results data, literature review, refurbished market and product base analysis. Market analysis of target competitors can be analyzed from technology-based analysis to market portfolio strategies. We can add as many competitors that you require data about in the format and data style you are looking for. Our team of analysts can also provide you data in crude raw excel files pivot tables (Fact book) or can assist you in creating presentations from the data sets available in the report.