Global Programmable Rna Therapeutics Market
Market Size in USD Billion
CAGR :
% 
 USD
1.16 Billion
USD
4.92 Billion
2025
2033
USD
1.16 Billion
USD
4.92 Billion
2025
2033
| 2026 - 2033 | |
| USD 1.16 Billion | |
| USD 4.92 Billion | |
|
|
|
|
Programmable RNA Therapeutics Market Size
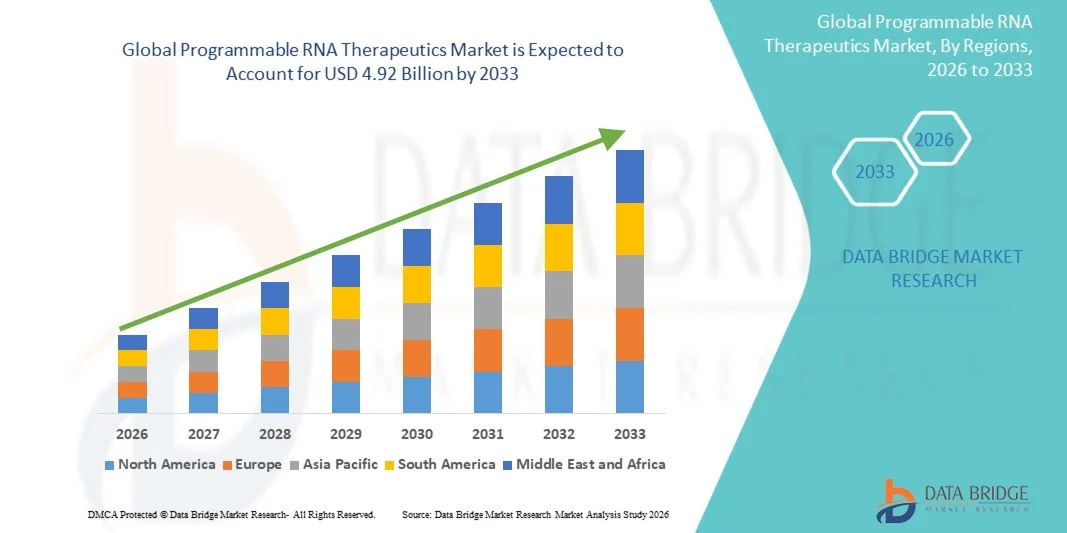
- The global Programmable RNA Therapeutics market size was valued at USD 1.16 billion in 2025 and is expected to reach USD 4.92 billion by 2033, at a CAGR of19.80% during the forecast period
- The market growth is largely fueled by rapid advancements in RNA-based technologies, growing research in gene therapies, and increasing adoption of precision medicine approaches, leading to higher demand for programmable RNA therapeutics in treating complex diseases
- Furthermore, rising investment from pharmaceutical and biotechnology companies, coupled with increasing clinical trials targeting oncology, infectious diseases, and rare genetic disorders, is establishing programmable RNA therapeutics as a critical component of next-generation therapeutics. These converging factors are accelerating the uptake of programmable RNA therapeutics solutions, thereby significantly boosting the market’s growth
Programmable RNA Therapeutics Market Analysis
- Programmable RNA therapeutics, including mRNA, siRNA, and antisense RNA therapies, are increasingly vital in modern medicine due to their potential to treat complex diseases such as cancer, genetic disorders, and infectious diseases with high precision and personalized approaches
- The escalating demand for programmable RNA therapeutics is primarily fueled by rapid advancements in RNA-based technology, increasing clinical trials, and growing adoption by pharmaceutical and biotechnology companies seeking next-generation therapeutics
- North America dominated the Programmable RNA Therapeutics market with the largest revenue share of approximately 42.3% in 2025, driven by high R&D investments, early adoption of RNA therapies, and a strong presence of leading pharmaceutical and biotechnology companies
- Asia-Pacific is expected to be the fastest-growing region in the Programmable RNA Therapeutics market during the forecast period, with a projected CAGR of 10.2%, supported by increasing clinical research activities, expanding biotech infrastructure, and rising healthcare investments in countries like China, Japan, and India
- The mRNA therapeutics segment dominated the largest market revenue share of 45.3% in 2025, driven by widespread adoption in vaccines and therapeutic applications
Report Scope and Programmable RNA Therapeutics Market Segmentation
|
Attributes |
Programmable RNA Therapeutics Key Market Insights |
|
Segments Covered |
|
|
Countries Covered |
North America
Europe
Asia-Pacific
Middle East and Africa
South America
|
|
Key Market Players |
• Moderna, Inc. (U.S.) |
|
Market Opportunities |
|
|
Value Added Data Infosets |
In addition to the insights on market scenarios such as market value, growth rate, segmentation, geographical coverage, and major players, the market reports curated by the Data Bridge Market Research also include in-depth expert analysis, patient epidemiology, pipeline analysis, pricing analysis, and regulatory framework. |
Programmable RNA Therapeutics Market Trends
“Enhanced Focus on Targeted and Personalized Therapies”
- A significant and accelerating trend in the global Programmable RNA Therapeutics market is the increasing focus on developing targeted and personalized RNA-based treatments for rare diseases, oncology, and infectious diseases
- In 2025, mRNA-based therapeutics accounted for approximately 35% of the global market revenue, highlighting the growing preference for precision medicine approaches
- For instance, Moderna and BioNTech expanded their RNA platforms in 2024–2025 to target multiple cancer indications and rare genetic disorders, enabling highly personalized treatment regimens across North America, Europe, and Asia-Pacific. Similarly, CureVac entered clinical trials for novel RNA therapeutics targeting rare metabolic diseases, showcasing global investment in personalized solutions
- Another trend is the expansion of RNA-based platforms to include self-amplifying RNA, circular RNA, and small interfering RNA (siRNA) technologies
- These advancements increase therapeutic efficacy, reduce dosing frequency, and improve patient compliance. By 2025, siRNA therapeutics contributed roughly 20% of the total RNA therapeutics pipeline
- Increased collaboration between pharmaceutical companies and research institutions worldwide is driving innovation. Partnerships are enabling access to advanced RNA delivery technologies, scalable manufacturing platforms, and novel biomarker strategies, supporting global adoption across emerging and developed markets
Programmable RNA Therapeutics Market Dynamics
Driver
“Rising Prevalence of Target Diseases and Biotech Investments”
- The market growth is primarily driven by the increasing prevalence of genetic disorders, cancers, and infectious diseases worldwide, which necessitate innovative RNA-based therapeutics. Chronic and rare diseases accounted for nearly 45% of global RNA therapeutic research programs in 2025
- For instance, BioNTech initiated multiple oncology and rare disease RNA therapeutic trials across Europe, North America, and Asia-Pacific in 2025, reflecting the high global demand for targeted RNA solutions. Similarly, Moderna expanded its infectious disease RNA pipeline in emerging regions, including Latin America and Southeast Asia
- Rising investments in biotechnology and pharmaceutical R&D are further accelerating market growth. Global investment in RNA-based drug development increased by 12% CAGR from 2022 to 2025, with North America contributing roughly 50% of total funding
- Expansion of contract development and manufacturing organizations (CDMOs) globally is supporting large-scale production of RNA therapeutics, reducing time-to-market and increasing accessibility
- Government initiatives, such as the U.S. National Institutes of Health (NIH) funding programs and the European Union’s Innovative Medicines Initiative (IMI), are driving innovation and adoption of RNA therapeutics, particularly for rare and unmet medical conditions
Restraint/Challenge
“High Costs, Regulatory Hurdles, and Delivery Limitations”
- High development and manufacturing costs for RNA therapeutics remain a major barrier, especially for emerging markets
- For instance, In 2025, production costs accounted for approximately 60% of the total therapy expenditure globally
- Stringent regulatory requirements across regions, including the FDA (U.S.), EMA (Europe), and PMDA (Japan), can slow clinical trial approvals and limit rapid commercialization. Differences in regulatory frameworks between countries also complicate multi-regional trial planning
- Challenges associated with safe and efficient delivery of RNA molecules, including stability, immunogenicity, and targeted cellular uptake, can impact therapeutic efficacy and limit market penetration. Approximately 25% of RNA therapeutics in early-stage trials faced delivery-related challenges in 2025
- Intellectual property and patent disputes, particularly in high-demand RNA technology areas, can further delay commercialization
- Overcoming these challenges requires continued innovation in delivery systems, streamlined regulatory pathways, scalable manufacturing, and strategic collaborations to ensure broader global accessibility
Programmable RNA Therapeutics Market Scope
The market is segmented on the basis of type, therapeutic area/application, and end user.
- By Type of RNA Therapeutics
On the basis of type, the Programmable RNA Therapeutics market is segmented into mRNA therapeutics, siRNA therapeutics, antisense RNA (ASO) therapeutics, and others. The mRNA therapeutics segment dominated the largest market revenue share of 45.3% in 2025, driven by widespread adoption in vaccines and therapeutic applications. The segment benefits from strong R&D pipelines, high efficacy, and scalable manufacturing processes. Pharmaceutical and biotechnology companies prioritize mRNA therapeutics due to their versatility across multiple diseases. Strong regulatory approvals and ongoing clinical trials further support growth. Collaborative efforts between biotech firms and research institutes expand its commercial reach. Moreover, mRNA therapeutics are favored for their rapid development timelines and adaptability in emerging infectious diseases. High investments in RNA-based drug development and government incentives in key regions reinforce dominance. mRNA technology also demonstrates robust performance in immuno-oncology and cardiovascular therapies. Commercial launches of mRNA vaccines for COVID-19 have accelerated global adoption. Demand for personalized medicine applications contributes to strong market penetration.
The siRNA therapeutics segment is anticipated to witness the fastest CAGR of 18.9% from 2026 to 2033, fueled by innovations in gene silencing therapies and targeted treatments for genetic disorders. Advanced delivery systems, including lipid nanoparticles and conjugates, improve efficacy and safety. Biotech collaborations and partnerships with CROs for clinical trials accelerate development. Expansion of RNA therapeutics in rare diseases and oncology increases adoption. Governments and private investors are supporting clinical research, providing funding and regulatory facilitation. Awareness campaigns and scientific publications promote therapeutic benefits. Emerging markets are witnessing rapid uptake due to affordability and accessibility improvements. Precision medicine applications further drive growth. Strong pipeline development across Asia-Pacific, North America, and Europe boosts momentum. Technological advancements in RNA stabilization and delivery enhance market potential. Increasing licensing and technology transfer agreements among pharmaceutical companies expand global reach. The segment is becoming a key focus area for innovation and long-term growth in RNA therapeutics.
- By Therapeutic Area/Application
On the basis of therapeutic area, the market is segmented into oncology, infectious diseases, cardiovascular diseases, genetic disorders, neurological disorders, and others. The oncology segment held the largest market revenue share of 38.7% in 2025, driven by high prevalence of cancers and increasing adoption of RNA-based therapies. RNA therapeutics allow targeted therapy, immuno-oncology applications, and combination regimens. Pharmaceutical and biotechnology companies prioritize oncology applications due to strong clinical outcomes and commercial potential. Government and private funding for oncology research support market growth. Increasing global clinical trials for RNA-based cancer therapies contribute to expansion. Technological advances in RNA delivery and formulation improve efficacy. The availability of mRNA vaccines and RNA-based immunotherapies further boosts adoption. Oncology-focused RNA therapeutics pipelines remain strong across North America, Europe, and Asia-Pacific. Collaborations between biotech firms and research institutions accelerate development and commercialization. Real-world evidence demonstrates improved patient outcomes, reinforcing preference for RNA therapies. The segment benefits from favorable reimbursement policies in key regions. Rising awareness among healthcare providers and patients enhances acceptance and market penetration.
The genetic disorders segment is expected to witness the fastest CAGR of 19.3% from 2026 to 2033, driven by the increasing need for effective treatments for rare and inherited diseases. RNA-based therapies enable gene-specific targeting and precision medicine approaches. Innovations in delivery systems and chemical modifications enhance stability and therapeutic effect. Collaborations between biotech firms, academic research centers, and CROs accelerate clinical development. Government initiatives and incentives support orphan drug development, expanding adoption. Expanding awareness of RNA therapeutics for inherited disorders drives patient demand. Advances in diagnostics and genomic profiling facilitate personalized treatments. The growing prevalence of rare genetic conditions globally reinforces market potential. Asia-Pacific and North America are emerging as key growth hubs due to regulatory support. Funding for clinical research and public-private partnerships accelerates development. Pharmaceutical companies continue to expand pipelines targeting rare diseases. Increasing licensing and commercialization agreements further fuel market expansion.
- By End User
On the basis of end user, the market is segmented into pharmaceutical & biotechnology companies, CROs, academic & research institutes, and hospitals & clinics. The pharmaceutical & biotechnology companies segment dominated the largest market revenue share of 52.1% in 2025, due to extensive R&D pipelines, advanced manufacturing, and strategic collaborations. Companies leverage RNA therapeutics for clinical and commercial applications across oncology, infectious diseases, and genetic disorders. Strong investment in RNA technology, patent portfolios, and regulatory approvals support dominance. Strategic partnerships with CROs facilitate clinical trials and global expansion. Access to advanced delivery technologies and scalable production enables faster commercialization. The segment benefits from government grants and incentives for innovative therapeutics. High market competition encourages continuous pipeline development. Adoption of RNA therapeutics for personalized medicine enhances segment growth. Pharmaceutical and biotechnology companies play a critical role in commercializing RNA-based therapies. Increasing focus on mRNA vaccines and gene-silencing therapies boosts revenue share. The segment maintains dominance through technological expertise and strategic alliances.
The CROs segment is expected to witness the fastest CAGR of 17.8% from 2026 to 2033, driven by outsourcing of RNA therapeutic development, specialized expertise in RNA delivery, and global clinical trial management. Partnerships with pharmaceutical companies for trial execution, regulatory compliance, and large-scale production accelerate growth. CROs offer cost-effective solutions, shortening timelines for RNA-based therapy development. Increasing clinical research activities, contract manufacturing, and regional expansion contribute to adoption. Emerging markets in Asia-Pacific provide opportunities for CRO growth due to favorable regulations. Technological platforms for RNA stability and delivery enhance service offerings. Rising demand for rare disease and oncology trials expands opportunities. CROs benefit from collaborations with academic institutions for early-stage research. Digitalization and AI-driven trial management improve operational efficiency. Increasing pharmaceutical outsourcing strengthens segment growth globally. Regulatory support for clinical trials in key regions accelerates adoption. CROs continue to expand capabilities to meet growing RNA therapeutics demand.
Programmable RNA Therapeutics Market Regional Analysis
- North America dominated the programmable RNA therapeutics market with the largest revenue share of approximately 42.3% in 2025, driven by high R&D investments, early adoption of RNA therapies, and a strong presence of leading pharmaceutical and biotechnology companies
- The region benefits from advanced clinical trial infrastructure, availability of skilled research professionals, and a supportive regulatory environment, which facilitates rapid development and commercialization of novel RNA therapeutics
- Major players such as Moderna, BioNTech, Pfizer, and Arcturus Therapeutics are actively expanding their RNA pipelines across oncology, infectious diseases, and rare genetic disorders, contributing to sustained market growth
U.S. Programmable RNA Therapeutics Market Insight
The U.S. programmable RNA therapeutics market captured the largest revenue share in 2025 within North America, fueled by extensive clinical research programs, high patient awareness, and robust adoption of RNA-based therapeutics. Leading companies such as Moderna, BioNTech, Pfizer, and CureVac are leveraging advanced lipid nanoparticle delivery systems, collaborations with academic institutions, and expansive clinical trial networks to drive innovation. The country’s strong biotech ecosystem, coupled with early regulatory approvals and supportive government funding for RNA research, is accelerating the introduction of new RNA therapeutics into both domestic and global markets.
Europe Programmable RNA Therapeutics Market Insight
The Europe programmable RNA therapeutics market is projected to expand at a substantial CAGR throughout the forecast period, driven by the presence of a mature biotechnology sector, high patient awareness, and supportive healthcare policies. Key markets such as the U.K. and Germany are leading regional growth. Companies including CureVac, BioNTech, Evotec, and Moderna Europe are focusing on expanding clinical trials and commercial operations in oncology, rare diseases, and infectious disease therapeutics. Urbanization, increased healthcare expenditure, and demand for personalized medicine are further fostering the adoption of RNA therapeutics across both private and public healthcare settings.
U.K. Programmable RNA Therapeutics Market Insight
The U.K. programmable RNA therapeutics market is anticipated to grow at a noteworthy CAGR during the forecast period, driven by the strong presence of biotechnology firms, advanced healthcare infrastructure, and growing patient acceptance of RNA-based therapies. Rising concerns for effective treatment options in oncology and rare diseases are encouraging both public and private sector investment. The U.K.’s robust research collaborations and commercial pipelines, supported by companies like BioNTech and Moderna, continue to accelerate clinical trials and therapeutic adoption.
Germany Programmable RNA Therapeutics Market Insight
The Germany programmable RNA therapeutics market is expected to expand at a considerable CAGR during the forecast period, fueled by a strong emphasis on innovation, digital healthcare adoption, and regulatory support for advanced therapeutics. Germany’s well-developed pharmaceutical manufacturing capabilities, combined with demand for personalized medicine, are driving the growth of RNA therapeutics. Key players such as CureVac, BioNTech, and Evotec are actively investing in local R&D, clinical trials, and commercialization efforts to strengthen the country’s position in the global RNA therapeutics market.
Asia-Pacific Programmable RNA Therapeutics Market Insight
The Asia-Pacific programmable RNA therapeutics market is poised to grow at the fastest CAGR of 10.2% during the forecast period, supported by increasing clinical research activities, expanding biotechnology infrastructure, and rising healthcare investments in countries like China, Japan, and India. The region’s large patient population, government-backed RNA research initiatives, and increasing adoption of RNA therapeutics in oncology and rare diseases are key growth drivers. Companies such as Walvax Biotechnology, Suzhou Abogen Biosciences, Moderna Japan, and BioNTech Japan are expanding clinical trials and manufacturing capacities, further accelerating market development.
Japan Programmable RNA Therapeutics Market Insight
Japan’s programmable RNA therapeutics market is gaining momentum due to its advanced healthcare infrastructure, strong R&D capabilities, and emphasis on precision medicine. The country’s focus on innovative RNA therapeutics, coupled with rising adoption in oncology and rare genetic conditions, is driving growth. Major players including Moderna Japan and BioNTech Japan are actively collaborating with local hospitals and research institutions to expand clinical trials and commercial availability.
China Programmable RNA Therapeutics Market Insight
China programmable RNA therapeutics market accounted for the largest market revenue share in Asia-Pacific in 2025, driven by rapid urbanization, increasing healthcare investments, and strong domestic manufacturing capabilities. The country is rapidly becoming a hub for RNA clinical trials and production, with companies like Walvax Biotechnology, Suzhou Abogen Biosciences, and Fosun Pharma leading development. The government’s focus on biotechnology innovation, combined with increasing patient access and affordability, is further propelling the adoption of RNA therapeutics across both public and private healthcare sectors.
Programmable RNA Therapeutics Market Share
The Programmable RNA Therapeutics industry is primarily led by well-established companies, including:
• Moderna, Inc. (U.S.)
• BioNTech SE (Germany)
• CureVac N.V. (Germany)
• Alnylam Pharmaceuticals, Inc. (U.S.)
• Arrowhead Pharmaceuticals, Inc. (U.S.)
• Dicerna Pharmaceuticals, Inc. (U.S.)
• Ribometrix, Inc. (U.S.)
• eTheRNA immunotherapies (Belgium)
• Beam Therapeutics (U.S.)
• Sarepta Therapeutics, Inc. (U.S.)
• Ionis Pharmaceuticals, Inc. (U.S.)
• Sanofi Genzyme (France)
• Pfizer Inc. (U.S.)
• GlaxoSmithKline plc (GSK) (U.K.)
Latest Developments in Global Programmable RNA Therapeutics Market
- In March 2025, clinical trial data showed that lepodisiran, an extended‑duration small interfering RNA (siRNA) therapy developed by Eli Lilly, significantly reduced lipoprotein(a) levels by nearly 94 % at the highest tested dose in adults with elevated cardiovascular risk, highlighting the therapeutic potential of programmable RNA agents in cardiometabolic diseases
- In April 2025, Novartis announced the acquisition of Regulus Therapeutics, a company developing microRNA‑17 inhibitor farabursen (RGLS8429) for autosomal dominant polycystic kidney disease, expanding its RNA‑based drug development portfolio and reinforcing investment in RNA‑targeted medicine
- In June 2025, Eli Lilly completed its acquisition of Verve Therapeutics, bolstering its pipeline of in vivo RNA gene‑editing therapies targeting PCSK9 for cardiovascular disease and signalling strategic expansion into programmable RNA‑based genetic treatments
- In August 2025, RNA industry reports identified strong growth trends in the programmable RNA therapeutics and vaccines market, driven by ongoing expansion of mRNA and RNAi technologies, advanced delivery systems such as lipid nanoparticles (LNPs), and broader application in oncology, rare diseases, and infectious disease treatments, reflecting accelerating innovation and commercialisation of RNA therapeutics
- In September 2025, biotech firm Mana.bio launched Mina, a large‑language model platform designed for lipid nanoparticle (LNP) design and optimisation to accelerate RNA therapy delivery research, while also entering collaborative research partnerships to enhance RNA delivery efficiency for cancer and autoimmune disease applications
- In October 2025, shares of Avidity Biosciences surged more than 40 % after a major acquisition announcement by Novartis to buy the company, emphasizing increased investor confidence and strategic focus on RNA‑based drug platforms including Antibody‑Oligonucleotide Conjugates
SKU-
Get online access to the report on the World's First Market Intelligence Cloud
- Interactive Data Analysis Dashboard
- Company Analysis Dashboard for high growth potential opportunities
- Research Analyst Access for customization & queries
- Competitor Analysis with Interactive dashboard
- Latest News, Updates & Trend analysis
- Harness the Power of Benchmark Analysis for Comprehensive Competitor Tracking
Research Methodology
Data collection and base year analysis are done using data collection modules with large sample sizes. The stage includes obtaining market information or related data through various sources and strategies. It includes examining and planning all the data acquired from the past in advance. It likewise envelops the examination of information inconsistencies seen across different information sources. The market data is analysed and estimated using market statistical and coherent models. Also, market share analysis and key trend analysis are the major success factors in the market report. To know more, please request an analyst call or drop down your inquiry.
The key research methodology used by DBMR research team is data triangulation which involves data mining, analysis of the impact of data variables on the market and primary (industry expert) validation. Data models include Vendor Positioning Grid, Market Time Line Analysis, Market Overview and Guide, Company Positioning Grid, Patent Analysis, Pricing Analysis, Company Market Share Analysis, Standards of Measurement, Global versus Regional and Vendor Share Analysis. To know more about the research methodology, drop in an inquiry to speak to our industry experts.
Customization Available
Data Bridge Market Research is a leader in advanced formative research. We take pride in servicing our existing and new customers with data and analysis that match and suits their goal. The report can be customized to include price trend analysis of target brands understanding the market for additional countries (ask for the list of countries), clinical trial results data, literature review, refurbished market and product base analysis. Market analysis of target competitors can be analyzed from technology-based analysis to market portfolio strategies. We can add as many competitors that you require data about in the format and data style you are looking for. Our team of analysts can also provide you data in crude raw excel files pivot tables (Fact book) or can assist you in creating presentations from the data sets available in the report.