Global Prothrombin Time Testing Market
Market Size in USD Billion
CAGR :
% 
 USD
1.31 Billion
USD
2.05 Billion
2025
2033
USD
1.31 Billion
USD
2.05 Billion
2025
2033
| 2026 –2033 | |
| USD 1.31 Billion | |
| USD 2.05 Billion | |
|
|
|
|
Prothrombin Time Testing Market Size
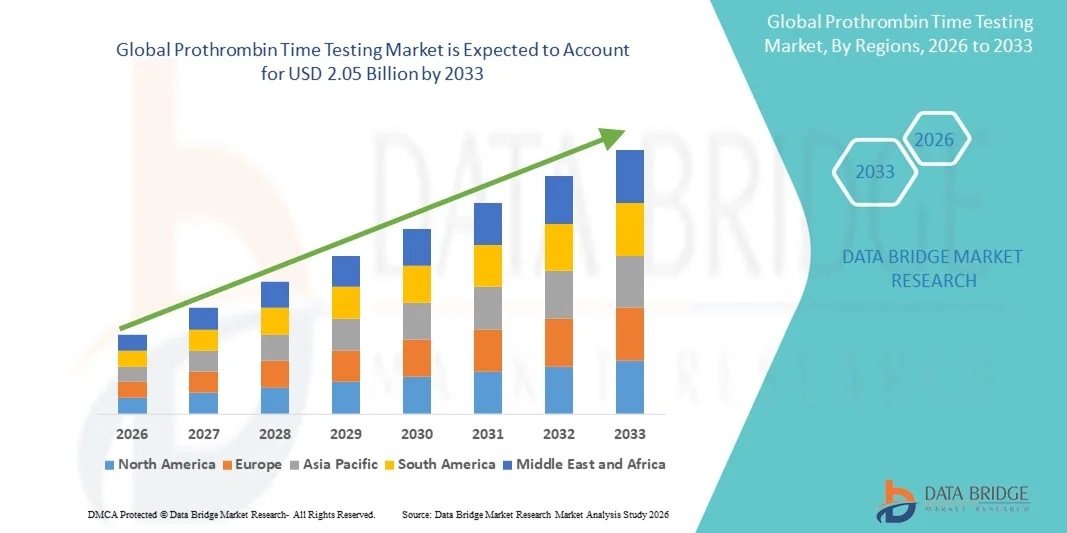
- The global prothrombin time testing market size was valued at USD 1.31 billion in 2025 and is expected to reach USD 2.05 billion by 2033, at a CAGR of 5.77% during the forecast period
- The market growth is largely fueled by increasing awareness of cardiovascular disorders, rising demand for rapid diagnostic solutions, and technological advancements in point-of-care testing, leading to improved patient management in both clinical and home care settings
- Furthermore, the growing emphasis on early detection of blood clotting abnormalities and enhanced laboratory efficiency is accelerating the adoption of Prothrombin Time Testing solutions, thereby significantly boosting the industry's growth
Prothrombin Time Testing Market Analysis
- Prothrombin Time Testing, providing rapid and accurate assessment of blood coagulation, is increasingly vital in both hospital and pre-hospital settings due to its ability to guide anticoagulant therapy, detect clotting disorders, and support critical patient care decisions
- The escalating demand for prothrombin time testing is primarily fueled by growing prevalence of cardiovascular and bleeding disorders, rising adoption of point-of-care diagnostic technologies, and increasing awareness among healthcare providers about the importance of timely coagulation monitoring
- North America dominated the prothrombin time testing market with the largest revenue share of 43% in 2025, driven by advanced healthcare infrastructure, high adoption of diagnostic technologies, and the presence of key market players, with the U.S. experiencing substantial growth in Prothrombin Time Testing installations, particularly in hospitals and diagnostic laboratories, fueled by innovations in point-of-care testing and laboratory automation
- Asia-Pacific is expected to be the fastest growing region in the prothrombin time testing market during the forecast period due to increasing healthcare access, rising government investments in diagnostics, and growing awareness of cardiovascular and bleeding disorders in countries such as China, India, and Japan
- The prothrombin time (PT) segment dominated the largest market revenue share of 48.1% in 2025, supported by the growing prevalence of cardiovascular disorders and widespread use of anticoagulant therapies
Report Scope and Prothrombin Time Testing Market Segmentation
|
Attributes |
Prothrombin Time Testing Key Market Insights |
|
Segments Covered |
|
|
Countries Covered |
North America
Europe
Asia-Pacific
Middle East and Africa
South America
|
|
Key Market Players |
|
|
Market Opportunities |
|
|
Value Added Data Infosets |
In addition to the insights on market scenarios such as market value, growth rate, segmentation, geographical coverage, and major players, the market reports curated by the Data Bridge Market Research also include in-depth expert analysis, patient epidemiology, pipeline analysis, pricing analysis, and regulatory framework. |
Prothrombin Time Testing Market Trends
Growing Adoption of Point-of-Care and Portable Diagnostic Devices
- A significant and accelerating trend in the global prothrombin time testing market is the increasing adoption of point-of-care (POC) and portable testing devices, enabling rapid coagulation monitoring in hospitals, clinics, and home care settings
- Portable PT/INR analyzers provide timely results, reducing dependency on central laboratories and facilitating immediate clinical decisions for anticoagulated patients
- For instance, in March 2023, Roche Diagnostics launched the CoaguChek INRange system, a portable PT/INR testing device designed for home use, enabling patients to self-monitor anticoagulation therapy safely and accurately
- The trend toward minimally invasive and easy-to-use devices is driving user preference, particularly in outpatient and emergency care scenarios, where fast results are critical for patient safety
- Integration with electronic health records (EHR) and cloud-based reporting platforms is also becoming a key feature, supporting seamless data management and monitoring for healthcare providers
- The market is further benefiting from increasing awareness among physicians and patients regarding the importance of regular coagulation monitoring for cardiovascular and thromboembolic condition
- Overall, the shift toward portable, user-friendly, and connected PT testing solutions is fundamentally reshaping diagnostic workflows and patient management strategies
Prothrombin Time Testing Market Dynamics
Driver
Rising Incidence of Cardiovascular and Thromboembolic Disorders
- The increasing prevalence of cardiovascular diseases, deep vein thrombosis, pulmonary embolism, and atrial fibrillation is a major driver for the global Prothrombin Time Testing market
- For instance, in April 2025, Roche Diagnostics expanded its CoaguChek platform with enhanced test strips and improved accuracy for anticoagulation therapy management, addressing growing patient demand
- Chronic conditions requiring long-term anticoagulation, such as atrial fibrillation and mechanical heart valves, are fueling demand for regular PT/INR monitoring in both clinical and home-care settings
- Healthcare providers are increasingly recommending routine monitoring to reduce bleeding and thrombotic risks, which boosts adoption of automated and rapid PT testing systems
- The growing geriatric population and rising awareness about preventive healthcare further amplify demand, particularly in developed and urban markets
- In addition, emerging markets are witnessing increased hospital infrastructure development and point-of-care adoption, which is expected to drive market expansion in the forecast period
Restraint/Challenge
High Device Costs and Reimbursement Constraints
- The relatively high cost of advanced PT testing devices and consumables remains a challenge for widespread adoption, especially in cost-sensitive regions
- For instance, some portable coagulation analyzers carry premium prices due to precision engineering and regulatory compliance requirements, limiting accessibility in smaller clinics and rural areas
- Reimbursement policies for PT/INR testing vary significantly across countries, impacting the affordability of routine monitoring and adoption in home-care settings
- The need for regular calibration, maintenance, and trained personnel to ensure accurate results also presents operational challenges for healthcare facilities
- While prices for basic PT testing kits are gradually decreasing, high-end automated analyzers with connectivity features remain relatively expensive
- Overcoming these barriers through device cost optimization, simplified operation, and expanded reimbursement coverage will be essential to sustain long-term growth of the Prothrombin Time Testing market
Prothrombin Time Testing Market Scope
The market is segmented on the basis of product, technology, application, and end-user.
- By Product
On the basis of product, the Prothrombin Time Testing market is segmented into instruments, consumables, and systems. The instruments segment dominated the largest market revenue share of 45.6% in 2025, driven by the widespread adoption of automated coagulation analyzers in hospitals and diagnostic laboratories. Instruments provide reliable and accurate measurements, reducing human error and facilitating standardized testing across multiple settings. The growing prevalence of cardiovascular diseases and anticoagulant therapies is driving steady demand for high-quality testing instruments. Healthcare providers prioritize instruments for their durability, precision, and integration capabilities with laboratory information systems (LIS). Manufacturers are also offering portable and semi-automated instruments to cater to point-of-care and home-based monitoring. In addition, innovations in user-friendly interfaces and rapid analysis functions further enhance adoption. Instruments are compatible with various test kits and reagents, providing flexibility and reducing operational costs. Training programs and service support offered by top manufacturers are boosting market confidence. Government initiatives promoting cardiovascular health and preventive care also support instrument adoption. Overall, instruments continue to dominate the PT testing market due to their reliability, accuracy, and institutional preference.
The consumables segment is expected to witness the fastest CAGR of 19.2% from 2026 to 2033, fueled by rising testing volumes and frequent replacement requirements. Consumables include reagents, test strips, and cuvettes essential for accurate PT measurements. Increasing patient monitoring and anticoagulant therapy programs drive steady consumable usage. Innovations in reagent stability and storage further support the rapid adoption of consumables. Point-of-care settings and home-use kits contribute to rising demand. Manufacturers are focusing on reducing assay time and enhancing shelf-life, increasing overall market appeal. Expanding awareness of self-monitoring in chronic disease management also drives growth. Rising adoption in emerging regions, where testing volumes are rapidly increasing, further accelerates consumable demand.
- By Technology
On the basis of technology, the market is segmented into mechanical, electrochemical, optical, and others. The electrochemical technology segment held the largest revenue share of 42.8% in 2025, owing to its high sensitivity, reliability, and compatibility with automated analyzers. Electrochemical analyzers offer rapid, precise measurements with minimal sample requirements, ideal for hospitals and diagnostic laboratories. Adoption is driven by the need for efficient and reproducible results in anticoagulant therapy management. Integration with electronic health records and laboratory systems enhances workflow efficiency. Advancements in miniaturization and portable electrochemical analyzers are further supporting market demand. Standardization and regulatory approvals contribute to widespread acceptance. Hospitals and large clinical laboratories prefer electrochemical technology for consistent performance and reproducibility. In addition, cost-effective maintenance and reagent usage make electrochemical analyzers a preferred choice.
The optical technology segment is expected to witness the fastest CAGR of 18.5% from 2026 to 2033, driven by non-invasive, rapid, and accurate detection capabilities. Optical analyzers are increasingly used in point-of-care testing and outpatient diagnostics. Continuous innovations in LED-based detection and miniaturized spectrophotometers are boosting adoption. Optical systems also facilitate multiplex testing and integration with automated platforms, increasing efficiency. Rising use in home-care and ambulatory monitoring settings contributes to high growth rates.
- By Application
On the basis of application, the market is segmented into thrombin time (TT), activated partial thromboplastin time (APTT), prothrombin time (PT), partial thromboplastin time (PTT), and others. The prothrombin time (PT) segment dominated the largest market revenue share of 48.1% in 2025, supported by the growing prevalence of cardiovascular disorders and widespread use of anticoagulant therapies. PT testing is essential for monitoring warfarin therapy and managing coagulation disorders, making it a critical diagnostic parameter in hospitals and clinical laboratories. The segment benefits from standardized testing protocols and frequent testing requirements. Rising awareness about thrombotic conditions and pre-surgical coagulation assessment further drives demand. Advanced PT testing instruments and integration with hospital systems support increased adoption. The availability of home-use PT testing devices also enhances market penetration.
The activated partial thromboplastin time (APTT) segment is expected to witness the fastest CAGR of 17.9% from 2026 to 2033, driven by its critical role in monitoring heparin therapy and detecting a wide range of coagulation abnormalities. APTT testing is increasingly adopted across point-of-care settings, hospitals, and home-based environments, providing healthcare professionals and patients with rapid and reliable results for timely clinical decision-making. The growing prevalence of thrombotic disorders and the need for continuous anticoagulant management further boost the demand for APTT tests. The development of automated analyzers enhances throughput and reduces human error, while portable and compact devices facilitate testing in non-traditional settings, including remote or resource-limited locations. Rising awareness among clinicians and patients about the importance of early detection and regular monitoring of coagulation profiles is encouraging wider adoption
- By End-User
On the basis of end-user, the market is segmented into diagnostic laboratories, hospitals, clinical laboratories, point-of-care testing (POCT), home care settings, and others. The hospitals segment held the largest market revenue share of 44.3% in 2025, supported by the concentration of advanced diagnostic infrastructure, trained personnel, and high patient throughput. Hospitals prefer integrated PT and APTT testing solutions to monitor anticoagulant therapy, manage emergencies, and perform preoperative assessments. High adoption is fueled by government funding, implementation of standardized protocols, and the presence of automated analyzers. Hospitals benefit from rapid turnaround times, reliability, and comprehensive reporting capabilities, which improve patient care outcomes. Frequent testing requirements, particularly for cardiovascular and critical care patients, ensure consistent demand. The presence of advanced laboratory facilities and trained personnel enables hospitals to handle high volumes efficiently. Integration with hospital information systems streamlines workflow and data management. Hospitals also invest in both portable and fixed analyzers for flexible deployment. Patient awareness programs and preventive healthcare initiatives further support the segment. In addition, hospitals increasingly adopt POC integration to enhance bedside testing. Strong infrastructure, regulatory compliance, and clinical expertise contribute to the dominance of this end-user segment.
The home care settings segment is expected to witness the fastest CAGR of 20.1% from 2026 to 2033, driven by the increasing prevalence of chronic cardiovascular conditions and patient preference for self-monitoring. The growing availability of portable PT/INR and APTT devices enables patients to manage anticoagulation therapy independently at home. Telemedicine integration and remote monitoring solutions facilitate real-time feedback and reduce hospital visits. Rising awareness among patients about anticoagulation management and the importance of regular testing accelerates adoption. Easy-to-use home-based analyzers with digital connectivity allow data sharing with clinicians, ensuring accurate and timely care. Growing geriatric population and increased prevalence of thrombotic disorders further contribute to segment growth. Home care monitoring reduces healthcare costs and supports chronic disease management. Continuous innovation in miniaturized and portable devices increases convenience and accuracy for patients. Remote guidance from healthcare professionals enhances confidence in home-based testing. The segment also benefits from rising adoption in rural and semi-urban areas where access to laboratories is limited. Education and training for patients and caregivers strengthen segment acceptance. Overall, the home care settings segment is the fastest-growing due to patient-centric healthcare trends and technology-enabled self-monitoring solutions.
Prothrombin Time Testing Market Regional Analysis
- North America dominated the prothrombin time testing market with the largest revenue share of 43% in 2025, driven by advanced healthcare infrastructure, high adoption of diagnostic technologies, and the presence of key market players
- The market, in particular, is witnessing substantial growth in prothrombin time testing installations, particularly in hospitals and diagnostic laboratories, fueled by innovations in point-of-care testing, laboratory automation, and integration with electronic health records
- The increasing prevalence of cardiovascular and bleeding disorders is further accelerating demand for rapid and accurate coagulation monitoring solutions
U.S. Prothrombin Time Testing Market Insight
The U.S. prothrombin time testing market captured the largest revenue share in 2025 within North America, driven by the rising demand for point-of-care PT devices and advanced laboratory systems. Growing awareness of cardiovascular health, the aging population, and increased government support for healthcare diagnostics are fueling market growth. The integration of PT testing with digital health platforms and mobile reporting solutions is further enhancing operational efficiency in hospitals and clinics.
Europe Prothrombin Time Testing Market Insight
The Europe prothrombin time testing market is projected to expand at a substantial CAGR throughout the forecast period, primarily driven by increasing awareness of coagulation disorders, rising prevalence of cardiovascular diseases, and supportive healthcare infrastructure. Adoption of modern PT testing instruments in hospitals, clinical laboratories, and research centers is increasing across major European countries. Technological advancements, such as automated PT testing and data connectivity, are enhancing efficiency and accuracy in diagnostics.
U.K. Prothrombin Time Testing Market Insight
The U.K. prothrombin time testing market is anticipated to grow at a noteworthy CAGR during the forecast period, supported by government initiatives for early detection of cardiovascular diseases and coagulation disorders. Hospitals and diagnostic labs are increasingly implementing advanced PT testing devices to ensure faster turnaround times and accurate monitoring, while healthcare professionals emphasize preventive care and risk management.
Germany Prothrombin Time Testing Market Insight
The Germany prothrombin time testing market is expected to expand at a considerable CAGR during the forecast period, fueled by growing patient awareness regarding cardiovascular health, rising adoption of automated PT systems, and increasing investments in modern laboratory facilities. Hospitals and clinics are focusing on faster and more accurate coagulation monitoring, with technological innovation and sustainability being key drivers.
Asia-Pacific Prothrombin Time Testing Market Insight
The Asia-Pacific prothrombin time testing market is poised to grow at the fastest CAGR during the forecast period of 2026 to 2033, driven by expanding healthcare access, rising government investments in diagnostics, and growing awareness of cardiovascular and bleeding disorders in countries such as China, India, and Japan. Increasing hospital infrastructure, modernization of clinical laboratories, and adoption of cost-effective PT testing solutions are boosting market penetration. Furthermore, the rising middle-class population and growing focus on preventive healthcare are contributing to market expansion.
Japan Prothrombin Time Testing Market Insight
The Japan prothrombin time testing market is witnessing steady growth due to the country’s advanced healthcare ecosystem, high adoption of modern laboratory technologies, and an aging population with increased susceptibility to cardiovascular and coagulation disorders. Hospitals and diagnostic centers are implementing automated PT testing systems to enhance accuracy and efficiency, while government programs supporting preventive healthcare continue to drive demand.
China Prothrombin Time Testing Market Insight
The China prothrombin time testing market accounted for the largest market revenue share in Asia-Pacific in 2025, supported by government initiatives for healthcare modernization, rapid urbanization, and the rising prevalence of cardiovascular and bleeding disorders. Hospitals, clinics, and diagnostic laboratories are increasingly adopting PT testing systems to ensure accurate coagulation monitoring, while local manufacturers and affordable diagnostic solutions are expanding accessibility to a broader patient base.
Prothrombin Time Testing Market Share
The Prothrombin Time Testing industry is primarily led by well-established companies, including:
• Roche Diagnostics (Switzerland)
• Siemens Healthineers (Germany)
• Abbott (U.S.)
• Helena Laboratories (U.S.)
• Stago Group (France)
• Instrumentation Laboratory (U.S.)
• Ortho Clinical Diagnostics (U.S.)
• Werfen Group (Spain)
• Thermo Fisher Scientific (U.S.)
• BioMedomics (U.S.)
Latest Developments in Global Prothrombin Time Testing Market
- In July 2023, iLine Microsystems S.L. received clearance for its microINR System in the U.S., a capillary blood finger‑stick device with Bluetooth connectivity. This system allows patients on anticoagulants to easily monitor PT/INR levels outside traditional laboratory settings, enabling greater convenience and timely management of anticoagulation therapy
- In July 2023, ARKRAY USA, Inc., in collaboration with CoaguSense, Inc., launched a new PT/INR monitoring system aimed at improving operational efficiency and connectivity for anticoagulation care. The device enhances both clinical workflow and patient self-testing capabilities, offering accurate and rapid results
- In May 2024, Universal Biosensors Inc. introduced the Xprecia Prime 4U coagulation analyzer, which supports patient self-monitoring of PT/INR at home. The system integrates with telehealth platforms, enabling healthcare providers to remotely track anticoagulation therapy and make timely adjustment
- In August 2024, Universal Biosensors Inc. finalized a revised distribution agreement in India for its Xprecia Prime PT/INR device, allowing for faster regional rollout. This expansion helps increase access to rapid PT/INR testing in emerging markets, supporting early intervention and better patient management
SKU-
Get online access to the report on the World's First Market Intelligence Cloud
- Interactive Data Analysis Dashboard
- Company Analysis Dashboard for high growth potential opportunities
- Research Analyst Access for customization & queries
- Competitor Analysis with Interactive dashboard
- Latest News, Updates & Trend analysis
- Harness the Power of Benchmark Analysis for Comprehensive Competitor Tracking
Research Methodology
Data collection and base year analysis are done using data collection modules with large sample sizes. The stage includes obtaining market information or related data through various sources and strategies. It includes examining and planning all the data acquired from the past in advance. It likewise envelops the examination of information inconsistencies seen across different information sources. The market data is analysed and estimated using market statistical and coherent models. Also, market share analysis and key trend analysis are the major success factors in the market report. To know more, please request an analyst call or drop down your inquiry.
The key research methodology used by DBMR research team is data triangulation which involves data mining, analysis of the impact of data variables on the market and primary (industry expert) validation. Data models include Vendor Positioning Grid, Market Time Line Analysis, Market Overview and Guide, Company Positioning Grid, Patent Analysis, Pricing Analysis, Company Market Share Analysis, Standards of Measurement, Global versus Regional and Vendor Share Analysis. To know more about the research methodology, drop in an inquiry to speak to our industry experts.
Customization Available
Data Bridge Market Research is a leader in advanced formative research. We take pride in servicing our existing and new customers with data and analysis that match and suits their goal. The report can be customized to include price trend analysis of target brands understanding the market for additional countries (ask for the list of countries), clinical trial results data, literature review, refurbished market and product base analysis. Market analysis of target competitors can be analyzed from technology-based analysis to market portfolio strategies. We can add as many competitors that you require data about in the format and data style you are looking for. Our team of analysts can also provide you data in crude raw excel files pivot tables (Fact book) or can assist you in creating presentations from the data sets available in the report.