Global Rna Editing Therapeutics Market
Market Size in USD Billion
CAGR :
% 
 USD
1.14 Billion
USD
6.37 Billion
2025
2033
USD
1.14 Billion
USD
6.37 Billion
2025
2033
| 2026 - 2033 | |
| USD 1.14 Billion | |
| USD 6.37 Billion | |
|
|
|
|
RNA Editing Therapeutics Market Size
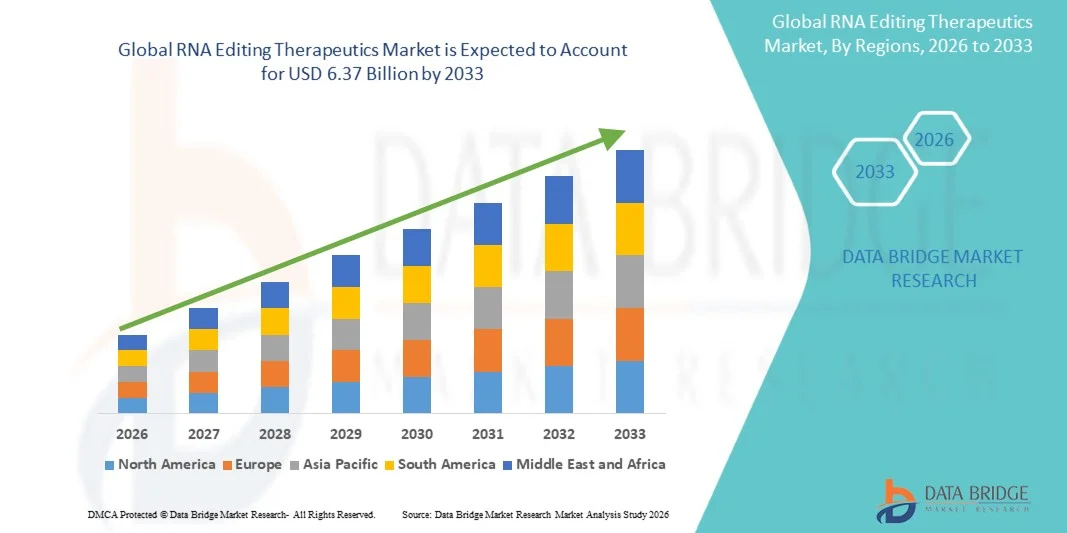
- The global RNA editing therapeutics market size was valued at USD 1.14 billion in 2025 and is expected to reach USD 6.37 billion by 2033, at a CAGR of 24.00% during the forecast period
- The market growth is largely fueled by rapid advancements in molecular biology, gene editing technologies, and increasing research into RNA-level interventions, along with growing prevalence of genetic and chronic diseases, leading to rising demand for innovative RNA Editing Therapeutics solutions
- Furthermore, increasing investments in biotechnology research, expanding clinical trials, and growing focus on precision medicine are establishing RNA-based therapies as a promising next-generation treatment approach. These converging factors are accelerating the uptake of RNA Editing Therapeutics solutions, thereby significantly boosting the industry's growth
RNA Editing Therapeutics Market Analysis
- RNA Editing Therapeutics, an advanced branch of precision medicine focused on modifying RNA sequences to correct genetic errors without altering DNA, is gaining significant traction due to rapid advancements in gene editing technologies and increasing research into targeted and reversible therapies for complex diseases
- The escalating demand for RNA Editing Therapeutics is primarily fueled by rising prevalence of genetic and chronic diseases, increasing investments in biotechnology and pharmaceutical R&D, and growing focus on precision and personalized medicine approaches
- North America dominated the RNA editing therapeutics market with the largest revenue share of approximately 44.5% in 2025, characterized by strong biotech infrastructure, high R&D funding, and early adoption of advanced gene and RNA-based therapies, with the U.S. leading in clinical trials and innovation in RNA editing platforms
- Asia-Pacific is expected to be the fastest growing region in the RNA editing therapeutics market during the forecast period due to expanding biotechnology research capabilities, increasing government support for genomics, and rising healthcare investments across emerging economies
- The CRISPR-based RNA editing segment dominated the largest market revenue share of 52.6% in 2025, driven by its high precision, programmability, and expanding applications in targeted gene regulation and transcript-level modifications
Report Scope and RNA Editing Therapeutics Market Segmentation
|
Attributes |
RNA Editing Therapeutics Key Market Insights |
|
Segments Covered |
|
|
Countries Covered |
North America
Europe
Asia-Pacific
Middle East and Africa
South America
|
|
Key Market Players |
|
|
Market Opportunities |
|
|
Value Added Data Infosets |
In addition to the insights on market scenarios such as market value, growth rate, segmentation, geographical coverage, and major players, the market reports curated by the Data Bridge Market Research also include in-depth expert analysis, patient epidemiology, pipeline analysis, pricing analysis, and regulatory framework. |
RNA Editing Therapeutics Market Trends
“Advancements in Precision Medicine and RNA-Based Therapeutic Innovation”
- A significant and accelerating trend in the global RNA Editing Therapeutics market is the growing advancement of precision medicine and RNA-based therapeutic technologies. These innovations are transforming the treatment landscape by enabling highly targeted and reversible modifications at the RNA level
- For instance, emerging RNA editing approaches such as ADAR-mediated editing and CRISPR-associated RNA targeting are being studied to correct disease-causing mutations without permanently altering the DNA sequence
- Increasing research activity in transcriptomics and molecular biology is expanding the understanding of RNA function in genetic disorders, cancer, and rare diseases
- Furthermore, pharmaceutical and biotechnology companies are investing heavily in RNA platforms to develop next-generation therapeutics with improved safety and specificity compared to traditional gene-editing approaches
- Growing collaboration between academic research institutes and biotech firms is accelerating clinical trials and pipeline development for RNA-based therapies across multiple disease areas
- This trend toward highly precise, programmable, and adaptable therapeutic approaches is significantly reshaping the global RNA Editing Therapeutics market
RNA Editing Therapeutics Market Dynamics
Driver
“Rising Prevalence of Genetic Disorders and Advancements in Genomic Research”
- The increasing global prevalence of genetic disorders, rare diseases, and chronic conditions is a major driver for the growth of the RNA Editing Therapeutics market
- For instance, conditions caused by single-gene mutations, such as certain metabolic, neurological, and hematological disorders, are creating strong demand for innovative treatment approaches like RNA editing
- Rapid advancements in genomic sequencing and molecular diagnostics are enabling earlier and more accurate identification of disease-causing mutations, supporting targeted therapeutic development
- In addition, rising investment in biotechnology research and increasing funding for precision medicine initiatives are accelerating innovation in RNA-based therapies
- Expanding clinical research programs and supportive regulatory frameworks for advanced biologics are further encouraging pharmaceutical companies to develop RNA editing solutions
- Growing awareness of personalized medicine among healthcare providers and patients is also contributing to increased adoption of next-generation therapeutic approaches globally
Restraint/Challenge
“High Development Costs and Technical Complexity of RNA Editing Technologies”
- The high cost and technical complexity associated with RNA editing therapeutic development remain key challenges for market growth, as these therapies require advanced infrastructure, specialized expertise, and extensive research investment
- A significant challenge is the difficulty in achieving precise, efficient, and safe RNA editing in vivo, as off-target effects and delivery limitations continue to be major scientific hurdles
- For instance, many RNA-based therapies are still in early-stage clinical trials due to challenges in ensuring stable and targeted delivery to specific tissues
- In addition, the lack of standardized manufacturing processes and regulatory frameworks for RNA editing technologies can slow down commercialization and approval timelines
- The long development cycles and high failure rates in clinical trials also increase financial risk for biotechnology and pharmaceutical companies
- Limited scalability of production and challenges in maintaining therapeutic stability further restrict widespread adoption
- Addressing these challenges through technological innovation, improved delivery systems, increased funding, and stronger global collaboration will be essential for sustained growth in the RNA Editing Therapeutics market
RNA Editing Therapeutics Market Scope
The market is segmented on the basis of technology and application.
• By Technology
On the basis of technology, the RNA Editing Therapeutics market is segmented into CRISPR-based RNA editing, ADAR-mediated RNA editing, and others. The CRISPR-based RNA editing segment dominated the largest market revenue share of 52.6% in 2025, driven by its high precision, programmability, and expanding applications in targeted gene regulation and transcript-level modifications. This technology enables reversible and flexible editing without permanently altering DNA, making it highly attractive for therapeutic development. Increasing investments in genomic research and biotechnology innovation are further strengthening adoption. Pharmaceutical companies are actively developing CRISPR-based RNA editing pipelines for rare and chronic diseases. Rising clinical trial activity and regulatory support for advanced gene-editing platforms also contribute to segment growth. In addition, improved delivery systems such as lipid nanoparticles are enhancing therapeutic efficiency. Growing demand for personalized medicine and precision therapeutics is accelerating market expansion. Strong research funding from government and private institutions further supports dominance. The segment continues to benefit from rapid technological advancements and expanding therapeutic indications.
The ADAR-mediated RNA editing segment is expected to witness the fastest CAGR of 13.8% from 2026 to 2033, driven by its endogenous editing mechanism and lower risk of genomic instability. ADAR-based systems utilize natural cellular enzymes, making them a safer and potentially more biocompatible approach for RNA modification. Increasing research into neurological and genetic disorders is expanding its application scope. Advances in RNA-guided ADAR recruitment technologies are improving specificity and efficiency. Rising interest in transient and reversible gene therapies is also boosting adoption. Pharmaceutical and biotech collaborations are accelerating clinical development. In addition, growing focus on non-permanent therapeutic interventions is driving demand. Expanding pipeline studies in rare disease treatment is further supporting growth. As delivery technologies improve, ADAR-mediated editing is expected to gain strong momentum globally.
• By Application
On the basis of application, the RNA Editing Therapeutics market is segmented into genetic disorders, oncology, neurological disorders, infectious diseases, and others. The genetic disorders segment accounted for the largest market revenue share of 41.9% in 2025, driven by the high prevalence of inherited diseases and the strong potential of RNA editing in correcting disease-causing mutations at the transcript level. Conditions such as cystic fibrosis, sickle cell disease, and muscular dystrophies are key targets for RNA-based therapeutic interventions. Increasing adoption of precision medicine and advancements in genomic sequencing are supporting early diagnosis and treatment development. Pharmaceutical companies are heavily investing in rare disease pipelines, further strengthening segment growth. Regulatory incentives for orphan drugs also contribute to market expansion. Growing awareness among healthcare providers and patients about gene-targeted therapies is boosting adoption. Continuous innovation in delivery systems enhances therapeutic efficiency. Rising clinical trial activity is accelerating progress in this segment. Strong unmet medical needs ensure sustained demand for RNA editing solutions.
The neurological disorders segment is expected to witness the fastest CAGR of 14.6% from 2026 to 2033, driven by the growing burden of conditions such as Alzheimer’s disease, Parkinson’s disease, and amyotrophic lateral sclerosis (ALS). RNA editing offers a promising approach for targeting disease-related protein expression in the central nervous system. Advances in blood-brain barrier (BBB) delivery technologies are significantly improving therapeutic potential. Increasing research funding for neurodegenerative diseases is accelerating innovation. The rising aging population globally further contributes to segment growth. Pharmaceutical companies are increasingly focusing on CNS-targeted RNA therapies. Expanding clinical trials and preclinical studies are validating efficacy in neurological applications. Growing demand for disease-modifying therapies is also boosting adoption. As delivery systems improve, neurological applications are expected to dominate future innovation pipelines.
RNA Editing Therapeutics Market Regional Analysis
- North America dominated the RNA editing therapeutics market with the largest revenue share of approximately 44.5% in 2025, characterized by strong biotechnology infrastructure, high R&D funding, and early adoption of advanced gene and RNA-based therapies
- The region benefits from a robust ecosystem of pharmaceutical companies, research institutes, and biotechnology startups actively engaged in RNA editing innovation
- The presence of well-established regulatory pathways for advanced therapeutics, along with significant investments in genomic medicine, further supports market growth. In addition, increasing collaboration between academia and industry is accelerating the development and clinical translation of RNA editing technologies across North America
U.S. RNA Editing Therapeutics Market Insight
The U.S. RNA editing therapeutics market captured the largest revenue share within North America in 2025, driven by its leadership in biotechnology innovation and strong clinical research activity. The country hosts a high concentration of biotech firms and academic institutions pioneering RNA editing platforms and next-generation gene therapies. Substantial funding from both public and private sectors is accelerating clinical trials and therapeutic development. Furthermore, the U.S. remains at the forefront of regulatory approvals and early adoption of advanced molecular medicine approaches.
Europe RNA Editing Therapeutics Market Insight
The Europe RNA editing therapeutics market is projected to expand at a substantial CAGR during the forecast period, supported by increasing investments in biotechnology research and strong regulatory frameworks for advanced therapies. Government-funded genomics programs and cross-border research collaborations are strengthening innovation in RNA-based treatments. In addition, growing focus on precision medicine is enhancing the adoption of novel therapeutic approaches across the region.
U.K. RNA Editing Therapeutics Market Insight
The U.K. RNA editing therapeutics market is anticipated to grow at a noteworthy CAGR, driven by strong academic research capabilities and expanding biotech innovation hubs. Public funding and private investment in genomic medicine are supporting early-stage research and clinical translation. Moreover, collaborations between universities, biotech companies, and healthcare institutions are accelerating advancements in RNA editing technologies.
Germany RNA Editing Therapeutics Market Insight
The Germany RNA editing therapeutics market is expected to expand at a considerable CAGR, fueled by the country’s strong pharmaceutical sector and emphasis on biomedical innovation. Germany’s advanced research infrastructure and focus on precision medicine are enabling rapid progress in gene and RNA-based therapies. Increasing participation in EU-wide genomics initiatives is further strengthening the market.
Asia-Pacific RNA Editing Therapeutics Market Insight
The Asia-Pacific RNA editing therapeutics market is poised to grow at the fastest CAGR during the forecast period, driven by expanding biotechnology research capabilities, increasing government support for genomics, and rising healthcare investments across emerging economies. The region’s growing focus on precision medicine and improving access to advanced research infrastructure are fostering rapid market expansion. In addition, rising collaboration with global biotech firms is accelerating technology transfer and innovation.
Japan RNA Editing Therapeutics Market Insight
The Japan RNA editing therapeutics market is gaining momentum due to its strong biotechnology sector and advanced research environment. Increasing focus on regenerative medicine and genomic research is supporting the development of RNA-based therapies. In addition, Japan’s aging population is driving demand for innovative treatments targeting chronic and genetic diseases, further supporting market growth.
China RNA Editing Therapeutics Market Insight
The China RNA editing therapeutics market accounted for the largest revenue share in Asia-Pacific in 2025, attributed to rapid expansion of biotechnology research, strong government support for genomics, and increasing investment in advanced therapeutic development. The country’s growing biotech industry and expanding clinical research capabilities are accelerating innovation in RNA editing platforms. Furthermore, partnerships between domestic firms and global biotech companies are strengthening the development pipeline and market adoption.
RNA Editing Therapeutics Market Share
The RNA Editing Therapeutics industry is primarily led by well-established companies, including:
- Pfizer Inc. (U.S.)
- Novartis AG (Switzerland)
- F. Hoffmann-La Roche Ltd. (Switzerland)
- Sanofi S.A. (France)
- AstraZeneca plc (U.K.)
- GlaxoSmithKline plc (U.K.)
- Bayer AG (Germany)
- Merck & Co., Inc. (U.S.)
- Eli Lilly and Company (U.S.)
- Takeda Pharmaceutical Company Limited (Japan)
- Moderna, Inc. (U.S.)
- BioNTech SE (Germany)
- Alnylam Pharmaceuticals, Inc. (U.S.)
- Ionis Pharmaceuticals, Inc. (U.S.)
- Editas Medicine, Inc. (U.S.)
- Intellia Therapeutics, Inc. (U.S.)
- CRISPR Therapeutics AG (Switzerland)
- Beam Therapeutics Inc. (U.S.)
- Sangamo Therapeutics, Inc. (U.S.)
- Regeneron Pharmaceuticals, Inc. (U.S.)
Latest Developments in Global RNA Editing Therapeutics Market
- In June 2021, Wave Life Sciences reported early clinical proof-of-concept results for RNA editing therapy WVE-006, demonstrating for the first time that ADAR-mediated RNA editing could restore functional protein expression in humans with alpha-1 antitrypsin deficiency (AATD). This marked one of the earliest clinical validations of RNA editing therapeutics in a genetic disease setting and significantly accelerated industry interest in programmable RNA modification approaches
- In January 2023, researchers from Shape Therapeutics and academic collaborators published a major scientific review in Molecular Therapy describing the advancement of ADAR-based programmable RNA editing platforms, highlighting their ability to correct disease-causing mutations at the RNA level without altering DNA. The publication emphasized RNA editing as a reversible and safer alternative to genome editing for genetic diseases and inflammatory disorders
- In June 2023, Wave Life Sciences advanced its RNA editing pipeline into higher-dose clinical evaluation, building on earlier human proof-of-concept data and expanding investigations in liver-targeted AATD therapy (WVE-006/WVE-007 programs). This represented a key step in moving RNA editing from early feasibility to dose-optimization clinical development
- In April 2024, Prime Medicine announced FDA IND clearance for its prime editing–based RNA/DNA editing therapeutic PM359 for chronic granulomatous disease (CGD), enabling initiation of a first-in-human clinical trial. Although primarily DNA-focused, this milestone is considered highly relevant to RNA editing therapeutics due to shared programmable editing architecture and delivery systems used in RNA-level correction strategies
- In June 2024, Roche entered into a strategic partnership with Ascidian Therapeutics to develop RNA exon-editing therapies, a next-generation RNA modification approach that enables replacement of faulty RNA segments. This collaboration strengthened industry investment in RNA editing technologies for neurological and rare genetic diseases
- In September 2024, Wave Life Sciences’ RNA editing program faced a major clinical setback when its AATD RNA editing candidate failed to demonstrate sufficient efficacy in later-stage human studies, leading to program restructuring and highlighting the translational challenges of ADAR-based editing technologies
- In May 2025, Prime Medicine reported early clinical success of its prime editing platform in patients with chronic granulomatous disease, showing restoration of immune cell function in treated patients. This was a landmark demonstration of durable therapeutic gene correction, reinforcing confidence in programmable editing modalities relevant to RNA-targeted therapeutic development
- In October 2025, a major peer-reviewed review in Frontiers in Genetics described the current state of RNA therapeutics, highlighting emerging RNA editing technologies such as CRISPR-Cas13 and ADAR-based systems, along with improvements in lipid nanoparticle delivery systems enabling more precise and stable RNA modification in vivo
SKU-
Get online access to the report on the World's First Market Intelligence Cloud
- Interactive Data Analysis Dashboard
- Company Analysis Dashboard for high growth potential opportunities
- Research Analyst Access for customization & queries
- Competitor Analysis with Interactive dashboard
- Latest News, Updates & Trend analysis
- Harness the Power of Benchmark Analysis for Comprehensive Competitor Tracking
Research Methodology
Data collection and base year analysis are done using data collection modules with large sample sizes. The stage includes obtaining market information or related data through various sources and strategies. It includes examining and planning all the data acquired from the past in advance. It likewise envelops the examination of information inconsistencies seen across different information sources. The market data is analysed and estimated using market statistical and coherent models. Also, market share analysis and key trend analysis are the major success factors in the market report. To know more, please request an analyst call or drop down your inquiry.
The key research methodology used by DBMR research team is data triangulation which involves data mining, analysis of the impact of data variables on the market and primary (industry expert) validation. Data models include Vendor Positioning Grid, Market Time Line Analysis, Market Overview and Guide, Company Positioning Grid, Patent Analysis, Pricing Analysis, Company Market Share Analysis, Standards of Measurement, Global versus Regional and Vendor Share Analysis. To know more about the research methodology, drop in an inquiry to speak to our industry experts.
Customization Available
Data Bridge Market Research is a leader in advanced formative research. We take pride in servicing our existing and new customers with data and analysis that match and suits their goal. The report can be customized to include price trend analysis of target brands understanding the market for additional countries (ask for the list of countries), clinical trial results data, literature review, refurbished market and product base analysis. Market analysis of target competitors can be analyzed from technology-based analysis to market portfolio strategies. We can add as many competitors that you require data about in the format and data style you are looking for. Our team of analysts can also provide you data in crude raw excel files pivot tables (Fact book) or can assist you in creating presentations from the data sets available in the report.