Global Smart Pills Market
Market Size in USD Billion
CAGR :
% 
 USD
2.31 Billion
USD
7.95 Billion
2025
2033
USD
2.31 Billion
USD
7.95 Billion
2025
2033
| 2026 –2033 | |
| USD 2.31 Billion | |
| USD 7.95 Billion | |
|
|
|
|
Smart Pills Market Size
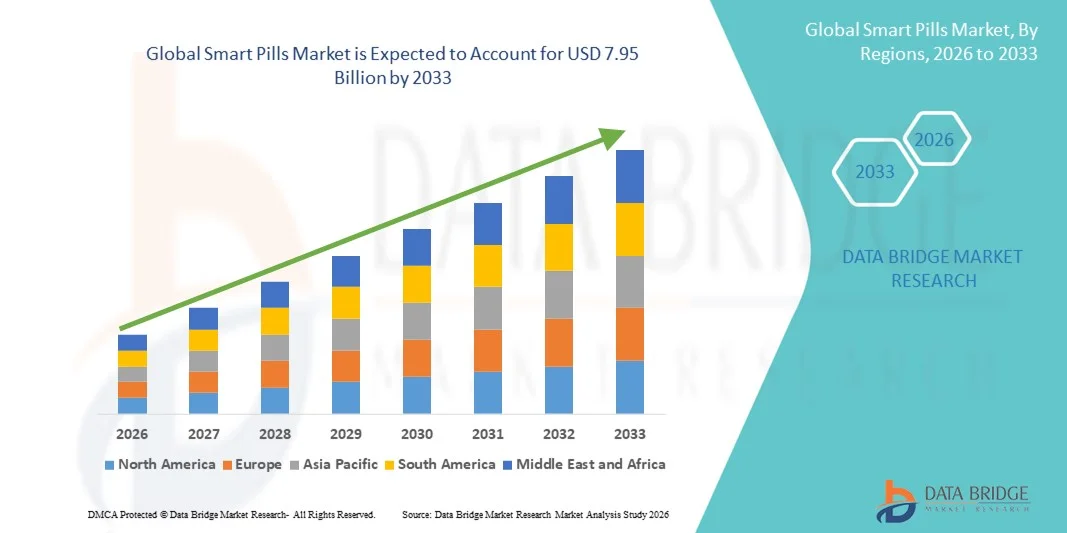
- The global smart pills market size was valued at USD 2.31 billion in 2025 and is expected to reach USD 7.95 billion by 2033, at a CAGR of 16.71% during the forecast period
- The market growth is largely driven by the increasing prevalence of gastrointestinal disorders, growing demand for minimally invasive diagnostic procedures, and rapid advancements in capsule endoscopy and ingestible sensor technologies across healthcare systems worldwide
- Furthermore, rising adoption of digital health solutions, expanding applications in drug delivery and patient monitoring, and strong investments in medical device innovation are positioning smart pills as a transformative solution in modern diagnostics and therapeutics. These converging factors are accelerating the adoption of smart pill technologies, thereby significantly propelling overall market expansion
Smart Pills Market Analysis
- Smart pills, encompassing ingestible medical devices equipped with miniature sensors, cameras, or drug delivery components, are increasingly transforming modern healthcare by enabling real-time gastrointestinal imaging, targeted drug delivery, and remote patient monitoring, offering minimally invasive alternatives to traditional diagnostic and therapeutic procedures across hospitals and specialty clinics
- The rising demand for smart pills is primarily fueled by the growing prevalence of gastrointestinal disorders, increasing preference for non-invasive diagnostic techniques, expanding geriatric population, and the rapid integration of digital health technologies that enhance patient compliance, disease monitoring, and personalized treatment approaches
- North America dominated the smart pills market with the largest revenue share of 38.76% in 2025, supported by advanced healthcare infrastructure, high healthcare expenditure, strong regulatory approvals, and the presence of leading medical device manufacturers, with the U.S. witnessing significant adoption of capsule endoscopy and ingestible sensor technologies driven by innovation and favorable reimbursement frameworks
- Asia-Pacific is expected to be the fastest growing region in the smart pills market during the forecast period due to improving healthcare infrastructure, rising awareness of early disease diagnosis, expanding medical tourism, and increasing investments in advanced medical technologies across emerging economies
- Capsule endoscopy segment dominated the smart pills market with a market share of 47.8% in 2025, driven by its widespread clinical acceptance for small bowel imaging, superior patient comfort compared to conventional endoscopy, and growing utilization in diagnosing Crohn’s disease, obscure gastrointestinal bleeding, and colorectal abnormalities
Report Scope and Smart Pills Market Segmentation
|
Attributes |
Smart Pills Key Market Insights |
|
Segments Covered |
|
|
Countries Covered |
North America
Europe
Asia-Pacific
Middle East and Africa
South America
|
|
Key Market Players |
|
|
Market Opportunities |
|
|
Value Added Data Infosets |
In addition to the insights on market scenarios such as market value, growth rate, segmentation, geographical coverage, and major players, the market reports curated by the Data Bridge Market Research also include in-depth expert analysis, patient epidemiology, pipeline analysis, pricing analysis, and regulatory framework |
Smart Pills Market Trends
Rising Adoption of AI-Enabled Capsule Endoscopy and Digital Health Integration
- A significant and accelerating trend in the global smart pills market is the integration of artificial intelligence (AI) and advanced digital health platforms with capsule endoscopy and ingestible sensor technologies, substantially enhancing diagnostic accuracy, workflow efficiency, and patient-centric care delivery across healthcare systems
- For instance, AI-powered image analysis software integrated with capsule endoscopy systems enables faster detection of gastrointestinal abnormalities, while digital tracking platforms allow physicians to remotely review real-time patient data captured by ingestible sensors
- AI integration in smart pills enables automated lesion detection, improved image classification, and enhanced data interpretation accuracy, reducing physician workload and minimizing diagnostic errors. For instance, certain capsule endoscopy platforms leverage AI algorithms to flag potential bleeding sites or inflammatory lesions and generate prioritized review frames for clinicians. Furthermore, digital connectivity allows seamless data transmission to secure cloud-based systems for comprehensive analysis
- The seamless integration of smart pills with hospital information systems and telemedicine platforms facilitates centralized data management and coordinated care pathways. Through unified dashboards, healthcare providers can monitor diagnostic findings alongside patient history, laboratory data, and treatment plans, creating a more synchronized and data-driven healthcare environment
- This trend toward more intelligent, data-driven, and minimally invasive diagnostic solutions is fundamentally reshaping expectations for gastrointestinal and chronic disease management. Consequently, companies such as Medtronic and Olympus Corporation are advancing AI-enabled capsule endoscopy platforms designed to enhance detection rates and streamline clinical workflows
- The demand for smart pills offering AI-enhanced diagnostics and seamless digital integration is expanding rapidly across hospitals, specialty clinics, and ambulatory care centers, as healthcare systems increasingly prioritize precision medicine, early disease detection, and operational efficiency
- Growing collaborations between medical device manufacturers and digital health technology firms are accelerating innovation cycles, fostering the development of multifunctional smart pills capable of combining imaging, sensing, and drug delivery capabilities within a single ingestible platform
Smart Pills Market Dynamics
Driver
Growing Demand Due to Rising Gastrointestinal Disorders and Preference for Minimally Invasive Diagnostics
- The increasing prevalence of gastrointestinal disorders and chronic diseases, coupled with the growing preference for minimally invasive diagnostic procedures, is a significant driver for the expanding demand for smart pills worldwide
- For instance, in recent years, several leading medical device manufacturers have expanded their capsule endoscopy portfolios and invested in next-generation ingestible sensor technologies to enhance diagnostic precision and patient comfort. Such strategic advancements by key companies are expected to drive the smart pills market growth during the forecast period
- As patients and healthcare providers seek safer and more comfortable diagnostic alternatives, smart pills offer non-invasive visualization of the gastrointestinal tract, reduced need for sedation, and improved patient compliance compared to conventional endoscopy procedures
- Furthermore, the increasing adoption of digital health solutions and remote monitoring technologies is positioning smart pills as integral components of modern healthcare ecosystems, enabling real-time data collection and improved clinical decision-making
- The ability to perform comprehensive small bowel imaging, monitor medication adherence through ingestible sensors, and support personalized treatment strategies are key factors propelling adoption in both developed and emerging healthcare markets. The expansion of reimbursement frameworks and growing awareness regarding early disease detection further contribute to market growth
- Rising healthcare expenditure in emerging economies and government initiatives promoting early disease screening programs are further strengthening demand for advanced diagnostic technologies such as smart pills
- The expanding geriatric population, which is more susceptible to gastrointestinal and chronic disorders, is also contributing significantly to the sustained adoption of capsule-based diagnostic and monitoring solutions
Restraint/Challenge
High Device Costs and Regulatory Approval Complexities
- Concerns surrounding high development costs, stringent regulatory requirements, and data privacy issues associated with ingestible medical devices pose significant challenges to broader market penetration. As smart pills function as advanced medical devices, they must comply with rigorous clinical validation and safety standards, increasing time-to-market and overall expenses
- For instance, regulatory approvals for capsule endoscopy systems and ingestible sensors often require extensive clinical trials and post-market surveillance, which can delay commercialization timelines and elevate operational costs for manufacturers
- Addressing these regulatory and financial barriers through streamlined approval pathways, strategic partnerships, and scalable manufacturing processes is essential for expanding access. Companies such as CapsoVision and Medtronic emphasize clinical validation studies and compliance with international medical device standards to strengthen market credibility. In addition, the relatively high procedural and device costs compared to conventional diagnostic tools can limit adoption in price-sensitive healthcare systems, particularly in developing regions
- While technological advancements are gradually improving affordability and efficiency, the perception of smart pills as premium diagnostic solutions may still restrict widespread utilization, especially in facilities with constrained healthcare budgets
- Overcoming these challenges through cost optimization strategies, stronger reimbursement support, enhanced regulatory collaboration, and increased awareness among healthcare providers will be critical for sustaining long-term market growth
- Limited availability of skilled professionals trained in interpreting capsule endoscopy data and managing digital diagnostic platforms can further slow adoption in certain healthcare settings
- Potential concerns regarding data security, patient privacy, and interoperability between smart pill systems and existing hospital IT infrastructure may also hinder seamless large-scale implementation
Smart Pills Market Scope
The market is segmented on the basis of type, disease indication, application, and end user.
- By Type
On the basis of type, the smart pills market is segmented into multiparameter monitoring and single parameter monitoring. The multiparameter monitoring segment dominated the market with the largest revenue share in 2025, driven by its ability to capture multiple physiological and diagnostic parameters simultaneously, including pH levels, temperature, pressure, and imaging data. These advanced capsules provide comprehensive gastrointestinal insights, improving diagnostic precision and reducing the need for multiple procedures. Hospitals and specialty clinics prefer multiparameter smart pills due to their enhanced clinical value and efficiency in complex cases. Their integration with digital health platforms further strengthens their adoption in developed healthcare systems. Continuous technological advancements in sensor miniaturization and wireless transmission capabilities also support segment dominance.
The single parameter monitoring segment is anticipated to witness the fastest growth rate from 2026 to 2033, fueled by increasing demand for cost-effective and targeted diagnostic solutions. These smart pills are particularly useful in specific applications such as pH monitoring or temperature tracking, where focused data collection is sufficient for clinical decision-making. Their relatively lower cost compared to multiparameter devices makes them more accessible in emerging markets and smaller healthcare facilities. Growing adoption in outpatient settings and research applications further contributes to segment expansion. In addition, simplified regulatory pathways for single-function devices may accelerate product launches and commercialization.
- By Disease Indication
On the basis of disease indication, the smart pills market is segmented into occult GI bleeding, Crohn’s disease, small bowel tumors, celiac disease, inherited polyposis syndromes, and mental disorders. The occult GI bleeding segment dominated the market in 2025, primarily due to the widespread use of capsule endoscopy for detecting hidden sources of gastrointestinal bleeding. Smart pills provide non-invasive visualization of the small intestine, which is often inaccessible through traditional endoscopy. Early detection capabilities significantly improve patient outcomes and reduce hospitalization rates. Increasing prevalence of gastrointestinal disorders and anemia-related conditions further strengthens demand in this segment. Strong clinical validation and established reimbursement frameworks also support its leading position.
The Crohn’s disease segment is projected to witness the fastest growth during the forecast period, driven by rising global incidence rates and the need for continuous disease monitoring. Smart pills offer detailed mucosal imaging, enabling physicians to assess inflammation severity and treatment effectiveness. The growing emphasis on personalized medicine and early intervention strategies further accelerates adoption. Technological advancements in AI-based lesion detection enhance diagnostic reliability for inflammatory bowel diseases. Increasing awareness and improved access to advanced diagnostic tools in emerging economies are also contributing to segment growth.
- By Application
On the basis of application, the smart pills market is segmented into capsule endoscopy, drug delivery, and patient monitoring of cancer. The capsule endoscopy segment dominated the market in 2025 with a market share of 47.8% due to its extensive clinical adoption for small bowel imaging and gastrointestinal diagnostics. It offers a minimally invasive alternative to traditional endoscopic procedures, significantly improving patient comfort and compliance. High diagnostic accuracy, coupled with continuous advancements in imaging resolution and battery life, reinforces its widespread usage. Hospitals and diagnostic centers increasingly rely on capsule endoscopy for early detection of tumors, bleeding, and inflammatory diseases. Favorable reimbursement policies in developed regions further support segment dominance.
The drug delivery segment is expected to register the fastest growth rate from 2026 to 2033, driven by expanding research in targeted and controlled therapeutic delivery systems. Smart pills designed for site-specific drug release improve treatment efficacy while minimizing systemic side effects. Growing applications in oncology and chronic disease management create new commercial opportunities. Pharmaceutical companies are increasingly investing in ingestible delivery technologies to enhance patient adherence and therapeutic precision. Continuous innovation in biodegradable materials and sensor-enabled release mechanisms further accelerates segment expansion.
- By End User
On the basis of end user, the smart pills market is segmented into hospitals, diagnostic centers, and others. The hospitals segment dominated the market in 2025, supported by the availability of advanced infrastructure, skilled professionals, and higher patient inflow for complex diagnostic procedures. Hospitals are primary adopters of capsule endoscopy and ingestible sensor technologies due to their capacity for comprehensive patient management. Integration with hospital information systems enables efficient data analysis and multidisciplinary treatment planning. Higher reimbursement coverage and strong investment in advanced medical technologies also contribute to segment leadership.
The diagnostic centers segment is projected to witness the fastest growth during the forecast period, driven by the rising trend toward outpatient diagnostic services and cost-effective care delivery models. Diagnostic centers increasingly adopt smart pill technologies to expand their specialized gastrointestinal testing capabilities. Shorter procedure times and improved patient convenience support higher adoption rates. Growing healthcare decentralization and expansion of specialty clinics in emerging economies further stimulate demand. Strategic partnerships with hospitals and technology providers also enhance service offerings and accelerate segment growth.
Smart Pills Market Regional Analysis
- North America dominated the smart pills market with the largest revenue share of 38.76% in 2025, supported by advanced healthcare infrastructure, high healthcare expenditure, strong regulatory approvals, and the presence of leading medical device manufacturers
- Healthcare providers in the region highly value the diagnostic accuracy, patient comfort, and seamless integration offered by smart pills with digital health platforms and hospital information systems
- This widespread adoption is further supported by favorable reimbursement frameworks, a strong presence of leading medical device manufacturers, and increasing awareness regarding early disease detection, establishing smart pills as a preferred solution for gastrointestinal diagnostics and patient monitoring across hospitals and specialty clinics
U.S. Smart Pills Market Insight
The U.S. smart pills market captured the largest revenue share in 2025 within North America, fueled by advanced healthcare infrastructure and rapid adoption of minimally invasive diagnostic technologies. Healthcare providers are increasingly prioritizing improved gastrointestinal diagnostics and patient compliance through capsule endoscopy and ingestible sensor systems. The growing prevalence of Crohn’s disease, occult GI bleeding, and colorectal disorders further propels market demand. Moreover, strong reimbursement frameworks and continuous technological advancements in AI-enabled imaging platforms are significantly contributing to the market's expansion.
Europe Smart Pills Market Insight
The Europe smart pills market is projected to expand at a substantial CAGR throughout the forecast period, primarily driven by rising awareness regarding early disease detection and stringent medical device regulations ensuring high-quality standards. The increase in gastrointestinal disorder cases, coupled with demand for patient-friendly diagnostic alternatives, is fostering the adoption of smart pills. European healthcare systems are also drawn to the clinical efficiency and improved diagnostic yield these technologies offer. The region is experiencing steady growth across hospitals and specialty diagnostic centers, with smart pills increasingly incorporated into both public and private healthcare facilities.
U.K. Smart Pills Market Insight
The U.K. smart pills market is anticipated to grow at a noteworthy CAGR during the forecast period, driven by the rising burden of inflammatory bowel diseases and growing emphasis on non-invasive diagnostic methods. In addition, increasing focus on reducing hospital stays and enhancing outpatient care is encouraging healthcare providers to adopt capsule-based diagnostic solutions. The UK’s strong healthcare research ecosystem, alongside supportive digital health initiatives, is expected to continue stimulating market growth.
Germany Smart Pills Market Insight
The Germany smart pills market is expected to expand at a considerable CAGR during the forecast period, fueled by increasing awareness of advanced diagnostic technologies and demand for precision-driven healthcare solutions. Germany’s well-established healthcare infrastructure, combined with its emphasis on medical innovation and regulatory compliance, promotes the adoption of smart pills, particularly in specialized gastroenterology centers. The integration of capsule endoscopy systems with digital health records is also becoming increasingly prevalent, aligning with the country’s focus on secure, data-driven medical practices.
Asia-Pacific Smart Pills Market Insight
The Asia-Pacific smart pills market is poised to grow at the fastest CAGR during the forecast period of 2026 to 2033, driven by improving healthcare infrastructure, rising disposable incomes, and growing awareness regarding early diagnosis in countries such as China, Japan, and India. The region's expanding patient population and increasing investments in advanced medical technologies are accelerating adoption. Furthermore, as APAC strengthens its position in medical device manufacturing and healthcare digitization initiatives, accessibility and affordability of smart pill technologies are expanding to a broader patient base.
Japan Smart Pills Market Insight
The Japan smart pills market is gaining momentum due to the country’s advanced healthcare system, aging population, and strong demand for precision diagnostics. The Japanese healthcare sector places significant emphasis on early disease detection, and adoption of capsule endoscopy is driven by the increasing number of elderly patients susceptible to gastrointestinal conditions. The integration of smart pills with hospital digital systems and AI-based diagnostic tools is fueling growth. Moreover, Japan’s focus on technological innovation is likely to spur further demand for highly accurate and patient-friendly diagnostic solutions.
India Smart Pills Market Insight
The India smart pills market accounted for a notable revenue share in Asia Pacific in 2025, attributed to the country’s expanding healthcare infrastructure, rising middle-class population, and increasing awareness of advanced diagnostic options. India stands as one of the rapidly growing markets for minimally invasive medical technologies, with smart pills gaining popularity in tertiary hospitals and specialized diagnostic centers. Government initiatives promoting digital health and early disease screening, alongside increasing private sector investments, are key factors propelling market expansion in India.
Smart Pills Market Share
The Smart Pills industry is primarily led by well-established companies, including:
- Medtronic (Ireland)
- CapsoVision, Inc. (U.S.)
- Check-Cap Ltd. (Israel)
- IntroMedic Co., Ltd. (South Korea)
- JINSHAN Science & Technology (Group) Co., Ltd. (China)
- Olympus Corporation (Japan)
- Koninklijke Philips N.V. (Netherlands)
- etectRx, Inc. (U.S.)
- Atmo Biosciences Pty Ltd (Australia)
- BodyCAP (France)
- PENTAX Medical (Japan)
- Motilis Medica SA (Switzerland)
- Rani Therapeutics, LLC (U.S.)
- Microchips Biotech, Inc. (U.S.)
- Medimetrics Personalized Drug Delivery B.V. (Netherlands)
- RF Co., Ltd. (China)
- HQ Inc. (U.S.)
- BioCam (China)
- Ovesco Endoscopy AG (Germany)
- AnX Robotica Corporation (U.S.)
What are the Recent Developments in Global Smart Pills Market?
- In December 2025, scientists from the Indian Institute of Technology (IIT) Delhi and All India Institute of Medical Sciences (AIIMS) New Delhi successfully tested a swallowable “smart pill” capable of sampling gut microbiome directly from within the intestine, offering a novel, non-invasive method to study and diagnose digestive diseases with improved accuracy
- In January 2025, CapsoVision’s CapsoCam Plus® capsule endoscopy system received FDA clearance for pediatric use, allowing non-invasive 360° panoramic small bowel imaging in children aged two years and older a notable expansion of clinical applications for capsule endoscopy diagnostics
- In January 2025, FDA granted CapsoVision’s CapsoCam Plus® system clearance to continue remote ingestion under medical supervision, enabling patients to swallow the capsule at home without clinic visits and significantly expanding telehealth-enabled diagnostic workflows
- In December 2024, Medtronic successfully reported the first-ever patient procedure ingestion of its next-generation PillCam™ Genius SB capsule endoscopy kit enabling advanced small bowel visualization that can be performed in a hospital, clinic, or even remotely during a telehealth appointment, enhancing accessibility and patient convenience
- In June 2024, researchers at the University of Southern California developed an AI-enabled ingestible smart pill system coupled with a wearable coil that tracks the device’s movement and real-time gut biomarkers, enabling remote gastrointestinal health monitoring through a smartphone interface, a significant step toward home-based diagnostics
SKU-
Get online access to the report on the World's First Market Intelligence Cloud
- Interactive Data Analysis Dashboard
- Company Analysis Dashboard for high growth potential opportunities
- Research Analyst Access for customization & queries
- Competitor Analysis with Interactive dashboard
- Latest News, Updates & Trend analysis
- Harness the Power of Benchmark Analysis for Comprehensive Competitor Tracking
Research Methodology
Data collection and base year analysis are done using data collection modules with large sample sizes. The stage includes obtaining market information or related data through various sources and strategies. It includes examining and planning all the data acquired from the past in advance. It likewise envelops the examination of information inconsistencies seen across different information sources. The market data is analysed and estimated using market statistical and coherent models. Also, market share analysis and key trend analysis are the major success factors in the market report. To know more, please request an analyst call or drop down your inquiry.
The key research methodology used by DBMR research team is data triangulation which involves data mining, analysis of the impact of data variables on the market and primary (industry expert) validation. Data models include Vendor Positioning Grid, Market Time Line Analysis, Market Overview and Guide, Company Positioning Grid, Patent Analysis, Pricing Analysis, Company Market Share Analysis, Standards of Measurement, Global versus Regional and Vendor Share Analysis. To know more about the research methodology, drop in an inquiry to speak to our industry experts.
Customization Available
Data Bridge Market Research is a leader in advanced formative research. We take pride in servicing our existing and new customers with data and analysis that match and suits their goal. The report can be customized to include price trend analysis of target brands understanding the market for additional countries (ask for the list of countries), clinical trial results data, literature review, refurbished market and product base analysis. Market analysis of target competitors can be analyzed from technology-based analysis to market portfolio strategies. We can add as many competitors that you require data about in the format and data style you are looking for. Our team of analysts can also provide you data in crude raw excel files pivot tables (Fact book) or can assist you in creating presentations from the data sets available in the report.