Global Stem Cell Derived Biologic Therapies Market
Market Size in USD Billion
CAGR :
% 
 USD
5.21 Billion
USD
16.38 Billion
2025
2033
USD
5.21 Billion
USD
16.38 Billion
2025
2033
| 2026 - 2033 | |
| USD 5.21 Billion | |
| USD 16.38 Billion | |
|
|
|
|
Stem Cell-Derived Biologic Therapies Market Size
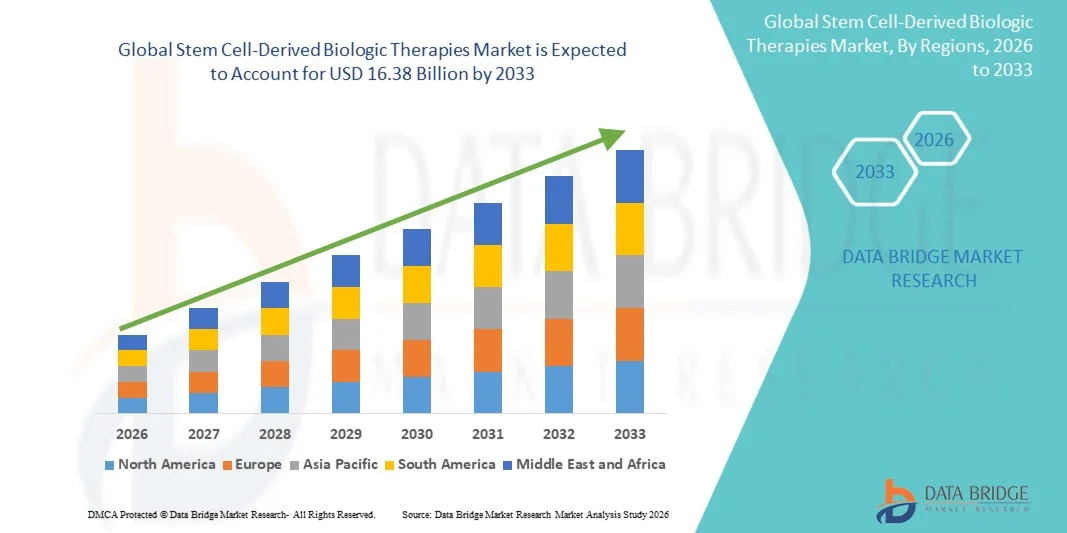
- The global stem cell-derived biologic therapies market size was valued at USD 5.21 billion in 2025 and is expected to reach USD 16.38 billion by 2033, at a CAGR of 15.40% during the forecast period
- The market growth is largely fueled by the increasing advancements in regenerative medicine and stem cell research, along with expanding clinical trials evaluating stem cell-derived biologics for the treatment of chronic and degenerative diseases
- Furthermore, the rising demand for innovative biologic therapies capable of repairing or regenerating damaged tissues, coupled with growing investments from biotechnology and pharmaceutical companies, is establishing stem cell-derived biologics as a transformative therapeutic approach. These converging factors are accelerating the development and adoption of such therapies, thereby significantly boosting the industry's growth
Stem Cell-Derived Biologic Therapies Market Analysis
- Stem cell-derived biologic therapies, which utilize biologically active components derived from stem cells to support tissue regeneration and disease treatment, are becoming increasingly significant in modern healthcare due to their potential to repair damaged tissues, regulate immune responses, and address complex chronic and degenerative diseases
- The escalating demand for stem cell-derived biologic therapies is primarily fueled by the growing prevalence of chronic conditions such as cardiovascular and neurological disorders, increasing investments in regenerative medicine research, and rising interest in advanced biologic treatments capable of offering long-term therapeutic benefits
- North America dominated the stem cell-derived biologic therapies market with the largest revenue share of 41.6% in 2025, characterized by strong biotechnology research capabilities, high healthcare spending, and the presence of major biotechnology and pharmaceutical companies, with the U.S. experiencing substantial growth in stem cell research programs and clinical trials supported by collaborations between academic institutions and industry players
- Asia-Pacific is expected to be the fastest growing region in the stem cell-derived biologic therapies market during the forecast period due to expanding biotechnology sectors, rising healthcare investments, and increasing government support for regenerative medicine research in countries such as China, Japan, and South Korea
- Allogeneic stem cell-derived therapies segment dominated the stem cell-derived biologic therapies market with a market share of 46.3% in 2025, driven by their scalability, availability of donor cell sources, and growing adoption in clinical research and therapeutic development for multiple disease indications
Report Scope and Stem Cell-Derived Biologic Therapies Market Segmentation
|
Attributes |
Stem Cell-Derived Biologic Therapies Key Market Insights |
|
Segments Covered |
|
|
Countries Covered |
North America
Europe
Asia-Pacific
Middle East and Africa
South America
|
|
Key Market Players |
|
|
Market Opportunities |
|
|
Value Added Data Infosets |
In addition to the insights on market scenarios such as market value, growth rate, segmentation, geographical coverage, and major players, the market reports curated by the Data Bridge Market Research also include in-depth expert analysis, patient epidemiology, pipeline analysis, pricing analysis, and regulatory framework |
Stem Cell-Derived Biologic Therapies Market Trends
“Rising Adoption of Regenerative Medicine and Advanced Cell-Based Therapeutics”
- A significant and accelerating trend in the global stem cell-derived biologic therapies market is the growing adoption of regenerative medicine approaches that utilize biologically active molecules derived from stem cells to repair or regenerate damaged tissues. This advancement is transforming treatment strategies across multiple therapeutic areas
- For instance, stem cell-derived biologics such as exosomes and growth factors are increasingly being explored for applications in neurological disorders, cardiovascular diseases, and musculoskeletal injuries, demonstrating promising results in clinical research and therapeutic development
- Continuous advancements in biotechnology and cell processing technologies are enabling the development of more targeted and effective biologic products derived from stem cells. These innovations are improving the scalability, consistency, and therapeutic potential of stem cell-derived biologics, making them more viable for clinical and commercial use
- Furthermore, increasing collaborations between biotechnology companies, pharmaceutical firms, and academic research institutes are accelerating the discovery and development of new stem cell-derived therapeutic candidates. These partnerships are supporting clinical trials, regulatory approvals, and the expansion of regenerative medicine pipelines globally
- The integration of advanced technologies such as gene editing, cell engineering, and biomaterial-based delivery systems is further enhancing the effectiveness and safety of stem cell-derived biologic therapies. These developments are contributing to the expansion of innovative treatment options for previously difficult-to-treat diseases
- The demand for stem cell-derived biologic therapies is increasing rapidly across healthcare systems as clinicians and patients seek more effective and regenerative treatment approaches for chronic and degenerative conditions, thereby driving continued market expansion
- Moreover, increasing government initiatives and funding programs supporting regenerative medicine research are encouraging biotechnology companies and research institutions to develop innovative stem cell-derived biologic therapies for a wide range of medical applications
Stem Cell-Derived Biologic Therapies Market Dynamics
Driver
“Increasing Prevalence of Chronic and Degenerative Diseases”
- The rising global burden of chronic and degenerative diseases such as cardiovascular disorders, neurological conditions, and musculoskeletal diseases is a major driver increasing the demand for stem cell-derived biologic therapies
- For instance, biotechnology companies and research institutions are actively investing in the development of stem cell-based biologics to address complex diseases that currently have limited treatment options, thereby accelerating innovation within the regenerative medicine sector
- As healthcare providers seek more advanced therapeutic solutions, stem cell-derived biologics offer promising capabilities for tissue repair, immune modulation, and long-term disease management, making them attractive alternatives to conventional treatments
- Furthermore, increasing research funding from governments, private investors, and pharmaceutical companies is supporting large-scale clinical trials and product development in the stem cell therapy field
- The expanding pipeline of regenerative medicine therapies, combined with technological advancements in stem cell research and biologic manufacturing, is expected to significantly drive market growth during the forecast period
- Growing awareness among healthcare professionals regarding the therapeutic potential of regenerative medicine is encouraging the adoption of stem cell-derived biologic therapies in clinical practice
- In addition, increasing investments from biotechnology startups and venture capital firms are accelerating innovation and commercialization of advanced stem cell-derived biologic products globally
Restraint/Challenge
“High Development Costs and Complex Regulatory Frameworks”
- The high cost associated with the research, development, and manufacturing of stem cell-derived biologic therapies presents a significant challenge to broader market adoption. These therapies often require advanced laboratory infrastructure, specialized expertise, and complex production processes
- For instance, conducting clinical trials for stem cell-based biologics involves extensive safety and efficacy evaluations, which can significantly increase development timelines and overall costs for biotechnology companies
- In addition, strict regulatory requirements governing stem cell therapies across different countries can create barriers for product approvals and commercialization. Regulatory agencies require extensive clinical evidence to ensure the safety, quality, and effectiveness of these advanced biologic treatments
- Differences in regulatory policies and ethical considerations related to certain stem cell sources can further complicate international development and market expansion efforts
- Addressing these challenges through improved manufacturing technologies, standardized regulatory frameworks, and increased investment in regenerative medicine research will be essential to support the long-term growth of the stem cell-derived biologic therapies market
- Limited reimbursement policies for advanced biologic therapies in several healthcare systems can restrict patient access and slow market adoption
- Moreover, technical challenges related to large-scale manufacturing, quality control, and product standardization can create additional hurdles for companies developing stem cell-derived biologic therapies
Stem Cell-Derived Biologic Therapies Market Scope
The market is segmented on the basis of therapy type, cell source, application, and end use
- By Therapy Type
On the basis of therapy type, the stem cell-derived biologic therapies market is segmented into allogeneic stem cell-derived therapies, autologous stem cell-derived therapies, and syngeneic stem cell-derived therapies. The allogeneic stem cell-derived therapies segment dominated the market with the largest market revenue share of 46.3% in 2025, primarily due to its scalability and ability to treat a large number of patients using donor-derived cells. These therapies allow manufacturers to develop standardized biologic products that can be produced in bulk, reducing production time compared to patient-specific approaches. The growing number of clinical trials evaluating allogeneic biologics for cardiovascular diseases, neurological disorders, and immune-related conditions is further supporting market expansion. In addition, biotechnology companies prefer allogeneic platforms because they offer greater commercial potential and streamlined manufacturing processes. The increasing collaboration between pharmaceutical companies and research institutions for the development of off-the-shelf regenerative treatments is also strengthening the dominance of this segment.
The autologous stem cell-derived therapies segment is anticipated to witness the fastest growth rate during the forecast period from 2026 to 2033, driven by the rising focus on personalized medicine and patient-specific treatment approaches. Autologous therapies use a patient’s own stem cells, which significantly reduces the risk of immune rejection and compatibility issues. Growing clinical research evaluating autologous biologics for orthopedic injuries, neurological disorders, and dermatological conditions is supporting segment expansion. Healthcare providers increasingly prefer these therapies due to their improved safety profile and long-term regenerative potential. Furthermore, advancements in cell isolation and processing technologies are making autologous therapies more efficient and accessible. These developments are expected to accelerate adoption across hospitals and regenerative medicine clinics worldwide.
- By Cell Source
On the basis of cell source, the stem cell-derived biologic therapies market is segmented into adult stem cells, embryonic stem cells, induced pluripotent stem cells (iPSCs), and others. The adult stem cells segment dominated the market with the largest market revenue share in 2025, primarily due to their extensive clinical use and relatively fewer ethical concerns compared to embryonic stem cells. Adult stem cells, particularly mesenchymal stem cells, are widely used in regenerative medicine for treating musculoskeletal disorders, cardiovascular diseases, and inflammatory conditions. These cells are easily obtainable from sources such as bone marrow and adipose tissue, making them highly practical for therapeutic development. In addition, adult stem cells have demonstrated strong immunomodulatory and regenerative properties, making them attractive for biologic therapy development. Many biotechnology companies are focusing on adult stem cell platforms due to their established clinical evidence and regulatory acceptance. These factors collectively contribute to the dominant position of this segment in the market.
The induced pluripotent stem cells (iPSCs) segment is expected to witness the fastest growth rate during the forecast period from 2026 to 2033, owing to their ability to generate patient-specific pluripotent cells without the ethical challenges associated with embryonic stem cells. iPSCs are increasingly used in advanced regenerative medicine research and drug discovery due to their capacity to differentiate into various cell types. Researchers and biotechnology firms are actively exploring iPSC-derived biologics for neurological diseases, cardiac repair, and oncology treatments. Technological advancements in gene editing and cell reprogramming are further enhancing the efficiency of iPSC production. In addition, the rising adoption of personalized medicine and disease modeling using iPSC platforms is accelerating research investments. These innovations are expected to significantly drive the growth of this segment in the coming years.
- By Application
On the basis of application, the stem cell-derived biologic therapies market is segmented into musculoskeletal disorders, cardiovascular diseases, oncology, neurological disorders, dermatology, and others. The musculoskeletal disorders segment dominated the market with the largest market revenue share in 2025, primarily driven by the increasing prevalence of orthopedic conditions such as osteoarthritis, bone injuries, and ligament damage. Stem cell-derived biologic therapies are widely explored for their regenerative capabilities in repairing bone, cartilage, and connective tissues. These therapies offer significant advantages over conventional treatments by promoting natural tissue healing and reducing recovery times. In addition, the growing aging population and increasing sports-related injuries are contributing to the rising demand for regenerative orthopedic treatments. Research institutions and biotechnology companies are actively conducting clinical trials for stem cell-derived biologics targeting joint and cartilage regeneration. As a result, this segment continues to hold a leading position within the market.
The neurological disorders segment is projected to witness the fastest growth rate from 2026 to 2033, driven by the increasing focus on developing regenerative therapies for complex neurological conditions such as Parkinson’s disease, spinal cord injuries, and Alzheimer’s disease. Stem cell-derived biologics are being studied for their ability to repair damaged neural tissues and support nerve regeneration. The rising burden of neurological disorders globally is encouraging governments and healthcare organizations to invest heavily in regenerative medicine research. In addition, ongoing clinical trials exploring stem cell-derived biologics for neurodegenerative diseases are expanding the therapeutic pipeline. Advancements in stem cell engineering and biomaterial delivery systems are also improving the effectiveness of treatments for neurological applications. These developments are expected to significantly accelerate growth in this segment.
- By End User
On the basis of end user, the stem cell-derived biologic therapies market is segmented into hospitals, biotechnology & pharmaceutical companies, research institutes, and stem cell banks. The biotechnology & pharmaceutical companies segment dominated the market with the largest revenue share in 2025, primarily due to their extensive involvement in research, product development, and commercialization of stem cell-derived biologic therapies. These companies invest heavily in clinical trials and advanced biotechnology platforms to develop innovative regenerative therapies. The increasing number of strategic partnerships between pharmaceutical firms and research institutions is also accelerating product development. Furthermore, biotechnology companies are actively expanding their regenerative medicine pipelines to address various chronic and degenerative diseases. Their strong financial resources and technological capabilities enable large-scale production and commercialization of biologic therapies. These factors collectively contribute to the dominance of this segment in the market.
The research institutes segment is anticipated to witness the fastest growth rate from 2026 to 2033, driven by the growing focus on stem cell research and regenerative medicine innovation worldwide. Academic and government research organizations play a critical role in discovering new therapeutic applications and advancing stem cell technologies. Increasing government funding and research grants for regenerative medicine projects are significantly supporting this segment. In addition, collaborations between research institutes and biotechnology companies are accelerating the development of novel stem cell-derived biologic products. Research institutes are also actively conducting preclinical and early-stage clinical studies to evaluate new treatment approaches. As scientific exploration continues to expand, this segment is expected to experience rapid growth during the forecast period.
Stem Cell-Derived Biologic Therapies Market Regional Analysis
- North America dominated the stem cell-derived biologic therapies market with the largest revenue share of 41.6% in 2025, characterized by strong biotechnology research capabilities, high healthcare spending, and the presence of major biotechnology and pharmaceutical companies
- Healthcare institutions and research organizations in the region place strong emphasis on advanced biologic therapies that support tissue regeneration and disease treatment, particularly for conditions such as neurological disorders, cardiovascular diseases, and musculoskeletal injuries
- This widespread adoption is further supported by well-established healthcare infrastructure, favorable research funding from government and private organizations, and a high concentration of clinical trials focused on stem cell-derived biologics, establishing these therapies as an important component of next-generation regenerative medicine solutions
U.S. Stem Cell-Derived Biologic Therapies Market Insight
The U.S. stem cell-derived biologic therapies market captured the largest revenue share of 79% in 2025 within North America, fueled by strong investments in regenerative medicine and the rapid expansion of biotechnology research. Healthcare institutions and biotechnology companies are increasingly focusing on the development of advanced biologic therapies for chronic and degenerative diseases. The growing number of clinical trials and research collaborations between academic institutions and pharmaceutical companies further accelerates market development. Moreover, supportive regulatory initiatives and funding programs for regenerative medicine research are significantly contributing to market expansion. The presence of leading biotechnology firms and well-established research infrastructure continues to strengthen the U.S. position in this market.
Europe Stem Cell-Derived Biologic Therapies Market Insight
The Europe stem cell-derived biologic therapies market is projected to expand at a substantial CAGR throughout the forecast period, primarily driven by increasing research activities and rising investments in regenerative medicine technologies. The growing burden of chronic diseases and the demand for advanced biologic treatment options are encouraging healthcare providers to adopt innovative stem cell-derived therapies. European governments and research organizations are actively supporting clinical studies and biotechnology development programs. The region is witnessing increased collaboration between biotechnology companies and academic research institutes to accelerate therapy development. In addition, favorable regulatory frameworks supporting advanced therapy medicinal products are promoting market growth across Europe.
U.K. Stem Cell-Derived Biologic Therapies Market Insight
The U.K. stem cell-derived biologic therapies market is anticipated to grow at a noteworthy CAGR during the forecast period, driven by strong government funding and a well-established biotechnology research ecosystem. The country has a strong focus on regenerative medicine innovation and advanced cell-based therapy development. Academic institutions and research organizations are actively collaborating with biotechnology companies to advance stem cell-derived biologic products. The increasing prevalence of chronic diseases and neurological disorders is further encouraging the development of regenerative treatment approaches. Moreover, government initiatives supporting biomedical innovation and clinical trials are expected to continue stimulating market growth.
Germany Stem Cell-Derived Biologic Therapies Market Insight
The Germany stem cell-derived biologic therapies market is expected to expand at a considerable CAGR during the forecast period, fueled by strong biotechnology infrastructure and increasing investments in advanced medical research. Germany’s emphasis on scientific innovation and pharmaceutical development supports the adoption of regenerative medicine technologies. Research institutes and biotechnology companies are actively exploring stem cell-derived biologics for applications in neurological disorders, cardiovascular diseases, and orthopedic conditions. The country’s strong healthcare infrastructure and focus on medical innovation further promote therapy development. In addition, increasing partnerships between academic institutions and biotechnology firms are supporting the growth of regenerative medicine research in Germany.
Asia-Pacific Stem Cell-Derived Biologic Therapies Market Insight
The Asia-Pacific stem cell-derived biologic therapies market is poised to grow at the fastest CAGR during the forecast period of 2026 to 2033, driven by increasing investments in biotechnology and expanding regenerative medicine research in countries such as China, Japan, and India. The region’s rapidly improving healthcare infrastructure and growing demand for advanced therapeutic solutions are accelerating the adoption of stem cell-derived biologics. Governments across Asia-Pacific are also actively promoting biotechnology innovation through funding programs and research initiatives. In addition, the rising prevalence of chronic diseases and increasing awareness of regenerative medicine are supporting market growth. These factors are contributing to the rapid expansion of the stem cell-derived biologic therapies market across the region.
Japan Stem Cell-Derived Biologic Therapies Market Insight
The Japan stem cell-derived biologic therapies market is gaining momentum due to the country’s strong leadership in regenerative medicine and advanced biotechnology research. Japan has implemented progressive regulatory frameworks to accelerate the development and commercialization of stem cell-based therapies. The country’s research institutions and biotechnology companies are actively conducting clinical studies focused on regenerative treatments for neurological and cardiovascular diseases. The increasing aging population is also creating demand for innovative therapies capable of addressing degenerative health conditions. Furthermore, strong government support for regenerative medicine initiatives continues to stimulate market growth in Japan.
India Stem Cell-Derived Biologic Therapies Market Insight
The India stem cell-derived biologic therapies market accounted for the largest market revenue share in Asia-Pacific in 2025, attributed to the country’s rapidly expanding biotechnology sector and increasing investments in medical research. India is emerging as a major hub for regenerative medicine studies and clinical research activities. The growing burden of chronic diseases and increasing awareness about advanced biologic therapies are encouraging healthcare institutions to explore stem cell-based treatment options. Government initiatives supporting biotechnology innovation and research collaborations are also contributing to market expansion. In addition, the presence of a large patient population and expanding healthcare infrastructure are key factors propelling the market in India.
Stem Cell-Derived Biologic Therapies Market Share
The Stem Cell-Derived Biologic Therapies industry is primarily led by well-established companies, including:
- Mesoblast Ltd. (Australia)
- Gamida Cell Ltd. (Israel)
- BrainStorm Cell Therapeutics Inc. (U.S.)
- Cynata Therapeutics Limited (Australia)
- Pluri Inc. (Israel)
- Lineage Cell Therapeutics, Inc. (U.S.)
- Vericel Corporation (U.S.)
- Fate Therapeutics, Inc. (U.S.)
- Athersys, Inc. (U.S.)
- BioRestorative Therapies, Inc. (U.S.)
- Aspen Neuroscience, Inc. (U.S.)
- BlueRock Therapeutics LP (U.S.)
- Century Therapeutics, Inc. (U.S.)
- Garuda Therapeutics, Inc. (U.S.)
- Cellular Biomedicine Group, Inc. (U.S.)
- Stemcell Technologies Inc. (Canada)
- Caladrius Biosciences, Inc. (U.S.)
- ReNeuron Group plc (U.K.)
- Orgenesis Inc. (U.S.)
- Sangamo Therapeutics, Inc. (U.S.)
What are the Recent Developments in Global Stem Cell-Derived Biologic Therapies Market?
- In December 2025, Capricor Therapeutics, a biotechnology company focused on regenerative medicine, announced positive topline results from its Phase 3 HOPE-3 clinical trial evaluating Deramiocel, a stem cell-derived therapy for Duchenne muscular dystrophy. The study demonstrated meaningful functional improvements in patients, highlighting the potential of stem cell-derived biologic therapies to treat rare degenerative diseases and advancing the therapy toward possible regulatory approval
- In December 2025, NurExone Biologic Inc. reported new laboratory findings showing the anti-inflammatory effects of its stem cell-derived exosome therapy, which aims to promote regeneration in the central nervous system. The results support the potential of exosome-based biologics derived from stem cells for treating neurological injuries and inflammatory conditions, strengthening the pipeline for cell-free regenerative therapies
- In September 2025, Bayer AG, through its subsidiary BlueRock Therapeutics, initiated a Phase III clinical trial for its stem cell-derived therapy targeting Parkinson’s disease. The therapy uses lab-grown dopamine-producing nerve cells derived from stem cells to restore damaged neural networks in patients, representing a significant step toward commercial regenerative treatments for neurodegenerative disorders
- In July 2025, Tulsi Therapeutics, a biotechnology startup incubated at the University of Hyderabad, developed a novel therapy called Tulsi-28X, combining stem cells with exosomes derived from umbilical cord tissue to treat chronic liver failure. Preclinical trials demonstrated significant liver regeneration in animal studies, and the company has applied for regulatory approval to begin human clinical trials
- In May 2025, XellSmart Biopharmaceutical, a regenerative medicine company, announced that its allogeneic induced pluripotent stem cell (iPSC)-derived therapy for spinal cord injury received approval to begin a registrational Phase I clinical trial in the United States. The therapy aims to regenerate damaged neural tissues and restore motor function, marking a milestone for iPSC-derived biologic therapies in neurological treatment development
SKU-
Get online access to the report on the World's First Market Intelligence Cloud
- Interactive Data Analysis Dashboard
- Company Analysis Dashboard for high growth potential opportunities
- Research Analyst Access for customization & queries
- Competitor Analysis with Interactive dashboard
- Latest News, Updates & Trend analysis
- Harness the Power of Benchmark Analysis for Comprehensive Competitor Tracking
Research Methodology
Data collection and base year analysis are done using data collection modules with large sample sizes. The stage includes obtaining market information or related data through various sources and strategies. It includes examining and planning all the data acquired from the past in advance. It likewise envelops the examination of information inconsistencies seen across different information sources. The market data is analysed and estimated using market statistical and coherent models. Also, market share analysis and key trend analysis are the major success factors in the market report. To know more, please request an analyst call or drop down your inquiry.
The key research methodology used by DBMR research team is data triangulation which involves data mining, analysis of the impact of data variables on the market and primary (industry expert) validation. Data models include Vendor Positioning Grid, Market Time Line Analysis, Market Overview and Guide, Company Positioning Grid, Patent Analysis, Pricing Analysis, Company Market Share Analysis, Standards of Measurement, Global versus Regional and Vendor Share Analysis. To know more about the research methodology, drop in an inquiry to speak to our industry experts.
Customization Available
Data Bridge Market Research is a leader in advanced formative research. We take pride in servicing our existing and new customers with data and analysis that match and suits their goal. The report can be customized to include price trend analysis of target brands understanding the market for additional countries (ask for the list of countries), clinical trial results data, literature review, refurbished market and product base analysis. Market analysis of target competitors can be analyzed from technology-based analysis to market portfolio strategies. We can add as many competitors that you require data about in the format and data style you are looking for. Our team of analysts can also provide you data in crude raw excel files pivot tables (Fact book) or can assist you in creating presentations from the data sets available in the report.