Global Synthetic Mrna Therapeutics Market
Market Size in USD Billion
CAGR :
% 
 USD
2.86 Billion
USD
11.73 Billion
2025
2033
USD
2.86 Billion
USD
11.73 Billion
2025
2033
| 2026 - 2033 | |
| USD 2.86 Billion | |
| USD 11.73 Billion | |
|
|
|
|
Synthetic mRNA Therapeutics Market Size
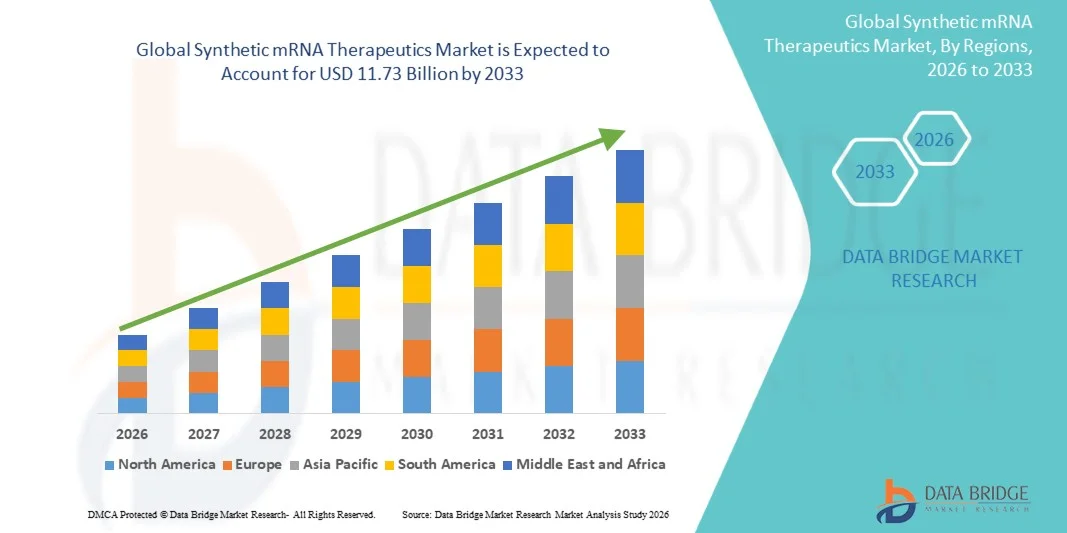
- The global synthetic mRNA therapeutics market size was valued at USD 2.86 billion in 2025 and is expected to reach USD 11.73 billion by 2033, at a CAGR of 19.30% during the forecast period
- The market growth is largely fueled by the rapid advancements in mRNA technology platforms and increasing adoption of precision medicine approaches, along with rising investments in biotechnology and vaccine development, leading to expanded applications of synthetic mRNA therapeutics in disease treatment and prevention
- Furthermore, growing prevalence of cancer, infectious diseases, and rare genetic disorders, coupled with strong clinical pipeline activity and increasing focus on personalized medicine, is establishing synthetic mRNA therapeutics as a transformative treatment modality. These converging factors are accelerating the uptake of Synthetic mRNA Therapeutics solutions, thereby significantly boosting the market growth
Synthetic mRNA Therapeutics Market Analysis
- Synthetic mRNA therapeutics, designed to deliver messenger RNA sequences that instruct cells to produce specific proteins for therapeutic or preventive purposes, are emerging as a transformative approach in modern medicine due to their precision, rapid development potential, and broad applicability across multiple disease areas
- The escalating demand for synthetic mRNA therapeutics is primarily fueled by increasing prevalence of cancer, infectious diseases, and rare genetic disorders, along with strong advancements in RNA delivery technologies, vaccine innovation, and growing investments in biotechnology research
- North America dominated the synthetic mRNA therapeutics market with the largest revenue share of approximately 44.2% in 2025, characterized by strong biotech infrastructure, high R&D investment, and early adoption of mRNA-based platforms, with the U.S. leading in clinical trials and commercialization efforts
- Asia-Pacific is expected to be the fastest growing region in the synthetic mRNA therapeutics market during the forecast period due to expanding biotechnology sector, increasing government funding, and rising focus on advanced therapeutics development
- The Cancer Immunotherapy segment dominated the largest market revenue share of 38.5% in 2025, driven by the rising global cancer burden and rapid adoption of personalized medicine approaches
Report Scope and Synthetic mRNA Therapeutics Market Segmentation
|
Attributes |
Synthetic mRNA Therapeutics Key Market Insights |
|
Segments Covered |
|
|
Countries Covered |
North America
Europe
Asia-Pacific
Middle East and Africa
South America
|
|
Key Market Players |
|
|
Market Opportunities |
|
|
Value Added Data Infosets |
In addition to the insights on market scenarios such as market value, growth rate, segmentation, geographical coverage, and major players, the market reports curated by the Data Bridge Market Research also include in-depth expert analysis, patient epidemiology, pipeline analysis, pricing analysis, and regulatory framework. |
Synthetic mRNA Therapeutics Market Trends
“Advancements in mRNA Platform Technologies and Next-Generation Therapeutic Development”
- A significant and accelerating trend in the global Synthetic mRNA Therapeutics market is the increasing adoption of advanced mRNA platform technologies for the development of next-generation vaccines and targeted therapies. These innovations are transforming the treatment landscape for infectious diseases, cancer, and rare genetic disorders
- For instance, synthetic mRNA platforms enable the rapid design and production of therapeutic candidates by encoding specific proteins that can trigger immune responses or replace defective proteins in the body
- Continuous improvements in lipid nanoparticle (LNP) delivery systems are significantly enhancing the stability and cellular uptake of mRNA-based drugs
- Furthermore, advancements in sequence optimization and nucleoside modification are improving protein expression efficiency and reducing immunogenicity, thereby increasing therapeutic effectiveness
- Growing investment in personalized medicine approaches is also accelerating the development of patient-specific mRNA therapies
- This trend toward highly adaptable, fast-developing, and precision-based therapeutic platforms is significantly shaping the global Synthetic mRNA Therapeutics market
Synthetic mRNA Therapeutics Market Dynamics
Driver
“Rising Prevalence of Chronic Diseases and Expanding Applications of mRNA Technology”
- The increasing global burden of chronic diseases such as cancer, infectious diseases, and rare genetic disorders is a major driver of the Synthetic mRNA Therapeutics market
- For instance, the success of mRNA-based vaccines during global health emergencies has significantly accelerated research and development in therapeutic applications beyond infectious diseases
- Growing demand for targeted and personalized treatment approaches is encouraging pharmaceutical companies to invest heavily in mRNA-based drug development
- Expanding clinical trials exploring mRNA therapies for oncology, cardiovascular diseases, and metabolic disorders are further supporting market growth
- Increasing government funding and supportive regulatory frameworks for advanced biologics are also contributing to market expansion
- Rapid technological advancements in biotechnology and molecular medicine are enabling faster commercialization of novel mRNA-based therapies globally
Restraint/Challenge
“Complex Manufacturing Processes and Stringent Regulatory Requirements”
- The complex manufacturing and storage requirements of synthetic mRNA therapeutics remain a significant challenge for large-scale commercialization
- These therapies require highly controlled production environments, advanced formulation techniques, and cold-chain logistics to maintain stability and efficacy
- For instance, maintaining the stability of mRNA molecules during transport and storage continues to be a major technical limitation for global distribution
- In addition, stringent regulatory approval processes for novel biologics can delay product development timelines and increase overall costs
- High research and development expenses associated with clinical trials and manufacturing scale-up further restrict market entry for smaller companies
- Addressing these challenges through improved delivery technologies, manufacturing innovation, and streamlined regulatory pathways will be essential for sustained growth of the Synthetic mRNA Therapeutics market
Synthetic mRNA Therapeutics Market Scope
The market is segmented on the basis of application and end user.
• By Application
On the basis of application, the Synthetic mRNA Therapeutics market is segmented into Infectious Diseases, Cancer Immunotherapy, Rare Genetic Disorders, Cardiovascular Diseases, and Others. The Cancer Immunotherapy segment dominated the largest market revenue share of 38.5% in 2025, driven by the rising global cancer burden and rapid adoption of personalized medicine approaches. Increasing use of mRNA-based cancer vaccines and immunotherapies is supporting strong clinical uptake due to their ability to stimulate targeted immune responses. Pharmaceutical companies are heavily investing in oncology-focused pipelines, which is accelerating innovation. Expanding clinical trials and favorable regulatory pathways are further strengthening segment growth. The availability of advanced lipid nanoparticle delivery systems is improving drug stability and effectiveness. Growing collaboration between biotech firms and research institutes is accelerating development timelines. Rising healthcare expenditure and unmet oncology needs are also major contributors. North America continues to dominate due to strong R&D infrastructure, while Europe is steadily expanding clinical adoption. Continuous pipeline expansion is improving long-term commercial potential. Overall, oncology remains the most lucrative application area for mRNA therapeutics.
The Infectious Diseases segment is expected to witness the fastest CAGR of 17.2% from 2026 to 2033, driven by increasing demand for rapid vaccine development platforms. The success of mRNA vaccines during global pandemics has significantly boosted confidence in this technology. Governments worldwide are investing in pandemic preparedness programs, further accelerating adoption. mRNA platforms allow faster design and production compared to traditional vaccine technologies. Rising prevalence of emerging infectious diseases is increasing demand for flexible and scalable vaccine solutions. Pharmaceutical companies are expanding manufacturing capacity to meet future outbreak requirements. Continuous advancements in antigen design are improving immune response efficiency. Strong pipeline development for influenza, RSV, and other viral infections is supporting growth. Regulatory agencies are also streamlining approval processes for emergency vaccines. Public health initiatives and awareness campaigns are improving vaccination coverage. Emerging economies are increasingly adopting modern vaccine platforms. Overall, this segment is expanding rapidly due to global health security priorities.
• By End User
On the basis of end user, the Synthetic mRNA Therapeutics market is segmented into Hospitals, Biotechnology & Pharmaceutical Companies, Research Institutes, and Others. The Biotechnology & Pharmaceutical Companies segment dominated the largest market revenue share of 52.6% in 2025, driven by strong R&D investments and large-scale drug development programs. These companies are the primary innovators in mRNA technology and lead most clinical trials globally. Significant funding availability allows them to invest in advanced delivery systems and scalable production technologies. Strategic collaborations, licensing deals, and mergers are further strengthening market dominance. Companies are increasingly focusing on oncology, infectious diseases, and rare genetic disorders. Integration of AI and bioinformatics tools is accelerating drug discovery processes. Established regulatory expertise helps in faster approvals and commercialization. North America remains the leading region due to strong biotech presence. Continuous expansion of biologics pipelines is enhancing growth prospects. High production capabilities enable large-scale vaccine and therapeutic manufacturing. Overall, this segment remains the backbone of mRNA therapeutic innovation.
The Research Institutes segment is expected to witness the fastest CAGR of 16.5% from 2026 to 2033, driven by increasing public and private funding for molecular and genomic research. Academic and clinical research collaborations are expanding rapidly across global institutions. These institutes play a crucial role in early-stage discovery and validation of mRNA-based therapies. Rising focus on precision medicine and genetic research is boosting demand for advanced tools and platforms. Government grants and international funding programs are accelerating innovation. Increasing participation in global research consortiums is strengthening knowledge sharing. Technological advancements in sequencing and molecular biology are enhancing research outcomes. Growing emphasis on translational research is bridging the gap between lab discoveries and clinical applications. Asia-Pacific is emerging as a fast-growing hub for academic research expansion. Strong infrastructure development in universities and laboratories is supporting growth. Overall, this segment is gaining momentum due to rising scientific collaboration and funding support
Synthetic mRNA Therapeutics Market Regional Analysis
- North America dominated the Synthetic mRNA Therapeutics market with the largest revenue share of approximately 44.2% in 2025, characterized by strong biotechnology infrastructure, high R&D investment, and early adoption of mRNA-based platforms
- The region benefits from a robust ecosystem of biotech companies, leading research institutions, and favorable regulatory pathways supporting rapid clinical development and commercialization of mRNA-based therapies. The U.S. leads in clinical trials, innovation, and large-scale therapeutic pipeline development
- Increasing prevalence of chronic and infectious diseases, rising demand for personalized medicine, and growing investments in advanced genetic and molecular therapeutics are key factors driving market growth in North America. Continuous advancements in mRNA delivery technologies and strong funding from both public and private sectors further support market expansion
U.S. Synthetic mRNA Therapeutics Market Insight
The U.S. Synthetic mRNA Therapeutics market captured the largest revenue share within North America in 2025, driven by its leadership in biotechnology innovation and strong clinical research activity. The country has a well-established ecosystem of pharmaceutical and biotech companies actively developing mRNA-based vaccines and therapeutics. High R&D funding, strong academic-industry collaboration, and rapid regulatory approvals are accelerating market growth.
Europe Synthetic mRNA Therapeutics Market Insight
The Europe Synthetic mRNA Therapeutics market is projected to expand at a substantial CAGR during the forecast period, supported by increasing investments in biotechnology research and strong government backing for advanced therapeutic development. The region is witnessing growing collaboration between academic institutions and biotech firms, particularly in Germany, France, and the U.K., which is accelerating innovation in mRNA platforms.
U.K. Synthetic mRNA Therapeutics Market Insight
The U.K. Synthetic mRNA Therapeutics market is expected to grow at a notable CAGR, driven by strong biomedical research infrastructure and increasing funding for genomic medicine. The presence of leading research universities and biotech startups is fostering innovation in mRNA-based drug development. In addition, national healthcare initiatives supporting advanced therapeutics are contributing to market expansion.
Germany Synthetic mRNA Therapeutics Market Insight
The Germany Synthetic mRNA Therapeutics market is anticipated to expand at a considerable CAGR, fueled by strong pharmaceutical manufacturing capabilities and a well-established biotechnology sector. Germany is a key hub for mRNA research and production, supported by substantial investments in life sciences and a strong focus on translational research.
Asia-Pacific Synthetic mRNA Therapeutics Market Insight
The Asia-Pacific Synthetic mRNA Therapeutics market is expected to grow at the fastest CAGR during the forecast period, driven by expanding biotechnology capabilities, increasing government funding, and rising focus on advanced therapeutics development. Countries such as China, Japan, and South Korea are investing heavily in mRNA research infrastructure and clinical development programs, accelerating regional growth.
Japan Synthetic mRNA Therapeutics Market Insight
The Japan Synthetic mRNA Therapeutics market is gaining momentum due to strong government support for regenerative medicine and advanced biologics. The country’s aging population and high healthcare standards are driving demand for innovative therapeutic solutions. Japan’s biotechnology firms are increasingly investing in mRNA technology for vaccines and precision medicine applications.
China Synthetic mRNA Therapeutics Market Insight
The China Synthetic mRNA Therapeutics market accounted for the largest revenue share in Asia-Pacific in 2025, supported by rapid expansion of the domestic biotechnology sector and increasing investment in advanced drug development. Strong government initiatives, rising clinical research activity, and growing collaborations with global biotech firms are key factors driving market growth.
Synthetic mRNA Therapeutics Market Share
The Synthetic mRNA Therapeutics industry is primarily led by well-established companies, including:
- Moderna, Inc. (U.S.)
- BioNTech SE (Germany)
- Pfizer Inc. (U.S.)
- CureVac N.V. (Germany)
- Arcturus Therapeutics Holdings Inc. (U.S.)
- Translate Bio (Sanofi) (U.S.)
- Acuitas Therapeutics Inc. (Canada)
- Ethris GmbH (Germany)
- GSK plc (U.K.)
- Sanofi S.A. (France)
- ModernaTx (U.S.)
- Beam Therapeutics Inc. (U.S.)
- AstraZeneca plc (U.K.)
- Daiichi Sankyo Company, Ltd. (Japan)
- RiboX Therapeutics (China)
- eTheRNA Immunotherapies (Belgium)
- Replicate Bioscience (U.S.)
- Providence Therapeutics (Canada)
- Arcturus (Singapore operations)
- Genentech (Roche) (U.S.)
Latest Developments in Global Synthetic mRNA Therapeutics Market
- In December 2021, Moderna began clinical development of next-generation mRNA vaccine candidates beyond COVID-19, including multivalent and variant-adapted mRNA platforms designed to expand therapeutic applications into infectious diseases and personalized immunotherapy. This marked a shift from pandemic response to broader synthetic mRNA therapeutic pipelines
- In July 2022, Japan approved the first self-amplifying mRNA (saRNA) vaccine (ARCT-154 by CSL and Arcturus Therapeutics), representing a major advancement in synthetic mRNA technology with improved durability and lower dosage requirements compared to conventional mRNA vaccines
- In March 2023, BioNTech expanded its synthetic mRNA cancer immunotherapy pipeline through global clinical collaborations, including large-scale personalized mRNA vaccine trials for oncology, targeting up to 10,000 patients by 2030 in partnership with the UK government
- In June 2024, CSPC Pharmaceutical Group received regulatory approval in China to begin clinical trials for an mRNA-LNP-based cancer therapy (SYS6020), marking one of the first major mRNA-based cell therapy programs targeting malignancies beyond infectious diseases
- In July 2024, GSK advanced its seasonal influenza mRNA vaccine candidate into late-stage clinical trials after demonstrating strong immune responses across age groups, reinforcing pharmaceutical investment in next-generation synthetic mRNA vaccines
- In December 2024, BioNTech strengthened its synthetic mRNA therapeutic portfolio by acquiring Biotheus, gaining full rights to advanced oncology mRNA candidates including BNT327/PM8002, supporting expansion into immuno-oncology applications
- In May 2025, Moderna received U.S. FDA approval for mNEXSPIKE (mRNA-1283), a next-generation synthetic mRNA vaccine demonstrating enhanced immune response and extended platform capability for high-risk populations
SKU-
Get online access to the report on the World's First Market Intelligence Cloud
- Interactive Data Analysis Dashboard
- Company Analysis Dashboard for high growth potential opportunities
- Research Analyst Access for customization & queries
- Competitor Analysis with Interactive dashboard
- Latest News, Updates & Trend analysis
- Harness the Power of Benchmark Analysis for Comprehensive Competitor Tracking
Research Methodology
Data collection and base year analysis are done using data collection modules with large sample sizes. The stage includes obtaining market information or related data through various sources and strategies. It includes examining and planning all the data acquired from the past in advance. It likewise envelops the examination of information inconsistencies seen across different information sources. The market data is analysed and estimated using market statistical and coherent models. Also, market share analysis and key trend analysis are the major success factors in the market report. To know more, please request an analyst call or drop down your inquiry.
The key research methodology used by DBMR research team is data triangulation which involves data mining, analysis of the impact of data variables on the market and primary (industry expert) validation. Data models include Vendor Positioning Grid, Market Time Line Analysis, Market Overview and Guide, Company Positioning Grid, Patent Analysis, Pricing Analysis, Company Market Share Analysis, Standards of Measurement, Global versus Regional and Vendor Share Analysis. To know more about the research methodology, drop in an inquiry to speak to our industry experts.
Customization Available
Data Bridge Market Research is a leader in advanced formative research. We take pride in servicing our existing and new customers with data and analysis that match and suits their goal. The report can be customized to include price trend analysis of target brands understanding the market for additional countries (ask for the list of countries), clinical trial results data, literature review, refurbished market and product base analysis. Market analysis of target competitors can be analyzed from technology-based analysis to market portfolio strategies. We can add as many competitors that you require data about in the format and data style you are looking for. Our team of analysts can also provide you data in crude raw excel files pivot tables (Fact book) or can assist you in creating presentations from the data sets available in the report.