North America Depth Of Anesthesia Monitoring Market
Market Size in USD Million
CAGR :
% 
 USD
89.87 Million
USD
150.98 Million
2022
2030
USD
89.87 Million
USD
150.98 Million
2022
2030
| 2023 - 2030 | |
| USD 89.87 Million | |
| USD 150.98 Million | |
|
|
|
|
North America Depth of Anesthesia Monitoring Market Analysis and Size
The increasing health consciousness and rising need to monitor patients’ depth of general anesthesia is boosting the market forward. Presently, numerous research studies are taking place, which is anticipated to create a cutting-edge advantage for manufacturers to develop new, innovative, and advanced depth of anesthesia monitoring device, which is anticipated to provide numerous other opportunities in the market.
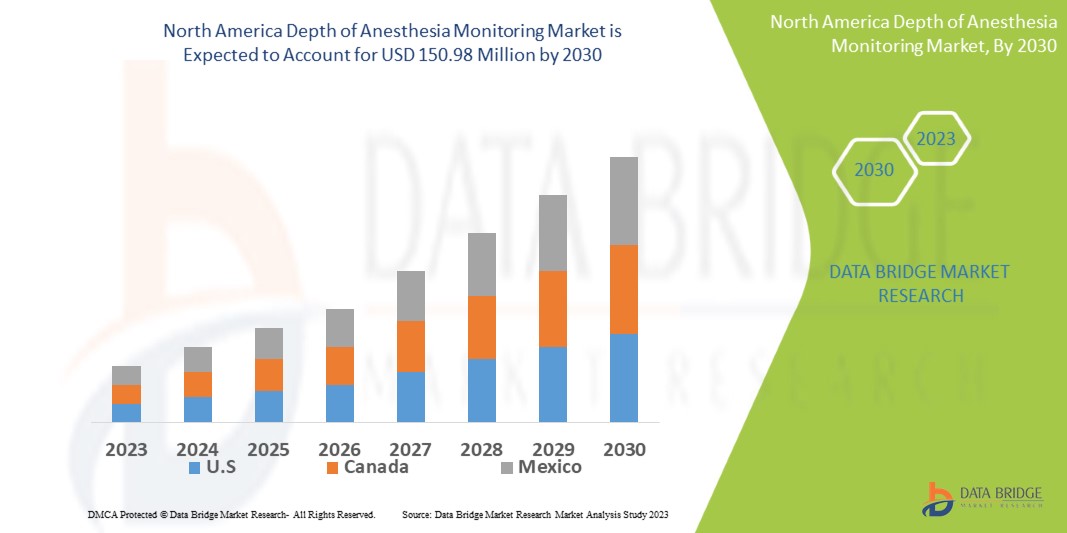
Data Bridge Market Research analyses that the depth of anesthesia monitoring market is expected to reach USD 150.98 million by 2030, which is USD 89.87 million in 2022, and is expected to undergo a CAGR of 6.7% during the forecast period 2022 to 2030. In addition to the insights on market scenarios such as market value, growth rate, segmentation, geographical coverage, and major players, the market reports curated by the Data Bridge Market Research also include depth expert analysis, patient epidemiology, pipeline analysis, pricing analysis, and regulatory framework.
North America Depth of Anesthesia Monitoring Market Scope and Segmentation
|
Report Metric |
Details |
|
Forecast Period |
2023 to 2030 |
|
Base Year |
2022 |
|
Historic Years |
2021 (Customizable to 2015 - 2020) |
|
Quantitative Units |
Revenue in USD Million, Volumes in Units, Pricing in USD |
|
Segments Covered |
Technology (Bispectral Index, Entropy and Patient State Index (PSI)), Product (Devices and Consumables), Mode of Purchase (Purchasing Organization and Direct Purchase), Application (Fluid Management, Cesarean Section, Laparoscopic Surgery, Vascular Surgery, Orthopedic Surgery, Cardiological Interventions, Emergency Medicine, Pediatric Surgery and Others), Distribution Channel (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies And Others) |
|
Countries Covered |
U.S., Canada and Mexico in North America |
|
Market Players Covered |
Drägerwerk AG & Co. KGaA (Germany), EDAN Instruments, Inc. (China), Masimo (U.S.), Shenzhen Mindray Bio-Medical Electronics Co., Ltd (China), Spacelabs Healthcare (India), Koninklijke Philips N.V (Netherlands), SCHILLER (Switzerland), Medtronic (Ireland), GE Healthcare (U.S.), NIHON KOHDEN CORPORATION (Japan) |
|
Market Opportunities |
|
Market Definition
During general anesthesia, a patient's anesthetic dose is monitored through depth of anesthesia monitors. The increased frequency of procedures requiring the injection of general anesthesia into the patient is crucial to the market's expansion. Heart surgery, knee and hip replacement surgery, brain surgery use general anesthesia regularly. Furthermore, with widespread cosmetic treatments, general anesthesia is injected.
North America Depth of Anesthesia Monitoring Market Dynamics
Drivers
- Increasing incidence of chronic diseases in elderly population
The elderly population is growing the demand for anesthesia depth monitoring equipment. This is mainly because of the elderly population's increased vulnerability to chronic diseases. An increase in the frequency of gynaecological, neurological, cardiovascular and urological problems in people aged 65 years and above is boosting the depth of anesthesia monitoring market forward. Around 45%, or 133 million, of all U.S. citizens suffer from at least one chronic disease, and the number is increasing. The incidence rate was highest among women and adults aged 65 or above, and those living in rural areas. Thus, this factor boosts the market growth.
- Increase rate in surgeries due to osteoporosis and road accidents
Osteoporosis is a major health issue, particularly among the aged population. The condition is related with joint and bone wear in the knee, hip, wrist and spine. Each year, osteoporosis causes around 8.6 million fractures in this region. Women are most likely to be affected by this illness. A rise in the prevalence of osteoporosis and osteoarthritis, and an increase in the number of knee and hip transplantation surgeries, are boosting the depth of anesthesia monitoring market.
Opportunities
- Growing product launches
There have been increasing drug launches for different surgeries, improving the market growth. For instance, 'Safira,' a Medovate medicine approved by the U.S. Food and Drug Administration (FDA) was launched In February 2020. For regional anesthetics, Safira stands for Safer Injection. The gadget contains a safety function that prevents anesthetic from being delivered at dangerous pressures, reducing the chance of peripheral nerve injury. Furthermore, GE Healthcare launched the new CS600 Carestation Series in Indonesia—the latest technology of anesthetic tool which is easier to operate, flexible, and capable of decreasing errors in surgery rooms in 2017. It is possible to increase the quality of anesthesia and perioperative care with safer processes of surgery. Thus, increasing product launches boost the growth of the market.
Restraints/Challenges
- Side-effects of anesthesia
There are various adverse effects that are associated with anesthesia that impedes the growth of the market. Effects such as nausea, dry mouth, itching, muscle aches, sleepiness, sore throat, and shivering hampers doctors' adoption of anesthesia. This factor is anticipated to limit the market growth.
This depth of anesthesia monitoring market report provides details of new recent developments, trade regulations, import-export analysis, production analysis, value chain optimization, market share, impact of domestic and localized market players, analyses opportunities in terms of emerging revenue pockets, changes in market regulations, strategic market growth analysis, market size, category market growths, application niches and dominance, product approvals, product launches, geographic expansions, technological innovations in the market. To gain more info on the depth of anesthesia monitoring market contact Data Bridge Market Research for an Analyst Brief, our team will help you take an informed market decision to achieve market growth.
Recent Developments:
- In 2019, NIHON KOHDEN CORPORATION launched new patient vital signs monitor which is mostly designed to enhance monitoring capabilities to outpatient facilities. After launching the new product, the company improved their product portfolio in the market.
- In 2019, SCHILLER announced that they have launched ECG device medilogAR. The patient can be screened for up to 14 days as the new product has dual battery concept. This leads to more production in the market because of its dual battery concept.
North America Depth of Anesthesia Monitoring Market Scope
The depth of anesthesia monitoring market is segmented on the basis of technology, product, mode of purchase, application, end user and distribution channel. The growth amongst these segments will help you analyze meagre growth segments in the industries and provide the users with a valuable market overview and market insights to help them make strategic decisions for identifying core market applications.
Product
- Devices
- Consumables
Technology
- Bispectral Index
- Entropy
- Patient State Index (PSI)
Mode of Purchase
- Purchasing Organization
- Direct Purchase
Application
- Fluid Management
- Cesarean Section
- Laparoscopic Surgery
- Vascular Surgery
- Orthopedic Surgery
- Cardiological Interventions
- Emergency Medicine
- Pediatric Surgery
Distribution Channel
- Direct Tender
- Retail Sales
End-User
- Hospitals
- Clinics
- Ambulatory Surgical Center
Depth of Anesthesia Monitoring Regional Analysis/Insights
The depth of anesthesia monitoring market is analyzed and market size insights and trends are provided by types of technology, product, mode of purchase, application, end user and distribution channel as referenced above.
The countries covered in the depth of anesthesia monitoring market report are U.S., Canada and Mexico in North America.
U.S. is expected to lead the market in the forecast period of 2023 to 2030 due to the incidence of large number of depth of anesthesia monitor manufacturing companies in the country and in the North America. Furthermore, the U.S. has skilled professionals and healthcare infrastructure to adopt advanced technologies in this region.
The country section of the report also provides individual market impacting factors and changes in regulation in the market domestically that impacts the current and future trends of the market. Data points like down-stream and upstream value chain analysis, technical trends and porter's five forces analysis, case studies are some of the pointers used to forecast the market scenario for individual countries. Also, the presence and availability of North America brands and their challenges faced due to large or scarce competition from local and domestic brands, impact of domestic tariffs and trade routes are considered while providing forecast analysis of the country data.
Competitive Landscape and North America Depth of Anesthesia Monitoring Market Share Analysis
The depth of anesthesia monitoring market competitive landscape provides details by competitor. Details included are company overview, company financials, revenue generated, market potential, investment in research and development, new market initiatives, North America presence, production sites and facilities, production capacities, company strengths and weaknesses, product launch, product width and breadth, application dominance. The above data points provided are only related to the companies' focus related to depth of anesthesia monitoring market.
Some of the major players operating in the depth of anesthesia monitoring market are:
- Drägerwerk AG & Co. KGaA (Germany)
- EDAN Instruments, Inc. (China)
- Masimo (U.S.)
- Shenzhen Mindray Bio-Medical Electronics Co., Ltd (China)
- Spacelabs Healthcare (India)
- Koninklijke Philips N.V (Netherlands)
- SCHILLER (Switzerland)
- Medtronic (Ireland)
- GE Healthcare (U.S.)
- NIHON KOHDEN CORPORATION (Japan)
SKU-
Get online access to the report on the World's First Market Intelligence Cloud
- Interactive Data Analysis Dashboard
- Company Analysis Dashboard for high growth potential opportunities
- Research Analyst Access for customization & queries
- Competitor Analysis with Interactive dashboard
- Latest News, Updates & Trend analysis
- Harness the Power of Benchmark Analysis for Comprehensive Competitor Tracking
Research Methodology
Data collection and base year analysis are done using data collection modules with large sample sizes. The stage includes obtaining market information or related data through various sources and strategies. It includes examining and planning all the data acquired from the past in advance. It likewise envelops the examination of information inconsistencies seen across different information sources. The market data is analysed and estimated using market statistical and coherent models. Also, market share analysis and key trend analysis are the major success factors in the market report. To know more, please request an analyst call or drop down your inquiry.
The key research methodology used by DBMR research team is data triangulation which involves data mining, analysis of the impact of data variables on the market and primary (industry expert) validation. Data models include Vendor Positioning Grid, Market Time Line Analysis, Market Overview and Guide, Company Positioning Grid, Patent Analysis, Pricing Analysis, Company Market Share Analysis, Standards of Measurement, Global versus Regional and Vendor Share Analysis. To know more about the research methodology, drop in an inquiry to speak to our industry experts.
Customization Available
Data Bridge Market Research is a leader in advanced formative research. We take pride in servicing our existing and new customers with data and analysis that match and suits their goal. The report can be customized to include price trend analysis of target brands understanding the market for additional countries (ask for the list of countries), clinical trial results data, literature review, refurbished market and product base analysis. Market analysis of target competitors can be analyzed from technology-based analysis to market portfolio strategies. We can add as many competitors that you require data about in the format and data style you are looking for. Our team of analysts can also provide you data in crude raw excel files pivot tables (Fact book) or can assist you in creating presentations from the data sets available in the report.