North America Pharmaceutical Isolator Market
Market Size in USD Billion
CAGR :
% 
 USD
1.74 Billion
USD
5.17 Billion
2025
2033
USD
1.74 Billion
USD
5.17 Billion
2025
2033
| 2026 –2033 | |
| USD 1.74 Billion | |
| USD 5.17 Billion | |
|
|
|
|
North America Pharmaceutical Isolator Market Size
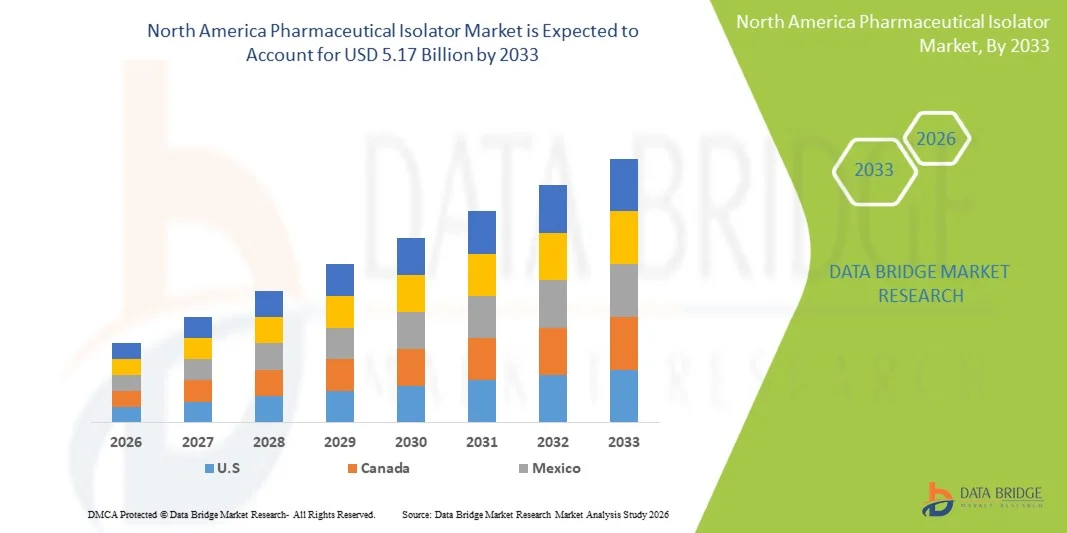
- The North America Pharmaceutical Isolator Market size was valued at USD 1.74 billion in 2025 and is expected to reach USD 5.17 billion by 2033, at a CAGR of 14.60% during the forecast period
- The market growth is largely fueled by increasing demand for sterile and contamination-free environments in pharmaceutical manufacturing, along with stringent regulatory requirements for drug safety and quality, leading to higher adoption of advanced containment solutions
- Furthermore, rising investments in biopharmaceutical production, growing focus on worker safety, and increasing need for aseptic processing are establishing pharmaceutical isolators as critical components in modern drug manufacturing. These converging factors are accelerating the uptake of Pharmaceutical Isolator solutions, thereby significantly boosting the industry's growth
North America Pharmaceutical Isolator Market Analysis
- Pharmaceutical isolators, used to maintain sterile and contamination-free environments, are increasingly vital in pharmaceutical and biopharmaceutical manufacturing due to their role in ensuring product safety, regulatory compliance, and operator protection
- The escalating demand for pharmaceutical isolators is primarily fueled by the growing need for aseptic processing, rising production of biologics and sterile drugs, and increasing focus on contamination control in manufacturing facilities
- The U.S. dominated the North America Pharmaceutical Isolator Market with the largest revenue share of approximately 38.1% in 2025, characterized by advanced pharmaceutical manufacturing infrastructure, strong presence of leading biotechnology companies, and high adoption of sterile processing technologies, with the country witnessing substantial growth driven by increasing investments in biologics and high-potency drug production
- Canada is expected to be the fastest growing country in the North America Pharmaceutical Isolator Market during the forecast period, with a projected CAGR of around 9.0%, due to expanding pharmaceutical and biotechnology industries, increasing focus on regulatory compliance, rising investments in healthcare infrastructure, and growing adoption of advanced manufacturing technologies
- The closed system segment held the largest market revenue share of 58.2% in 2025, driven by its superior contamination control and complete isolation capabilities
Report Scope and North America Pharmaceutical Isolator Market Segmentation
|
Attributes |
Pharmaceutical Isolator Key Market Insights |
|
Segments Covered |
|
|
Countries Covered |
North America
|
|
Key Market Players |
|
|
Market Opportunities |
|
|
Value Added Data Infosets |
In addition to the insights on market scenarios such as market value, growth rate, segmentation, geographical coverage, and major players, the market reports curated by the Data Bridge Market Research also include in-depth expert analysis, patient epidemiology, pipeline analysis, pricing analysis, and regulatory framework. |
North America Pharmaceutical Isolator Market Trends
“Increasing Adoption of Advanced Containment Technologies in Pharmaceutical Manufacturing”
- A significant and accelerating trend in the North America Pharmaceutical Isolator Market is the growing adoption of advanced containment and aseptic processing technologies to ensure product safety and regulatory compliance. These systems are increasingly being utilized to minimize contamination risks during the handling of potent compounds and sterile drug manufacturing
- For instance, pharmaceutical manufacturers in countries such as China and India are increasingly investing in closed isolator systems for aseptic filling and cytotoxic drug handling, ensuring enhanced operator and product protection
- The rising demand for biologics, high-potency active pharmaceutical ingredients (HPAPIs), and sterile injectables is further driving the integration of advanced isolator systems across production facilities
- In addition, the shift toward automation and integration of isolators with robotic systems is improving operational efficiency, reducing human intervention, and enhancing reproducibility in pharmaceutical processes
- This trend is also supported by increasing regulatory scrutiny and the need to comply with international standards such as GMP, which is encouraging manufacturers to upgrade their existing infrastructure
- As a result, the demand for technologically advanced pharmaceutical isolators is growing rapidly across the North America region, particularly in emerging economies with expanding pharmaceutical manufacturing capabilities
North America Pharmaceutical Isolator Market Dynamics
Driver
“Rising Pharmaceutical Manufacturing and Growing Demand for Sterile Drug Production”
- The rapid expansion of pharmaceutical manufacturing activities across the North America region is a major driver for the North America Pharmaceutical Isolator Market, supported by increasing investments in drug production and research
- For instance, the growing production of injectable drugs and vaccines in countries such as India and China has significantly increased the need for aseptic processing environments, thereby boosting the demand for isolator systems
- The increasing prevalence of chronic diseases and infectious conditions is driving the demand for advanced therapeutics, including biologics and biosimilars, which require highly controlled and sterile manufacturing environments
- Furthermore, the rising focus on high-potency drugs, including oncology treatments, is necessitating the use of isolators to ensure safe handling and prevent cross-contamination
- Government initiatives to strengthen domestic pharmaceutical production and reduce dependency on imports are also contributing to the growth of manufacturing facilities, thereby driving demand for isolators
- In addition, the increasing outsourcing of pharmaceutical manufacturing to contract manufacturing organizations (CMOs) in North America is further accelerating the adoption of advanced isolator technologies
Restraint/Challenge
“High Implementation Costs and Operational Complexities”
- The high initial investment required for the installation of advanced pharmaceutical isolator systems remains a key challenge, particularly for small and medium-sized manufacturers in developing economies
- For instance, the cost associated with setting up fully integrated aseptic isolator systems, along with validation and maintenance expenses, can be significantly higher compared to traditional cleanroom setups
- Operational complexities, including the need for specialized training, strict maintenance protocols, and adherence to regulatory standards, further increase the burden on manufacturers
- In addition, integrating isolator systems with existing manufacturing infrastructure can be challenging, especially in older facilities that require significant upgrades
- Ensuring continuous compliance with evolving regulatory requirements across different countries in the North America region also adds to the complexity of operations
- Addressing these challenges through cost optimization, technological advancements, and increased awareness of long-term benefits will be essential for driving broader adoption of pharmaceutical isolators in the region
North America Pharmaceutical Isolator Market Scope
The market is segmented on the basis of type, system type, pressure, configuration, application, end user, and distribution channel.
• By Type
On the basis of type, the North America Pharmaceutical Isolator Market is segmented into aseptic isolators, containment isolators, bio isolators, sampling and weighing isolators, active pharmaceutical ingredient (API) manufacturing isolators, radiopharmaceutical isolators, production isolators, and others. The aseptic isolators segment dominated the largest market revenue share of 34.7% in 2025, driven by increasing demand for sterile manufacturing environments across pharmaceutical industries. Aseptic isolators are critical in preventing microbial contamination during drug production. Rising demand for biologics and injectable drugs significantly supports adoption. Stringent regulatory frameworks regarding sterility assurance further enhance usage. Increasing investments in cleanroom technologies boost segment growth. Expansion of global pharmaceutical manufacturing capacities strengthens demand. Technological advancements improving automation and efficiency further support adoption. Growing emphasis on patient safety and product quality drives utilization. Increased outsourcing of sterile manufacturing enhances reliance on isolators. Continuous innovation in isolator design improves flexibility and performance. Rising adoption in vaccine production also contributes to growth. These factors collectively ensure the dominance of the aseptic isolators segment.
The containment isolators segment is expected to witness the fastest CAGR of 9.6% from 2026 to 2033, driven by increasing need for safe handling of high-potency active pharmaceutical ingredients (HPAPIs). Growing focus on occupational safety and regulatory compliance boosts demand. Rising production of oncology and cytotoxic drugs further supports adoption. Increasing awareness regarding hazardous drug exposure enhances usage. Expansion of pharmaceutical outsourcing and contract manufacturing strengthens growth. Technological advancements in containment systems improve efficiency and reliability. Growing pipeline of potent drugs fuels long-term demand. Increasing investments in advanced manufacturing facilities further accelerate adoption. Enhanced operator protection and environmental safety regulations drive utilization. Rising need for controlled environments in research laboratories supports growth. Continuous innovation in isolator technology strengthens performance. These factors position containment isolators as the fastest-growing segment.
• By System Type
On the basis of system type, the North America Pharmaceutical Isolator Market is segmented into closed system and open system. The closed system segment held the largest market revenue share of 58.2% in 2025, driven by its superior contamination control and complete isolation capabilities. Closed systems are widely used in sterile drug manufacturing and handling hazardous materials. Increasing regulatory emphasis on contamination prevention boosts adoption. Rising production of biologics and high-value drugs supports demand. Enhanced safety for both product and operator further strengthens usage. Expansion of pharmaceutical production facilities increases deployment. Growing preference for automated and controlled environments enhances growth. Technological advancements improving system efficiency contribute significantly. Increasing global clinical and commercial manufacturing activities boost demand. Strong compliance with international safety standards ensures widespread adoption. Rising need for maintaining sterility in advanced therapies further supports growth. These factors ensure dominance of the closed system segment.
The open system segment is expected to witness the fastest CAGR of 8.7% from 2026 to 2033, driven by its flexibility and cost-effectiveness in selected applications. Open systems provide easier accessibility for manual interventions. Increasing adoption in research laboratories and small-scale manufacturing units supports growth. Growing demand for adaptable and less complex systems enhances usage. Technological improvements improving safety features boost adoption. Rising investments in R&D facilities strengthen demand. Expansion of academic research activities contributes to growth. Increasing customization capabilities enhance usability. Demand for hybrid solutions combining flexibility and safety further supports expansion. Continuous innovation in system design improves efficiency. Growing acceptance in emerging markets boosts adoption. These factors position open systems as the fastest-growing segment.
• By Pressure
On the basis of pressure, the North America Pharmaceutical Isolator Market is segmented into positive pressure and negative pressure. The positive pressure segment dominated the largest market revenue share of 54.6% in 2025, driven by its extensive application in aseptic processing and sterile drug production. Positive pressure systems prevent ingress of contaminants, ensuring product safety. Increasing demand for sterile injectable drugs supports growth. Rising regulatory requirements for contamination control enhance adoption. Expansion of biologics manufacturing strengthens demand. Growing emphasis on high-quality pharmaceutical output further boosts usage. Technological advancements improving pressure control systems support efficiency. Increasing production of vaccines and biosimilars contributes to growth. Strong demand from large-scale manufacturing facilities enhances adoption. Rising investments in pharmaceutical infrastructure further support expansion. Continuous innovation in isolator systems improves reliability. These factors ensure dominance of the positive pressure segment.
The negative pressure segment is expected to witness the fastest CAGR of 9.1% from 2026 to 2033, driven by increasing need for safe handling of hazardous and toxic substances. Negative pressure isolators protect operators and the environment from exposure. Rising production of high-potency and cytotoxic drugs supports demand. Growing regulatory focus on workplace safety enhances adoption. Increasing awareness regarding hazardous drug handling boosts growth. Expansion of oncology drug manufacturing strengthens demand. Technological advancements improving containment efficiency support adoption. Rising investments in specialized manufacturing facilities further drive growth. Growing use in research and diagnostic laboratories enhances demand. Continuous development of advanced containment technologies improves performance. These factors position negative pressure as the fastest-growing segment.
• By Configuration
On the basis of configuration, the North America Pharmaceutical Isolator Market is segmented into floor standing, modular, mobile, compact, table top, portable, and others. The floor standing segment accounted for the largest market revenue share of 32.4% in 2025, driven by its widespread use in large-scale pharmaceutical production environments. These systems offer high capacity and robust operational efficiency. Increasing industrial-scale drug manufacturing supports demand. Expansion of pharmaceutical infrastructure globally enhances adoption. Rising need for stable and permanent installations boosts usage. Growing production of biologics strengthens demand. Technological advancements improving system durability support growth. Increasing investments in manufacturing plants contribute significantly. Strong preference in large facilities ensures continued dominance. Rising adoption in vaccine production facilities enhances usage. Continuous improvements in design and automation further support growth. These factors ensure dominance of the floor standing segment.
The modular segment is expected to witness the fastest CAGR of 10.3% from 2026 to 2033, driven by flexibility, scalability, and ease of customization. Modular systems allow quick installation and expansion. Increasing demand for adaptable manufacturing solutions supports growth. Rising investments in flexible production facilities boost adoption. Growing need for cost-efficient and scalable systems enhances demand. Expansion of contract manufacturing organizations strengthens growth. Technological advancements improving modular integration support adoption. Increasing focus on rapid deployment capabilities enhances usage. Growing adoption in emerging markets further boosts demand. Continuous innovation in modular design improves performance. Rising preference for customized solutions strengthens the segment. These factors position modular configuration as the fastest-growing segment.
• By Application
On the basis of application, the North America Pharmaceutical Isolator Market is segmented into sterility testing, manufacturing, sampling/weighing/distribution, and medical device manufacturing. The manufacturing segment held the largest market revenue share of 44.1% in 2025, driven by increasing pharmaceutical production activities worldwide. Isolators are essential for maintaining sterile environments during drug manufacturing. Rising demand for biologics and injectable drugs supports growth. Expansion of global pharmaceutical production enhances adoption. Increasing regulatory requirements for product safety boost usage. Growing investments in manufacturing infrastructure strengthen demand. Technological advancements improving production efficiency support growth. Rising adoption in vaccine manufacturing further enhances demand. Strong reliance on isolators for contamination control ensures dominance. Continuous growth in pharmaceutical pipelines boosts manufacturing needs. Increasing outsourcing of production further supports segment growth. These factors ensure dominance of the manufacturing segment.
The sterility testing segment is expected to witness the fastest CAGR of 9.8% from 2026 to 2033, driven by stringent regulatory requirements for quality assurance. Increasing focus on product safety and compliance supports growth. Rising pharmaceutical R&D activities enhance testing demand. Growing adoption of advanced testing technologies boosts usage. Expansion of biologics and biosimilars increases testing needs. Increasing regulatory audits and inspections strengthen demand. Technological advancements improving testing accuracy support growth. Rising need for contamination-free validation enhances adoption. Growing investments in laboratory infrastructure further drive growth. Continuous innovation in testing methodologies improves efficiency. These factors position sterility testing as the fastest-growing segment.
• By End User
On the basis of end user, the North America Pharmaceutical Isolator Market is segmented into hospitals, diagnostic laboratories, academic and research institutes, pharmaceutical and biotechnology companies, contract research organizations, and others. The pharmaceutical and biotechnology companies segment dominated the largest market revenue share of 49.3% in 2025, driven by extensive use of isolators in drug development and manufacturing. Increasing R&D investments significantly support growth. Rising demand for biologics and specialty drugs enhances adoption. Expansion of pharmaceutical pipelines boosts demand. Growing focus on contamination control strengthens usage. Increasing global manufacturing activities further support growth. Technological advancements improving isolator performance enhance adoption. Rising collaborations and partnerships drive demand. Continuous innovation in drug development supports usage. Strong reliance on isolators ensures segment dominance. These factors collectively ensure leadership of this segment.
The contract research organizations segment is expected to witness the fastest CAGR of 10.7% from 2026 to 2033, driven by increasing outsourcing of pharmaceutical research and manufacturing activities. CROs offer cost-effective and efficient solutions. Growing complexity of drug development supports outsourcing trends. Rising demand for faster clinical trial execution enhances adoption. Expansion of global clinical research activities boosts growth. Increasing partnerships between pharma companies and CROs strengthen demand. Technological advancements improving research capabilities support growth. Rising investments in CRO infrastructure further enhance adoption. Growing focus on efficiency and cost reduction drives outsourcing. Continuous expansion of CRO services boosts market presence. These factors position CROs as the fastest-growing segment.
• By Distribution Channel
On the basis of distribution channel, the North America Pharmaceutical Isolator Market is segmented into direct tender, retail sales, and third party distributors. The direct tender segment dominated the largest market revenue share of 51.8% in 2025, driven by bulk procurement by pharmaceutical companies and research organizations. Direct tenders ensure cost efficiency and long-term supply agreements. Increasing large-scale manufacturing projects support demand. Strong supplier relationships enhance procurement efficiency. Rising investments in pharmaceutical infrastructure boost growth. Increasing demand for customized solutions supports adoption. Expansion of global manufacturing networks further strengthens demand. Continuous supply requirements ensure stability. Growing preference for direct sourcing enhances control and quality. These factors ensure dominance of the direct tender segment.
The third party distributors segment is expected to witness the fastest CAGR of 8.9% from 2026 to 2033, driven by expanding global distribution networks and increasing demand from emerging markets. Third party distributors enhance product accessibility and availability. Growing need for efficient supply chain solutions supports growth. Increasing presence in developing regions boosts adoption. Rising demand from small and mid-sized enterprises strengthens the segment. Technological advancements improving logistics efficiency support growth. Expansion of e-commerce and digital platforms enhances reach. Increasing partnerships between manufacturers and distributors further drive demand. Continuous improvement in distribution infrastructure boosts efficiency. Growing globalization of pharmaceutical markets enhances adoption. These factors position third party distributors as the fastest-growing segment.
North America Pharmaceutical Isolator Market Regional Analysis
- The North America Pharmaceutical Isolator Market is poised to grow at a substantial CAGR during the forecast period of 2026 to 2033, driven by the rapid expansion of pharmaceutical and biotechnology industries, increasing healthcare investments, and rising demand for sterile and contamination-free production environments across the region
- The region’s growth is further supported by stringent regulatory frameworks, increasing adoption of Good Manufacturing Practices (GMP), and rising production of biologics, vaccines, and high-potency drugs
- In addition, North America is a key hub for advanced pharmaceutical manufacturing, significantly boosting the demand for high-performance isolator systems to ensure product safety and regulatory compliance
U.S. North America Pharmaceutical Isolator Market Insight
The U.S. North America Pharmaceutical Isolator Market dominated with the largest revenue share of approximately 38.1% in 2025, characterized by advanced pharmaceutical manufacturing infrastructure, strong presence of leading biotechnology companies, and high adoption of sterile processing technologies. The country is witnessing substantial growth driven by increasing investments in biologics and high-potency drug production, along with continuous technological advancements in aseptic processing systems.
Canada North America Pharmaceutical Isolator Market Insight
The Canada North America Pharmaceutical Isolator Market is expected to be the fastest growing, with a projected CAGR of around 9.0% during the forecast period. This growth is driven by expanding pharmaceutical and biotechnology industries, increasing focus on regulatory compliance, and rising investments in healthcare infrastructure. In addition, growing adoption of advanced manufacturing technologies and increasing emphasis on sterile production environments are further supporting the rapid growth of the market in Canada.
North America Pharmaceutical Isolator Market Share
The Pharmaceutical Isolator industry is primarily led by well-established companies, including:
- Getinge AB (Sweden)
- SKAN AG (Switzerland)
- Azbil Corporation (Japan)
- Comecer S.p.A. (Italy)
- Bioquell (U.K.)
- Hosokawa Micron Group (Japan)
- Extract Technology Ltd. (U.K.)
- Fedegari Autoclavi S.p.A. (Italy)
- Syntegon Technology GmbH (Germany)
- Tofflon Science and Technology Group Co., Ltd. (China)
- NuAire, Inc. (U.S.)
- Germfree Laboratories, Inc. (U.S.)
- ITECO Engineering (Italy)
- Labconco Corporation (U.S.)
- MBRAUN GmbH (Germany)
- Esco Lifesciences Group (Singapore)
- Jacomex (France)
- Weiss Technik (Germany)
- Ortner Reinraumtechnik GmbH (Austria)Angelantoni Life Science (Italy)
Latest Developments in North America Pharmaceutical Isolator Market
- In April 2021, ILC Dover launched its SoloPure Flexible Aseptic Isolator, designed to enhance sterile pharmaceutical manufacturing with improved flexibility and contamination control. The system supports safer handling of sensitive drugs and reflects growing adoption of advanced isolator technologies in pharmaceutical production
- In October 2022, Tema Sinergie introduced a new four-glove pharmaceutical isolator specifically designed for sterile powder sampling and handling of large containers. This launch improved operational precision and contamination control in pharmaceutical and biopharmaceutical manufacturing environments
- In July 2023, SKAN Group AG acquired an 80% stake in Aseptic Technologies to strengthen its aseptic processing and isolator capabilities. This strategic move enhanced SKAN’s portfolio in sterile manufacturing solutions and reinforced its position in advanced pharmaceutical isolator technologies
- In March 2024, Telstar launched a dual-mode pharmaceutical isolator system capable of operating for both containment and aseptic applications. This innovation provides increased flexibility for pharmaceutical manufacturers and supports evolving production requirements in biologics and high-potency drugs
SKU-
Get online access to the report on the World's First Market Intelligence Cloud
- Interactive Data Analysis Dashboard
- Company Analysis Dashboard for high growth potential opportunities
- Research Analyst Access for customization & queries
- Competitor Analysis with Interactive dashboard
- Latest News, Updates & Trend analysis
- Harness the Power of Benchmark Analysis for Comprehensive Competitor Tracking
Research Methodology
Data collection and base year analysis are done using data collection modules with large sample sizes. The stage includes obtaining market information or related data through various sources and strategies. It includes examining and planning all the data acquired from the past in advance. It likewise envelops the examination of information inconsistencies seen across different information sources. The market data is analysed and estimated using market statistical and coherent models. Also, market share analysis and key trend analysis are the major success factors in the market report. To know more, please request an analyst call or drop down your inquiry.
The key research methodology used by DBMR research team is data triangulation which involves data mining, analysis of the impact of data variables on the market and primary (industry expert) validation. Data models include Vendor Positioning Grid, Market Time Line Analysis, Market Overview and Guide, Company Positioning Grid, Patent Analysis, Pricing Analysis, Company Market Share Analysis, Standards of Measurement, Global versus Regional and Vendor Share Analysis. To know more about the research methodology, drop in an inquiry to speak to our industry experts.
Customization Available
Data Bridge Market Research is a leader in advanced formative research. We take pride in servicing our existing and new customers with data and analysis that match and suits their goal. The report can be customized to include price trend analysis of target brands understanding the market for additional countries (ask for the list of countries), clinical trial results data, literature review, refurbished market and product base analysis. Market analysis of target competitors can be analyzed from technology-based analysis to market portfolio strategies. We can add as many competitors that you require data about in the format and data style you are looking for. Our team of analysts can also provide you data in crude raw excel files pivot tables (Fact book) or can assist you in creating presentations from the data sets available in the report.