North America Rapid Diagnostic Tests Rdt Market
Market Size in USD Million
CAGR :
% 
 USD
13,942.76 Million
USD
29,455.56 Million
2022
2030
USD
13,942.76 Million
USD
29,455.56 Million
2022
2030
| 2023 - 2030 | |
| USD 13,942.76 Million | |
| USD 29,455.56 Million | |
|
|
|
North America Rapid Diagnostic Tests (RDT) Market Analysis and Size
The rapid diagnostic tests, also called rapid tests, are easy-to-use tests that offer quick results, usually in less than 20 minutes. Unlike other standard and conventional tests, where the diagnosis and sampling are sent to the laboratory, the results obtained from rapid diagnostic test kits are provided at the point of care. The point of care is a place where the patients get cured. The point of care can be a provider's office, a clinic, or a patient's own house. The RDTs are applicable for diagnosing and testing infectious diseases such as COVID-19, cardiology diseases, oncology, pregnancy, fertility testing, toxicology testing, drugs-of-abuse testing, cardiometabolic testing, and glucose monitoring.
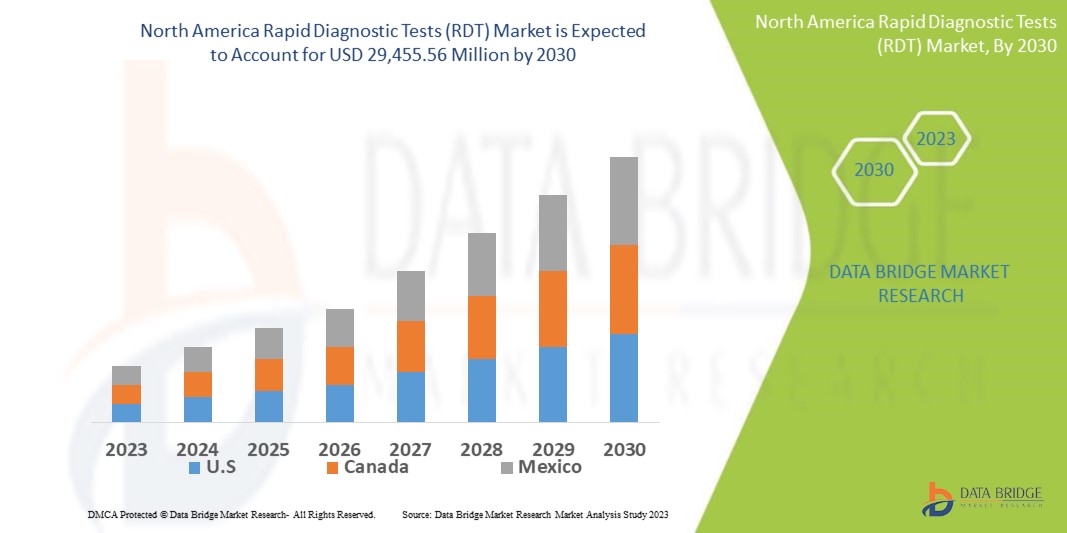
Data Bridge Market Research analyses that the North America rapid diagnostic tests (RDT) market which was USD 13,942.76 million in 2022, is expected to reach USD 29,455.56 million by 2030, and is expected to undergo a CAGR of 9.80% during the forecast period. “Consumable and Kits” dominates the product type segment of the North America rapid diagnostic tests (RDT) market owing to accuracy of the results offered by these products when rapid diagnosis is required. In addition to the insights on market scenarios such as market value, growth rate, segmentation, geographical coverage, and major players, the market reports curated by the Data Bridge Market Research also include depth expert analysis, patient epidemiology, pipeline analysis, pricing analysis, and regulatory framework.
North America Rapid Diagnostic Tests (RDT) Market Scope and Segmentation
|
Report Metric |
Details |
|
Forecast Period |
2023 to 2030 |
|
Base Year |
2022 |
|
Historic Years |
2021 (Customizable to 2015-2020) |
|
Quantitative Units |
Revenue in USD Million, Volumes in Units, Pricing in USD |
|
Segments Covered |
Product Type (Consumables and Kits, Instruments and Others), Mode (Professional Rapid Diagnostic Test Product and Over-The-Counter [OTC] Rapid Diagnostic Test Product), Technology (PCR-Based, Flow-Through Assays, Lateral Flow Immunochromatographic Assays, Agglutination Assay, Microfluidics, Substrate Technology and Others), Modality (Laboratory Based Test and Non-Laboratory Based Test), Age Group (Adult and Pediatric), Test Type (Determining Confirmation, Serological Testing and Viral Sequencing), Approach (In-Vitro Diagnostic, Molecular Diagnostic), Specimen (Swab, Blood, Urine, Saliva, Sputum and Others), Application (Infectious Disease Testing, Glucose Monitoring, Cardiology Testing, Oncology Testing, Cardiometabolic Testing, Drugs-of-Abuse Testing, Pregnancy & Fertility Testing, Toxicology Testing, Others), End User (Hospital & Clinic, Diagnostic Laboratory, Home Care Setting, Research and Academic Institutes and Others), Distribution Channel (Direct Tender, Retail Sales, Others) |
|
Countries Covered |
U.S., Canada and Mexico in North America |
|
Market Players Covered |
Abbott (U.S.), Danaher (U.S.), Cellex (U.S.), AdvaCare Pharma (U.S.), Access Bio (U.S.), Cardinal Health (U.S.), Bio-Rad Laboratories, Inc. (U.S.), BD (U.S.), F. Hoffmann-La Roche Ltd (Switzerland), bioMérieux SA (France), InBios International, Inc. (U.S.), Gnomegen LLC (U.S.), QIAGEN (Germany), Quidel Corporation (U.S.), Chembio Diagnostics Systems, Inc. (U.S.), Siemens Healthcare Gmbh (Germany), PerkinElmer Inc. (U.S.), Sekisui Diagnostics (U.S.), Fujirebio (Japan), PTS Diagnostics (U.S.), LamdaGen Corporation (U.S.), Werfen (Spain), Nova Biomedical (U.S.), Trinity Biotech (Ireland), Sysmex Europe GmbH (Germany), Luminex Corporation (U.S.) and DiaSorin (Italy) |
|
Market Opportunities |
|
Market Definition
Rapid diagnostic tests (RDTs) are quick, point-of-care diagnostic tools designed to deliver fast results, often within minutes, without the need for extensive laboratory infrastructure. These tests are commonly used for the prompt detection of infectious diseases, such as COVID-19, malaria, HIV, and various other conditions. RDTs typically involve simple procedures, such as lateral flow assays or immunoassays, and are employed in diverse settings, including clinics, emergency rooms, and at-home applications. Their speed and ease of use make them valuable for early diagnosis, enabling timely intervention and improving healthcare accessibility, particularly in resource-limited or remote areas.
North America Rapid Diagnostic Tests (RDT) Market Dynamics
- Rising Prevalence of Infectious Diseases
The increasing prevalence of infectious diseases, including the North America impact of pandemics like COVID-19, propels the demand for rapid diagnostic tests. RDTs play a crucial role in the swift identification of infectious agents, enabling timely containment measures, treatment initiation, and public health interventions.
- Advancements in Technology
Ongoing technological advancements contribute to the refinement of rapid diagnostic tests, enhancing their accuracy and reliability. Innovations in lateral flow assays, molecular diagnostics, and biosensor technologies ensure that RDTs provide sensitive and specific results, fostering confidence in their diagnostic capabilities and expanding their utility across various medical conditions.
Opportunities
- Chronic Disease Management
Rapid diagnostic tests in chronic disease management offer opportunities for quick and convenient monitoring of conditions like diabetes and cardiovascular diseases. Innovative tests can provide valuable data for healthcare professionals, aiding in personalized treatment plans and improving patient outcomes. The growth of this segment aligns with the broader trend of shifting healthcare towards proactive and preventive strategies.
- Rising Remote and Rural Healthcare
In remote and rural healthcare settings, rapid diagnostic tests play a pivotal role by offering accessible and timely diagnostic information. These tests can be deployed in areas with limited laboratory infrastructure, enabling healthcare providers to quickly assess and address health issues. Improved access to diagnostic capabilities contributes to more equitable healthcare delivery and better health outcomes in underserved regions.
Restraints/Challenges
- Affordability and Accessibility
While rapid diagnostic tests are generally more cost-effective than some traditional laboratory methods, affordability remains a concern, particularly in resource-limited settings. Ensuring accessibility to these tests, especially in low-income regions, is a challenge.
This North America rapid diagnostic tests (RDT) market report provides details of new recent developments, trade regulations, import-export analysis, production analysis, value chain optimization, market share, impact of domestic and localized market players, analyses opportunities in terms of emerging revenue pockets, changes in market regulations, strategic market growth analysis, market size, category market growths, application niches and dominance, product approvals, product launches, geographic expansions, technological innovations in the market. To gain more info on the North America rapid diagnostic tests (RDT) market Contact Data Bridge Market Research for an Analyst Brief, our team will help you take an informed market decision to achieve market growth.
Recent Developments
- In August 2021, Cardinal Health partnered with Abbott and Quidel Corporation to commercialize the over-the-counter (OTC) rapid COVID-19 tests. The partnership would allow Cardinal Health to expand Cardinal Health's COVID-19 testing and surveillance offerings and be able to access Quidel's QuickVue At-Home OTC COVID-19 Test and Abbott's BinaxNOW COVID-19 Antigen Self-Test, which allow the patients to perform tests without prescription easily
- In February 2020, BD announced a long-term strategic partnership agreement to bring laboratory-quality, small-volume blood collection to retail pharmacies with Babson Diagnostics. This helped both the company in improving their range of quality services
North America Rapid Diagnostic Tests (RDT) Market Scope
The North America rapid diagnostic tests (RDT) market is segmented on the basis of product type, mode technology, modality, age group, test type, approach, specimen, application, end user, and distribution channel. The growth amongst these segments will help you analyze meagre growth segments in the industries and provide the users with a valuable market overview and market insights to help them make strategic decisions for identifying core market applications.
Product Type
- Consumables and Kits
- Instruments
- Others
Mode
- Professional Rapid Diagnostic Test Product
- Over-The-Counter [OTC] Rapid Diagnostic Test Product
Technology
- PCR-Based
- Flow-Through Assays
- Lateral Flow Immunochromatographic Assays
- Agglutination Assay
- Microfluidics
- Substrate Technology
- Others
Modality
- Laboratory Based Test
- Non-Laboratory Based Test
Age Group
- Adult
- Pediatric
Test Type
- Determining Confirmation
- Serological Testing
- Viral Sequencing
Approach
- In-Vitro Diagnostic
- Molecular Diagnostic
Specimen
- Swab
- Blood
- Urine
- Saliva
- Sputum
- Others
Application
- Infectious Disease Testing
- Glucose Monitoring
- Cardiology Testing
- Oncology Testing
- Cardiometabolic Testing
- Drugs-of-Abuse Testing
- Pregnancy & Fertility Testing
- Toxicology Testing
- Others
End User
- Hospital & Clinic
- Diagnostic Laboratory
- Home Care Setting
- Research and Academic Institutes
- Others
Distribution Channel
- Direct Tender
- Retail Sales
- Others
North America Rapid diagnostic tests (RDT) Market Regional Analysis/Insights
The North America rapid diagnostic tests (RDT) market is analysed and market size insights and trends are provided by product type, mode technology, modality, age group, test type, approach, specimen, application, end user, and distribution channel as referenced above.
The countries covered in the North America rapid diagnostic tests (RDT) market report are U.S., Canada and Mexico in North America.
The U.S. is expected to dominate with highest market share and CAGR due to the presence of the largest consumer market with high GDP. Moreover, the U.S. has the highest household spending North Americaly. It offers trade agreements with several countries making it the largest market for consumer products, including rapid diagnostic tests due to major market players and increased technological advancements in the region.
The country section of the report also provides individual market impacting factors and changes in regulation in the market domestically that impacts the current and future trends of the market. Data points like down-stream and upstream value chain analysis, technical trends and porter's five forces analysis, case studies are some of the pointers used to forecast the market scenario for individual countries. Also, the presence and availability of North America brands and their challenges faced due to large or scarce competition from local and domestic brands, impact of domestic tariffs and trade routes are considered while providing forecast analysis of the country data.
Healthcare Infrastructure Growth Installed Base and New Technology Penetration
The North America rapid diagnostic tests (RDT) market also provides you with detailed market analysis for every country growth in healthcare expenditure for capital equipment, installed base of different kind of products for North America rapid diagnostic tests (RDT) market, impact of technology using life line curves and changes in healthcare regulatory scenarios and their impact on the North America rapid diagnostic tests (RDT) market. The data is available for historic period 2015-2020.
Competitive Landscape and North America Rapid Diagnostic Tests (RDT) Market Share Analysis
The North America rapid diagnostic tests (RDT) market competitive landscape provides details by competitor. Details included are company overview, company financials, revenue generated, market potential, investment in research and development, new market initiatives, North America presence, production sites and facilities, production capacities, company strengths and weaknesses, product launch, product width and breadth, application dominance. The above data points provided are only related to the companies' focus related to North America rapid diagnostic tests (RDT) market.
Some of the major players operating in the North America rapid diagnostic tests (RDT) market are:
- Abbott (U.S.)
- Danaher (U.S.)
- Cellex (U.S.)
- AdvaCare Pharma (U.S.)
- Access Bio (U.S.)
- Cardinal Health (U.S.)
- Bio-Rad Laboratories, Inc. (U.S.)
- BD (U.S.)
- F. Hoffmann-La Roche Ltd (Switzerland)
- bioMérieux SA (France)
- InBios International, Inc. (U.S.)
- Gnomegen LLC (U.S.)
- QIAGEN (Germany)
- Quidel Corporation (U.S.)
- Chembio Diagnostics Systems, Inc. (U.S.)
- Siemens Healthcare Gmbh (Germany)
- PerkinElmer Inc. (U.S.)
- Sekisui Diagnostics (U.S.)
- Fujirebio (Japan)
- PTS Diagnostics (U.S.)
- LamdaGen Corporation (U.S.)
- Werfen (Spain)
- Nova Biomedical (U.S.)
- Trinity Biotech (Ireland)
- Sysmex Europe GmbH (Germany)
- Luminex Corporation (U.S.)
- DiaSorin (Italy)
SKU-
Get online access to the report on the World's First Market Intelligence Cloud
- Interactive Data Analysis Dashboard
- Company Analysis Dashboard for high growth potential opportunities
- Research Analyst Access for customization & queries
- Competitor Analysis with Interactive dashboard
- Latest News, Updates & Trend analysis
- Harness the Power of Benchmark Analysis for Comprehensive Competitor Tracking
Research Methodology
Data collection and base year analysis are done using data collection modules with large sample sizes. The stage includes obtaining market information or related data through various sources and strategies. It includes examining and planning all the data acquired from the past in advance. It likewise envelops the examination of information inconsistencies seen across different information sources. The market data is analysed and estimated using market statistical and coherent models. Also, market share analysis and key trend analysis are the major success factors in the market report. To know more, please request an analyst call or drop down your inquiry.
The key research methodology used by DBMR research team is data triangulation which involves data mining, analysis of the impact of data variables on the market and primary (industry expert) validation. Data models include Vendor Positioning Grid, Market Time Line Analysis, Market Overview and Guide, Company Positioning Grid, Patent Analysis, Pricing Analysis, Company Market Share Analysis, Standards of Measurement, Global versus Regional and Vendor Share Analysis. To know more about the research methodology, drop in an inquiry to speak to our industry experts.
Customization Available
Data Bridge Market Research is a leader in advanced formative research. We take pride in servicing our existing and new customers with data and analysis that match and suits their goal. The report can be customized to include price trend analysis of target brands understanding the market for additional countries (ask for the list of countries), clinical trial results data, literature review, refurbished market and product base analysis. Market analysis of target competitors can be analyzed from technology-based analysis to market portfolio strategies. We can add as many competitors that you require data about in the format and data style you are looking for. Our team of analysts can also provide you data in crude raw excel files pivot tables (Fact book) or can assist you in creating presentations from the data sets available in the report.