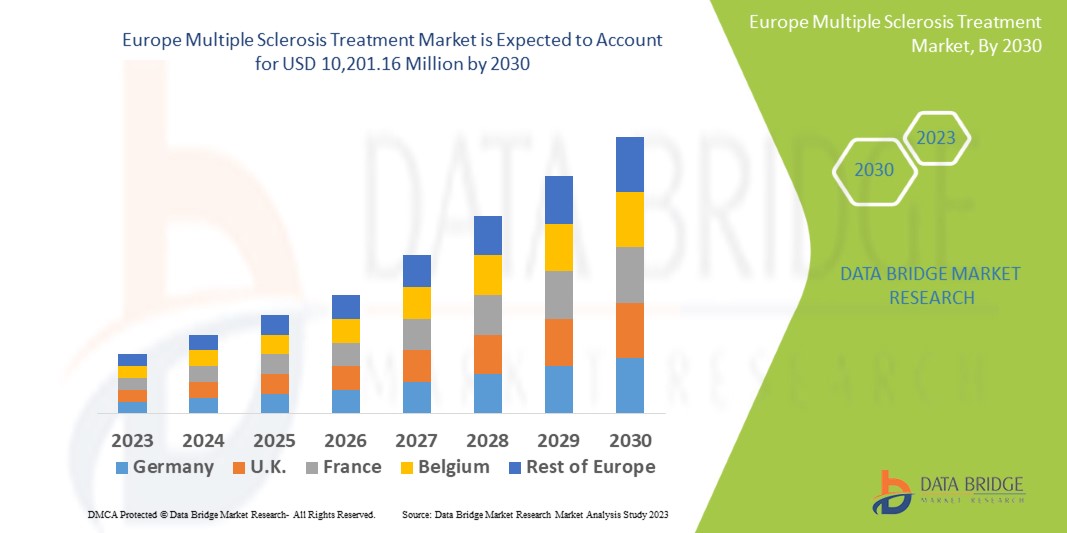
Europe Multiple Sclerosis Treatment Market
市场规模(十亿美元)
CAGR :
% 
 USD
6,026.70 Million
USD
10,201.16 Million
2022
2030
USD
6,026.70 Million
USD
10,201.16 Million
2022
2030
| 2023 –2030 | |
| USD 6,026.70 Million | |
| USD 10,201.16 Million | |
|
|
|
|
歐洲多發性硬化症治療市場,疾病類型(復發緩解型多發性硬化症 (RRMS)、繼發進展型多發性硬化症 (SPMS)、原發性進展型多發性硬化症 (PPMS)、重度復發緩解型多發性硬化症 (RES)),治療(預防性治療、終止性治療/急性治療、對症治療),藥物類型、品牌、實驗室、藥物和診所 20 年的產業趨勢
歐洲多發性硬化症治療市場分析及規模
多種傳染病對全球醫療衛生系統帶來的日益沉重的負擔是一個主要考慮因素,其中發展中國家承受的負擔尤其嚴重。根據印度肝膽科學研究所統計,全球約有3%的人口感染了C型肝炎病毒。丙型肝炎病毒感染率的全球性上升,尤其是在已開發國家和發展中國家,是推動多發性硬化症治療市場成長的主要因素之一。
根據Data Bridge Market Research分析,多發性硬化症治療市場預計到2030年將達到102.0116億美元,而2022年為60.267億美元,預計在2022年至2030年的預測期內,複合年增長率(CAGR)將達到6.8%。除了市場價值、成長率、市場區隔、地理覆蓋範圍和主要參與者等市場概況外,Data Bridge Market Research發布的市場報告還包括深入的專家分析、病患流行病學、研發管線分析、定價分析和監管框架分析。
歐洲多發性硬化症治療市場範圍和細分
|
報告指標 |
細節 |
|
預測期 |
2023年至2030年 |
|
基準年 |
2022 |
|
歷史年代 |
2021年(可自訂為2015-2020年) |
|
定量單位 |
營收單位:百萬美元;銷售單位:台;價格單位:美元 |
|
涵蓋部分 |
疾病類型(復發緩解型多發性硬化症 (RRMS)、繼發性進展型多發性硬化症 (SPMS)、原發性進展型多發性硬化症 (PPMS)、重度復發緩解型多發性硬化症 (RES))、治療(預防性治療、急性惡化治療/終止性治療、對症治療)、藥物類型(品牌藥、仿製藥)、最終使用者(醫院、手術中心) |
|
覆蓋國家/地區 |
德國、法國、英國、荷蘭、瑞士、比利時、俄羅斯、義大利、西班牙、土耳其、歐洲其他國家 |
|
涵蓋的市場參與者 |
艾伯維公司(美國)、博士倫公司(加拿大)、百奧拉製藥公司(美國)、勃林格殷格翰國際有限公司(德國)、安進公司(美國)、輝瑞公司(美國)、羅氏公司(瑞士)、邁蘭公司(美國)、諾華公司(瑞士)、拜耳公司(德國)、百時美施貴寶公司(美國)、百健公司(美國)、梯瓦製藥工業有限公司(以色列)、武田藥品工業株式會社(日本)、Jazz Pharmaceuticals公司(英國)、雅培公司(美國)、Bio-Rad Laboratories公司(美國)、邁蘭公司(美國) |
|
市場機遇 |
|
市場定義
多發性硬化症(MS)是一種慢性發炎性免疫介導疾病,可導致脊髓和腦神經細胞的軸突斷裂、脫髓鞘和神經退化。常用的治療方法包括注射、口服和輸注藥物、抗憂鬱藥物、物理治療和肌肉鬆弛劑。
歐洲多發性硬化症治療市場動態
司機
- 人們對多發性硬化症的認識不斷提高
人們對多發性硬化症的認識不斷提高,有助於推動市場成長。例如,多發性硬化症基金會及其附屬機構自2003年起,每年三月設立“全國多發性硬化症教育和宣傳月”,旨在提高公眾對該疾病的認識。該計畫的主要目標是增進大眾對多發性硬化症的了解,並幫助患者在治療方面做出明智的決定。此外,美國國家多發性硬化症協會發起並實施了「MS Kills Connection」(多發性硬化症致死:連接治癒多發性硬化症)活動,旨在連接多發性硬化症患者、多發性硬化症患者的醫護人員以及致力於創造一個沒有多發性硬化症的世界的人士。因此,這項因素也促進了市場成長。
- 多發性硬化症發病率不斷上升
該地區多發性硬化症的發生率呈上升趨勢。根據英國國家多發性硬化症協會的研究,2010年該地區多發性硬化症的盛行率約為每10萬人309例,成人多發性硬化症患者總數達727,344例。歐洲被認為是多發性硬化症的高發生率地區。英國多發性硬化症協會指出,2018年英國約有11萬人患有此病。此外,該地區女性多發性硬化症的發生率是男性的兩倍多。每年新增確診患者人數也不斷增加,從2017年的5,000人增加到2018年的6,700人。因此,此發病率的上升推高了對治療藥物的需求。
機會
- 各組織加強研發活動力度
目前,許多藥物正處於治療多發性硬化症(MS)的研究階段,其中許多藥物正在進行臨床試驗。美國食品藥物管理局(FDA)已批准幾種用於治療MS的藥物,包括芬戈莫德、幹擾素、那他珠單抗、醋酸格拉替雷、特立氟胺和富馬酸二甲酯。一些遺傳學家和其他研究人員正在研究人類基因組,以尋找肌肉營養不良症、阿茲海默症、癌症和糖尿病等疾病的病因和其他可能的治療方法。例如,他們發現攜帶特定基因的人更容易罹患阿茲海默症。因此,研究人員正在研發能夠修飾或修復該特定基因的藥物。同樣,許多研究人員發現,一些多發性硬化症患者攜帶某種基因,使他們更容易患上這種疾病。因此,他們正在持續研發能夠改變或修復該基因的藥物。
限制/挑戰
- 藥品價格高昂
儘管多發性硬化症的盛行率很高,但藥物價格仍是限制市場成長的一大挑戰。高昂的藥價使得滿足患者群體的需求變得效率低。疾病修飾療法(DMTs)是標準的治療方案,但這些藥物的極高價格導致患者依從性差,疾病管理不善。據美國國家多發性硬化症協會稱,多發性硬化症藥物的價格正在大幅上漲。預計這一因素將限制市場成長。
這份多發性硬化症治療市場報告詳細介紹了近期新進展、貿易法規、進出口分析、生產分析、價值鏈優化、市場份額、國內外市場參與者的影響、新興收入來源的機遇分析、市場法規變化、戰略市場增長分析、市場規模、各細分市場增長、應用領域及主導地位、產品審批、產品發布、地域擴張以及市場技術創新。如需了解更多關於多發性硬化症治療市場的信息,請聯繫 Data Bridge Market Research 獲取分析師簡報,我們的團隊將幫助您做出明智的市場決策,實現市場成長。
最新進展:
- 2021年,強生公司旗下的Ponvory獲得美國FDA批准。 Ponvory是一種每日一次口服的選擇性鞘氨醇-1-磷酸受體1(S1P1)調節劑,用於治療患有復發型多發性硬化症(MS)的成人患者,例如臨床孤立症候群、復發緩解型多發性硬化症和活動性繼發進展型多發性硬化症。
- 2020 年,諾華宣布其藥物 Kesimpta 獲得美國 FDA 批准,以皮下注射劑的形式用於治療成人復發型多發性硬化症 (RMS),包括臨床孤立綜合徵、復發緩解型疾病和活動性繼發進展型疾病。
歐洲多發性硬化症治療市場範圍
多發性硬化症治療市場按疾病類型、治療方法、藥物類型和最終用戶進行細分。這些細分市場的成長將有助於您分析行業中成長緩慢的細分市場,並為用戶提供有價值的市場概覽和市場洞察,以幫助他們制定策略決策,從而確定核心市場應用。
疾病類型
- 復發緩解型多發性硬化症(RRMS)
- 繼發性進展型多發性硬化症(SPMS)
- 原發性進行性多發性硬化症(PPMS)
- 重度復發緩解型多發性硬化症(RES)
治療
- 預防性療法
- 終止療法/急性惡化期的治療
- 症狀治療
藥物類型
- 品牌
- 通用的
給藥途徑
- 口服
- 腸外
終端用戶
- 醫院和診所
- 診斷實驗室
- 其他的
多發性硬化症治療區域分析/見解
如上所述,本文分析了多發性硬化症治療市場,並按疾病類型、治療方法、藥物類型和最終用戶提供了市場規模見解和趨勢。
多發性硬化症治療市場報告涵蓋的國家包括德國、法國、英國、荷蘭、瑞士、比利時、俄羅斯、義大利、西班牙、土耳其以及歐洲其他地區。
由於德國在該地區推出了全新的多發性硬化症治療方案,預計在2023年至2030年的預測期內,德國將引領市場。該地區不斷增長的藥物研發投入也推動了市場成長。此外,人們對多發性硬化症及其治療的認知度不斷提高,也有助於推動市場成長。
報告的國別部分還提供了影響各個市場的因素以及國內市場監管變化,這些因素和變化會影響市場的當前和未來趨勢。下游和上游價值鏈分析、技術趨勢和波特五力分析、案例研究等數據點是預測各國市場前景的基礎。此外,在進行國別數據預測分析時,還會考慮歐洲品牌的市場佔有率和可用性,以及它們因來自本地和國內品牌的激烈或稀缺競爭而面臨的挑戰,以及國內關稅和貿易路線的影響。
歐洲多發性硬化症治療市場競爭格局及份額分析
多發性硬化症治療市場競爭格局分析報告按競爭對手提供了詳細信息,包括公司概況、財務狀況、收入、市場潛力、研發投入、新市場拓展計劃、歐洲業務佈局、生產基地及設施、產能、公司優勢與劣勢、產品發布、產品線寬度與廣度以及應用領域優勢。以上數據僅與各公司在多發性硬化症治療市場的業務相關。
多發性硬化症治療市場的主要參與者包括:
- 艾伯維公司(美國)
- 博士倫健康公司(加拿大)
- Biora Therapeutics, Inc (美國)
- 勃林格殷格翰國際有限公司(德國)
- 安進公司(美國)
- 輝瑞公司(美國)
- F. Hoffmann-La Roche Ltd(瑞士)
- 邁蘭公司(美國)
- 諾華公司(瑞士)
- 拜耳股份公司(德國)
- 百時美施貴寶公司(美國)
- 百健(美國)
- 梯瓦製藥工業股份有限公司(以色列)
- 武田藥品工業株式會社(日本)
- Jazz Pharmaceuticals, Inc (英國)
- 雅培(美國)
- Bio-Rad Laboratories Inc.(美國)
- 邁蘭公司(美國)
SKU-
Get online access to the report on the World's First Market Intelligence Cloud
- Interactive Data Analysis Dashboard
- Company Analysis Dashboard for high growth potential opportunities
- Research Analyst Access for customization & queries
- Competitor Analysis with Interactive dashboard
- Latest News, Updates & Trend analysis
- Harness the Power of Benchmark Analysis for Comprehensive Competitor Tracking
目录
1 INTRODUCTION
1.1 OBJECTIVES OF THE STUDY
1.2 MARKET DEFINITION
1.3 OVERVIEW OF EUROPE MULTIPLE SCLEROSIS TREATMENT MARKET
1.4 CURRENCY AND PRICING
1.5 LIMITATION
1.6 MARKETS COVERED
2 MARKET SEGMENTATION
2.1 KEY TAKEAWAYS
2.2 ARRIVING AT THE EUROPE MULTIPLE SCLEROSIS TREATMENT MARKET SIZE
2.2.1 VENDOR POSITIONING GRID
2.2.2 TECHNOLOGY LIFE LINE CURVE
2.2.3 TRIPOD DATA VALIDATION MODEL
2.2.4 MARKET GUIDE
2.2.5 MULTIVARIATE MODELLING
2.2.6 TOP TO BOTTOM ANALYSIS
2.2.7 CHALLENGE MATRIX
2.2.8 APPLICATION COVERAGE GRID
2.2.9 STANDARDS OF MEASUREMENT
2.2.10 VENDOR SHARE ANALYSIS
2.2.11 EPIDEMIOLOGY BASED MODEL
2.2.12 DATA POINTS FROM KEY PRIMARY INTERVIEWS
2.2.13 DATA POINTS FROM KEY SECONDARY DATABASES
2.3 EUROPE MULTIPLE SCLEROSIS TREATMENT MARKET: RESEARCH SNAPSHOT
2.4 ASSUMPTIONS
3 MARKET OVERVIEW
3.1 DRIVERS
3.2 RESTRAINTS
3.3 OPPORTUNITIES
3.4 CHALLENGES
4 EXECUTIVE SUMMARY
5 PREMIUM INSIGHTS
5.1 PESTEL ANALYSIS
5.2 PORTER’S 5 FORCES
6 EPIDEMIOLOGY
6.1 INCIDENCE OF ALL BY GENDER
6.2 TREATMENT RATE
6.3 MORTALITY RATE
6.4 DRUG ADHERENCE AND THERAPY SWITCH MODEL
6.5 PATEINT TREATMENT SUCCESS RATES
7 INDUSTRY INSIGHTS
7.1 PATENT ANALYSIS
7.2 DRUG TREATMENT RATE BY MATURED MARKETS
7.3 DEMOGRAPHIC TRENDS: IMPACTS ON ALL INCIDENCE RATES
7.4 PATIENT FLOW DIAGRAM
7.5 KEY PRICING STRATEGIES
7.6 KEY PATIENT ENROLLMENT STRATEGIES
7.7 INTERVIEWS WITH HEMATOLOGISTS
7.8 INTERVIEWS WITH ONCOLOGISTS
7.9 INTERVIEWS WITH CANCER ORGANIZATIONS
7.1 OTHER KOL SNAPSHOTS
8 PIPELINE ANALYSIS
8.1 CLINICAL TRIALS AND PHASE ANALYSIS
8.2 DRUG THERAPY PIPELINE
8.3 PHASE III CANDIDATES
8.4 PHASE II CANDIDATES
8.5 PHASE I CANDIDATES
8.6 OTHERS (PRE-CLINICAL AND RESEARCH)
9 REGULATORY FRAMEWORK
10 EUROPE MULTIPLE SCLEROSIS TREATMENT MARKET, BY TYPE
10.1 OVERVIEW
10.2 RELAPSING–REMITTING MULTIPLE SCLEROSIS (RRMS)
10.3 CLINICALLY ISOLATED SYNDROME (CIS)
10.4 PRIMARY PROGRESSIVE MS (PPMS)
10.5 SECONDARY PROGRESSIVE MS (SPMS)
11 EUROPE MULTIPLE SCLEROSIS TREATMENT MARKET, BY TREATMENT
11.1 OVERVIEW
11.2 MEDICATIONS
11.2.1 DISEASE-MODIFYING DRUGS
11.2.1.1. MONOCLONAL ANTIBODY
11.2.1.1.1. MONOCLONAL ANTIBODY, BY TYPE
11.2.1.1.1.1 OCRELIZUMAB
11.2.1.1.1.2 NATALIZUMAB
11.2.1.1.1.3 DACLIZUMAB
11.2.1.1.1.4 OTHERS
11.2.1.1.2. MONOCLONAL ANTIBODY, BY DRUG TYPE
11.2.1.1.2.1 GENERIC
11.2.1.1.2.2 BRANDED
11.2.1.1.2.2.1. OCREVUS
11.2.1.1.2.2.2. TYSABRI
11.2.1.1.2.2.3. ZENAPAX
11.2.1.2. IMMUNOMODULATOR DRUGS
11.2.1.2.1. IMMUNOMODULATOR DRUGS, BY DRUG
11.2.1.2.1.1 GLATIRAMER
11.2.1.2.1.2 CLADRIBINE
11.2.1.2.1.3 TERIFLUNOMIDE
11.2.1.2.1.4 DIMETHYL FUMARATE
11.2.1.2.1.5 OTHERS
11.2.1.2.2. IMMUNOMODULATOR DRUGS, BY DRUG TYPE
11.2.1.2.2.1 GENERIC
11.2.1.2.2.2 BRANDED
11.2.1.2.2.2.1. GLATOPA
11.2.1.2.2.2.2. COPAXONE
11.2.1.2.2.2.3. MAVENCLAD
11.2.1.2.2.2.4. OTHERS
11.2.1.3. SPHINGOSINE 1-PHOSPHATE (S1P) RECEPTOR MODULATORS
11.2.1.3.1. SPHINGOSINE 1-PHOSPHATE (S1P) RECEPTOR MODULATORS, BY TYPE
11.2.1.3.1.1 PONESIMOD
11.2.1.3.1.2 FINGOLIMOD
11.2.1.3.1.3 SIPONIMOD
11.2.1.3.1.4 OZANIMOD
11.2.1.3.1.5 OTHERS
11.2.1.3.2. SPHINGOSINE 1-PHOSPHATE (S1P) RECEPTOR MODULATORS, BY DRUG TYPE
11.2.1.3.2.1 GENERIC
11.2.1.3.2.2 BRANDED
11.2.1.3.2.2.1. PONVORY
11.2.1.3.2.2.2. ZEPOSIA
11.2.1.3.2.2.3. MAYZENT
11.2.1.3.2.2.4. ZEPOSIA
11.2.1.4. ANTI CANCER DRUGS
11.2.1.4.1. ANTI CANCER DRUGS, BY TYPE
11.2.1.4.1.1 MITOXANTRONE
11.2.1.4.1.2 CYCLOPHOSPHAMIDE
11.2.1.4.1.3 MELPHALAN FLUFENAMIDE
11.2.1.4.1.4 OTHERS
11.2.1.4.2. ANTI CANCER DRUGS, BY DRUG TYPE
11.2.1.4.2.1 GENERIC
11.2.1.4.2.2 BRANDED
11.2.1.4.2.2.1. PEPAXTO
11.2.1.4.2.2.2. CYTOXAN
11.2.1.4.2.2.3. NEOSAR
11.2.1.4.2.2.4. OTHERS
11.2.1.5. INTERFERONE
11.2.1.5.1. INTERFERONE, BY TYPE
11.2.1.5.1.1 INTERFERON BETA-1B
11.2.1.5.1.2 INTERFERON BETA-1A
11.2.1.5.1.3 PEGYLATED INTERFERON BETA-1A
11.2.1.5.1.4 OTHERS
11.2.1.5.2. INTERFERONE, BY DRUG TYPE
11.2.1.5.2.1 GENERIC
11.2.1.5.2.2 BRANDED
11.2.1.5.2.2.1. ROFERON
11.2.1.5.2.2.2. VIRAFERON
11.2.1.5.2.2.3. AVONEX
11.2.1.5.2.2.4. BETASERON
11.2.1.6. IMMUNOSUPPRESSANTS
11.2.1.6.1. IMMUNOSUPPRESSANTS, BY TYPE
11.2.1.6.1.1 AZATHIOPRINE
11.2.1.6.1.2 METHOTREXATE
11.2.1.6.1.3 OTHERS
11.2.1.6.2. IMMUNOSUPPRESSANTS, BY DRUG TYPE
11.2.1.6.2.1 AZASAN
11.2.1.6.2.2 IMURAN
11.2.1.6.2.3 REDITREX
11.2.1.6.2.4 TREXALL
11.2.1.6.2.5 XATMEP
11.2.2 SYMPTOMATIC THERAPIES
11.2.2.1. MUSCLE RELAXANTS
11.2.2.1.1. MUSCLE RELAXANTS, BY TYPE
11.2.2.1.1.1 BACLOFEN
11.2.2.1.1.2 TIZANIDINE
11.2.2.1.1.3 CYCLOBENZAPRINE
11.2.2.1.1.4 OTHERS
11.2.2.1.2. MUSCLE RELAXANTS, BY DRUG TYPE
11.2.2.1.2.1 GENERIC
11.2.2.1.2.2 BRANDED
11.2.2.1.2.2.1. LIORESAL
11.2.2.1.2.2.2. ZANAFLEX
11.2.2.1.2.2.3. FLEXERIL
11.2.2.2. FATIGUE MEDICATIONS
11.2.2.2.1. FATIGUE MEDICATIONS, BY TYPE
11.2.2.2.1.1 AMANTADINE
11.2.2.2.1.2 MODAFINIL
11.2.2.2.1.3 METHYLPHENIDATE
11.2.2.2.1.4 OTHERS
11.2.2.2.2. FATIGUE MEDICATIONS, BY DRUG TYPE
11.2.2.2.2.1 GENERIC
11.2.2.2.2.2 BRANDED
11.2.2.2.2.2.1. GOCOVRI
11.2.2.2.2.2.2. PROVIGIL
11.2.2.2.2.2.3. RITALIN
11.2.2.3. NEUROPATHIC PAIN
11.2.2.3.1. GABAPENTIN
11.2.2.3.2. CARBAMAZEPINE
11.2.2.3.3. AMITRIPTYLINE
11.2.2.3.4. OTHERS
11.2.2.4. CORTICOSTEROIDS
11.2.2.4.1. PREDNISONE
11.2.2.4.2. METHYLPREDNISOLONE
11.2.2.4.3. DEXAMETHASONE
11.2.2.4.4. PREDNISOLONE
11.2.2.4.5. BETAMETHASONE
11.2.2.4.6. OTHERS
11.3 PHYSIOTHERAPY
11.3.1 MANUAL THERAPY
11.3.2 SPECIALIZED TREATMENTS
11.3.3 JOINT MOBILIZATION TECHNIQUES
11.3.4 SUSPENSION THERAPY
11.3.5 RELAXATION TECHNIQUES
11.3.6 POSTURE CORRECTION METHODS
11.4 PLASMAPHERESIS
11.4.1 PLASMA EXCHANGE (PE)
11.4.2 DOUBLE-MEMBRANE-FILTRATION PLASMAPHERESIS (DFP)
11.4.3 ADSORPTION PLASMAPHERESIS
11.5 OTHERS
12 GLOBAL MULTIPLE SCLEROSIS MARKET , BY DRUG TYPE
12.1 OVERVIEW
12.2 GENERIC
12.3 BRANDED
12.3.1 OCREVUS
12.3.2 TYSABRI
12.3.3 ZENAPAX
12.3.4 GLATOPA
12.3.5 COPAXONE
12.3.6 MAVENCLAD
12.3.7 PONVORY
12.3.8 ZEPOSIA
12.3.9 MAYZENT
12.3.10 ZEPOSIA
12.3.11 PEPAXTO
12.3.12 CYTOXAN
12.3.13 NEOSAR
12.3.14 ROFERON
12.3.15 VIRAFERON
12.3.16 AVONEX
12.3.17 BETASERON
12.3.18 BACLOFEN
12.3.19 TIZANIDINE
12.3.20 CYCLOBENZAPRINE
12.3.21 OTHERS
13 EUROPE MULTIPLE SCLEROSIS TREATMENT MARKET, BY ROUTE OF ADMINISTRATION
13.1 OVERVIEW
13.2 ORAL
13.2.1 TABLET
13.2.2 CAPSULE
13.2.3 OTHERS
13.3 PARENTERAL
13.3.1 INTRAVENOUS
13.3.2 SUBCUTANEOUS
13.3.3 OTHERS
13.4 OTHERS
14 EUROPE MULTIPLE SCLEROSIS TREATMENT MARKET, BY PRESCRIPTION TYPE
14.1 OVERVIEW
14.2 OVER THE COUNTER
14.3 PRESCRIPTION
15 EUROPE MULTIPLE SCLEROSIS TREATMENT MARKET, BY AGE GROUP
15.1 OVERVIEW
15.2 CHILDREN
15.3 ADULTS
15.4 GERIATRIC
16 EUROPE MULTIPLE SCLEROSIS TREATMENT MARKET, BY GENDER
16.1 OVERVIEW
16.2 MALE
16.3 FEMALE
17 EUROPE MULTIPLE SCLEROSIS TREATMENT MARKET, BY END USER
17.1 OVERVIEW
17.2 HOSPITALS
17.3 SPECIALTY CLINICS
17.4 HOME HEALTHCARE
17.5 AMBULATORY SURGICAL CENTERS
17.6 OTHERS
18 EUROPE MULTIPLE SCLEROSIS TREATMENT MARKET, BY DISTRIBUTION CHANNEL
18.1 OVERVIEW
18.2 HOSPITAL PHARMACY
18.3 RETAIL PHARMACY
18.3.1 ONLINE STORES
18.3.2 PHARMACY STORES
18.4 OTHERS
19 EUROPE MULTIPLE SCLEROSIS TREATMENT MARKET, BY GEOGRAPHY
EUROPE MULTIPLE SCLEROSIS TREATMENT MARKET, (ALL SEGMENTATION PROVIDED ABOVE IS REPRESENTED IN THIS CHAPTER BY COUNTRY)
19.1 EUROPE
19.1.1 GERMANY
19.1.2 U.K.
19.1.3 ITALY
19.1.4 FRANCE
19.1.5 SPAIN
19.1.6 RUSSIA
19.1.7 SWITZERLAND
19.1.8 TURKEY
19.1.9 BELGIUM
19.1.10 NETHERLANDS
19.1.11 DENMARK
19.1.12 SWEDEN
19.1.13 POLAND
19.1.14 NORWAY
19.1.15 FINLAND
19.1.16 REST OF EUROPE
20 EUROPE MULTIPLE SCLEROSIS TREATMENT MARKET, COMPANY LANDSCAPE
20.1 COMPANY SHARE ANALYSIS: GLOBAL
20.2 COMPANY SHARE ANALYSIS: NORTH AMERICA
20.3 COMPANY SHARE ANALYSIS: EUROPE
20.4 COMPANY SHARE ANALYSIS: EUROPE
20.5 MERGERS & ACQUISITIONS
20.6 NEW PRODUCT DEVELOPMENT & APPROVALS
20.7 EXPANSIONS
20.8 REGULATORY CHANGES
20.9 PARTNERSHIP AND OTHER STRATEGIC DEVELOPMENTS
21 EUROPE MULTIPLE SCLEROSIS TREATMENT MARKET, SWOT AND DBMR ANALYSIS
22 EUROPE MULTIPLE SCLEROSIS TREATMENT MARKET, COMPANY PROFILE
22.1 BIOGEN
22.1.1 COMPANY OVERVIEW
22.1.2 REVENUE ANALYSIS
22.1.3 GEOGRAPHIC PRESENCE
22.1.4 PRODUCT PORTFOLIO
22.1.5 RECENT DEVELOPMENTS
22.2 TEVA NEUROSCIENCE, INC.
22.2.1 COMPANY OVERVIEW
22.2.2 REVENUE ANALYSIS
22.2.3 GEOGRAPHIC PRESENCE
22.2.4 PRODUCT PORTFOLIO
22.2.5 RECENT DEVELOPMENTS
22.3 VIATRIS INC.
22.3.1 COMPANY OVERVIEW
22.3.2 REVENUE ANALYSIS
22.3.3 GEOGRAPHIC PRESENCE
22.3.4 PRODUCT PORTFOLIO
22.3.5 RECENT DEVELOPMENTS
22.4 NOVARTIS AG
22.4.1 COMPANY OVERVIEW
22.4.2 REVENUE ANALYSIS
22.4.3 GEOGRAPHIC PRESENCE
22.4.4 PRODUCT PORTFOLIO
22.4.5 RECENT DEVELOPMENTS
22.5 MERCK KGAA
22.5.1 COMPANY OVERVIEW
22.5.2 REVENUE ANALYSIS
22.5.3 GEOGRAPHIC PRESENCE
22.5.4 PRODUCT PORTFOLIO
22.5.5 RECENT DEVELOPMENTS
22.6 GLENMARK PHARMACEUTICALS LTD
22.6.1 COMPANY OVERVIEW
22.6.2 REVENUE ANALYSIS
22.6.3 GEOGRAPHIC PRESENCE
22.6.4 PRODUCT PORTFOLIO
22.6.5 RECENT DEVELOPMENTS
22.7 LUPIN
22.7.1 COMPANY OVERVIEW
22.7.2 REVENUE ANALYSIS
22.7.3 GEOGRAPHIC PRESENCE
22.7.4 PRODUCT PORTFOLIO
22.7.5 RECENT DEVELOPMENTS
22.8 CIPLA INC
22.8.1 COMPANY OVERVIEW
22.8.2 REVENUE ANALYSIS
22.8.3 GEOGRAPHIC PRESENCE
22.8.4 PRODUCT PORTFOLIO
22.8.5 RECENT DEVELOPMENTS
22.9 SANOFI
22.9.1 COMPANY OVERVIEW
22.9.2 REVENUE ANALYSIS
22.9.3 GEOGRAPHIC PRESENCE
22.9.4 PRODUCT PORTFOLIO
22.9.5 RECENT DEVELOPMENTS
22.1 BAYER
22.10.1 COMPANY OVERVIEW
22.10.2 REVENUE ANALYSIS
22.10.3 GEOGRAPHIC PRESENCE
22.10.4 PRODUCT PORTFOLIO
22.10.5 RECENT DEVELOPMENTS
22.11 ABBVIE, INC
22.11.1 COMPANY OVERVIEW
22.11.2 REVENUE ANALYSIS
22.11.3 GEOGRAPHIC PRESENCE
22.11.4 PRODUCT PORTFOLIO
22.11.5 RECENT DEVELOPMENTS
22.12 ABBOTT
22.12.1 COMPANY OVERVIEW
22.12.2 REVENUE ANALYSIS
22.12.3 GEOGRAPHIC PRESENCE
22.12.4 PRODUCT PORTFOLIO
22.12.5 RECENT DEVELOPMENTS
22.13 F. HOFFMANN-LA ROCHE LTD
22.13.1 COMPANY OVERVIEW
22.13.2 REVENUE ANALYSIS
22.13.3 GEOGRAPHIC PRESENCE
22.13.4 PRODUCT PORTFOLIO
22.13.5 RECENT DEVELOPMENTS
22.14 BRISTOL MYERS SQUIBB
22.14.1 COMPANY OVERVIEW
22.14.2 REVENUE ANALYSIS
22.14.3 GEOGRAPHIC PRESENCE
22.14.4 PRODUCT PORTFOLIO
22.14.5 RECENT DEVELOPMENTS
22.15 TG THERAPEUTICS, INC
22.15.1 COMPANY OVERVIEW
22.15.2 REVENUE ANALYSIS
22.15.3 GEOGRAPHIC PRESENCE
22.15.4 PRODUCT PORTFOLIO
22.15.5 RECENT DEVELOPMENTS
NOTE: THE COMPANIES PROFILED IS NOT EXHAUSTIVE LIST AND IS AS PER OUR PREVIOUS CLIENT REQUIREMENT. WE PROFILE MORE THAN 100 COMPANIES IN OUR STUDY AND HENCE THE LIST OF COMPANIES CAN BE MODIFIED OR REPLACED ON REQUEST
23 RELATED REPORTS
24 CONCLUSION
25 QUESTIONNAIRE
26 ABOUT DATA BRIDGE MARKET RESEARCH
1 INTRODUCTION
1.1 OBJECTIVES OF THE STUDY
1.2 MARKET DEFINITION
1.3 OVERVIEW OF EUROPE MULTIPLE SCLEROSIS TREATMENT MARKET
1.4 CURRENCY AND PRICING
1.5 LIMITATION
1.6 MARKETS COVERED
2 MARKET SEGMENTATION
2.1 KEY TAKEAWAYS
2.2 ARRIVING AT THE EUROPE MULTIPLE SCLEROSIS TREATMENT MARKET
2.2.1 VENDOR POSITIONING GRID
2.2.2 TECHNOLOGY LIFE LINE CURVE
2.2.3 TRIPOD DATA VALIDATION MODEL
2.2.4 MARKET GUIDE
2.2.5 MULTIVARIATE MODELLING
2.2.6 TOP TO BOTTOM ANALYSIS
2.2.7 CHALLENGE MATRIX
2.2.8 APPLICATION COVERAGE GRID
2.2.9 STANDARDS OF MEASUREMENT
2.2.10 VENDOR SHARE ANALYSIS
2.2.11 SALES VOLUME DATA
2.2.12 DATA POINTS FROM KEY PRIMARY INTERVIEWS
2.2.13 DATA POINTS FROM KEY SECONDARY DATABASES
2.3 EUROPE MULTIPLE SCLEROSIS TREATMENT MARKET: RESEARCH SNAPSHOT
2.4 ASSUMPTIONS
3 MARKET OVERVIEW
3.1 DRIVERS
3.2 RESTRAINTS
3.3 OPPORTUNITIES
3.4 CHALLENGES
4 EXECUTIVE SUMMARY
5 PREMIUM INSIGHTS
5.1 PESTEL ANALYSIS
5.2 PORTER’S FIVE FORCES MODEL
5.3 EVOLUTION MS TREATMENT MARKET IN EUROPE LAST 10-20 YEARS
6 INDUSTRY INSIGHTS
6.1 PATENT ANALYSIS
6.1.1 PATENT LANDSCAPE
6.1.2 USPTO NUMBER
6.1.3 PATENT EXPIRY
6.1.4 EPIO NUMBER
6.1.5 PATENT STRENGTH AND QUALITY
6.1.6 PATENT CLAIMS
6.1.7 PATENT CITATIONS
6.1.8 PATENT LITIGATION AND LICENSING
6.1.9 FILE OF PATENT
6.1.10 PATENT RECEIVED CONTRIES
6.1.11 TECHNOLOGY BACKGROUND
6.2 DRUG TREATMENT RATE BY MATURED MARKETS
6.3 DEMOGRAPHIC TRENDS: IMPACTS ON ALL INCIDENCE RATES
6.4 PATIENT FLOW DIAGRAM
6.5 KEY PRICING STRATEGIES
6.6 KEY PATIENT ENROLLMENT STRATEGIES
6.7 INTERVIEWS WITH SPECIALIST
6.8 OTHER KOL SNAPSHOTS
7 EPIDEMIOLOGY
7.1 INCIDENCE OF ALL BY GENDER
7.2 TREATMENT RATE
7.3 MORTALITY RATE
7.4 DRUG ADHERENCE AND THERAPY SWITCH MODEL
7.5 PATIENT TREATMENT SUCCESS RATES
8 MERGERS AND ACQUISITION
8.1 LICENSING
8.2 COMMERCIALIZATION AGREEMENTS
9 REGULATORY FRAMEWORK
9.1 REGULATORY APPROVAL PROCESS
9.2 GEOGRAPHIES’ EASE OF REGULATORY APPROVAL
9.3 REGULATORY APPROVAL PATHWAYS
9.4 LICENSING AND REGISTRATION
9.5 POST-MARKETING SURVEILLANCE
9.6 GOOD MANUFACTURING PRACTICES (GMPS) GUIDELINES
10 PIPELINE ANALYSIS
10.1 CLINICAL TRIALS AND PHASE ANALYSIS
10.2 DRUG THERAPY PIPELINE
10.3 PHASE III CANDIDATES
10.4 PHASE II CANDIDATES
10.5 PHASE I CANDIDATES
10.6 OTHERS (PRE-CLINICAL AND RESEARCH)
TABLE 1 EUROPE CLINICAL TRIAL MARKET FOR MULTIPLE SCLEROSIS TREATMENT MARKET
Company Name Therapeutic Area
XX XX
XX XX
XX XX
XX XX
XX XX
XX XX
Sources: Press Releases, Annual Reports, SEC Filings, Investor Presentations, Other Government Sources, Analysis Based on Inputs from Secondary, Expert Interviews
TABLE 2 DISTRIBUTION OF PRODUCTS AND PROJECTS BY PHASE MULTIPLE SCLEROSIS TREATMENT MARKET
Phase Number of Projects
Preclinical/Research Projects XX
Clinical Development XX
Phase I XX
Phase II XX
Phase III XX
U.S. Filed/Approved But Not Yet Marketed XX
Total XX
Sources: Press Releases, Annual Reports, SEC Filings, Investor Presentations, Other Government Sources, Analysis Based on Inputs from Secondary, Expert Interviews
TABLE 3 DISTRIBUTION OF PROJECTS BY THERAPEUTIC AREA AND PHASE MULTIPLE SCLEROSIS TREATMENT MARKET
Therapeutic Area Preclinical/ Research Project
XX XX
XX XX
XX XX
XX XX
XX XX
Total Projects XX
Sources: Press Releases, Annual Reports, SEC Filings, Investor Presentations, Other Government Sources, Analysis Based on Inputs from Secondary, Expert Interviews
TABLE 4 DISTRIBUTION OF PROJECTS BY SCIENTIFIC APPROACH AND PHASE MULTIPLE SCLEROSIS TREATMENT MARKET
Technology Preclinical/ Research Project
XX XX
XX XX
XX XX
XX XX
XX XX
Total Projects XX
Sources: Press Releases, Annual Reports, SEC Filings, Investor Presentations, Other Government Sources, Analysis Based on Inputs from Secondary, Expert Interviews
FIGURE 1 TOP ENTITIES BASED ON R&D GLANCE FOR MULTIPLE SCLEROSIS TREATMENT MARKET
Sources: Press Releases, Annual Reports, SEC Filings, Investor Presentations, Other Government Sources, Analysis Based on Inputs from Secondary, Expert Interviews
11 MARKETED DRUG ANALYSIS
11.1 DRUG
11.1.1 BRAND NAME
11.1.2 GENERICS NAME
11.2 THERAPEUTIC INDICTION
11.3 PHARMACOLOGICAL CLASS OF THE DRUG
11.4 DRUG PRIMARY INDICATION
11.5 MARKET STATUS
11.6 MEDICATION TYPE
11.7 DRUG DOSAGES FORM
11.8 DOSAGES AVAILABILITY
11.9 DRUG ROUTE OF ADMINISTRATION
11.1 DOSING FREQUENCY
11.11 DRUG INSIGHT
11.12 AN OVERVIEW OF THE DRUG DEVELOPMENT ACTIVITIES SUCH AS REGULATORY MILSTONE, SAFETY DATA AND EFFICACY DATA, MARKET EXCLUSIVITY DATA.
11.12.1 FORECAST MARKET OUTLOOK
11.12.2 CROSS COMPETITION
11.12.3 THERAPEUTIC PORTFOLIO
11.12.4 CURRENT DEVELOPMENT SCENARIO
12 MARKET ACCESS
12.1 10-YEAR MARKET FORECAST
12.2 CLINICAL TRIAL RECENT UPDATES
12.3 ANNUAL NEW FDA APPROVED DRUGS
12.4 DRUGS MANUFACTURER AND DEALS
12.5 MAJOR DRUG UPTAKE
12.6 CURRENT TREATMENT PRACTICES
12.7 IMPACT OF UPCOMING THERAPY
13 R & D ANALYSIS
13.1 COMPARATIVE ANALYSIS
13.2 DRUG DEVELOPMENTAL LANDSCAPE
13.3 IN-DEPTH INSIGHTS ON REGULATORY MILESTONES
13.4 THERAPEUTIC ASSESSMENT
13.5 ASSET-BASED COLLABORATIONS AND PARTNERSHIPS
14 MARKET OVERVIEW
14.1 DRIVERS
14.2 RESTRAINTS
14.3 OPPORTUNITIES
14.4 CHALLENGES
15 EUROPE MULTIPLE SCLEROSIS TREATMENT MARKET, BY TYPE
15.1 OVERVIEW
15.2 CLINICALLY ISOLATED SYNDROME (CIS)
15.3 RELAPSING–REMITTING MULTIPLE SCLEROSIS (RRMS)
15.4 SECONDARY PROGRESSIVE MULTIPLE SCLEROSIS (SPMS)
15.5 PRIMARY PROGRESSIVE MULTIPLE SCLEROSIS (PPMS)
15.6 SEVERE RELAPSING–REMITTING MULTIPLE SCLEROSIS (RES)
16 EUROPE MULTIPLE SCLEROSIS TREATMENT MARKET,BY THERAPEUTICS
16.1 OVERVIEW
16.2 APPROVED/ MARKETED MEDICATION
16.2.1 PREVENTIVE THERAPIES
16.2.1.1. ORAL MEDICATION
16.2.1.1.1. TECFIDERA
16.2.1.1.2. GILENYA
16.2.1.1.3. AUBAGIO
16.2.1.1.4. MAVENCLAD
16.2.1.1.5. MAYZENT
16.2.1.1.6. VUMERITY
16.2.1.1.7. OTHERS
16.2.1.2. INJECTABLE MEDICATION
16.2.1.2.1. INTERFERON BETA-1A
16.2.1.2.1.1 AVONEX
16.2.1.2.1.2 REBIF
16.2.1.2.1.3 OTHERS
16.2.1.2.2. GLATIRAMER ACETATE
16.2.1.2.2.1 COPAXONE
16.2.1.2.2.2 GLATOPA
16.2.1.2.2.3 OTHERS
16.2.1.2.3. INTERFERON BETA-1B
16.2.1.2.3.1 BETASERON
16.2.1.2.3.2 EXTAVIA
16.2.1.2.4. PLEGRIDY/PEGINTERFERON BETA-1A
16.2.1.2.5. CHEMOTHERAPY DRUG
16.2.1.2.6. MONOCLONAL ANTIBODIES
16.2.1.3. INFUSED MEDICATION
16.2.1.3.1. TYSABRI
16.2.1.3.2. OCREVUS
16.2.1.3.3. LEMTRADA
16.2.1.3.4. MITOXANTRONE
16.2.1.3.5. NOVANTRONE
16.2.1.3.6. OTHERS
16.2.1.4. HAEMATOPOIETIC STEM CELL TRANSPLANTATION (HSCT)
16.2.2 ABORTIVE THERAPIES/TREATMENT OF ACUTE EXACERBATIONS
16.2.2.1. CORTICOSTEROIDS
16.2.2.1.1. ORAL PREDNISONE
16.2.2.1.2. METHYLPREDNISOLONE
16.2.2.1.3. DEXAMETHASONE
16.2.2.1.4. PREDNISOLONE
16.2.2.1.5. BETAMETHASONE
16.2.2.1.6. OTHERS
16.2.2.2. BENZODIAZEPINES
16.2.2.2.1. CLONAZEPAM
16.2.2.2.2. DIAZEPAM
16.2.2.2.3. OTHERS
16.2.2.3. IMMUNOSUPPRESSANTS
16.2.2.3.1. AZATHIOPRINE
16.2.2.3.2. CYCLOPHOSPHAMIDE
16.2.2.3.3. METHOTREXATE
16.2.2.3.4. MITOXANTRONE
16.2.2.3.5. OTHERS
16.2.2.4. SPHINGOSINE 1-PHOSPHATE RECEPTOR MODULATORS
16.2.2.4.1. SIPONIMOD
16.2.2.4.2. FINGOLIMOD
16.2.2.4.3. OZANIMOD
16.2.2.4.4. OTHERS
16.2.2.5. DOPAMINE AGONISTS
16.2.2.5.1. OSMOLEX ER
16.2.2.5.2. GOCOVRI
16.2.2.5.3. OTHERS
16.2.2.6. NEUROMUSCULAR BLOCKERS
16.2.2.6.1. BOTOX
16.2.2.6.2. BOTOX COSMETIC
16.2.2.6.3. BOTULINUM TOXIN
16.2.2.7. ADRENOCORTICOTROPIC HORMONE
16.2.2.8. INTRAVENOUS IMMUNOGLOBULIN
16.2.2.9. PLASMAPHERESIS
16.2.2.10. OTHERS
16.2.3 SYMPTOMATIC THERAPIES
16.2.3.1. MUSCLE RELAXANTS
16.2.3.1.1. LIORESAL
16.2.3.1.2. ZANAFLEX
16.2.3.1.3. OTHERS
16.2.3.2. FATIGUE MEDICATIONS
16.2.3.2.1. GOCOVRI
16.2.3.2.2. OSMOLEX
16.2.3.2.3. PROVIGIL
16.2.3.2.4. RITALIN
16.2.3.2.5. OTHERS
16.2.3.3. STIMULANTS
16.2.3.3.1. MODAFINIL
16.2.3.3.2. ARMODAFINIL
16.2.3.3.3. OTHERS
16.2.3.4. ANTICONVULSANTS
16.2.3.4.1. GABAPENTIN
16.2.3.4.2. CARBAMAZEPINE
16.2.3.4.3. PREGABALIN
16.2.3.4.4. TOPIRAMATE
16.2.3.4.5. OTHERS
16.2.3.5. NONSTEROIDAL ANTI-INFLAMMATORY DRUGS
16.2.3.5.1. IBUPROFEN
16.2.3.5.2. NAPROXEN
16.2.3.5.3. DICLOFENAC
16.2.3.5.4. INDOMETHACIN
16.2.3.5.5. OTHERS
16.2.3.6. ANTIDIARRHEALS
16.2.3.7. POTASSIUM CHANNEL BLOCKERS
16.2.3.8. PHYSIOTHERAPY
16.2.3.9. LAXATIVES
16.2.3.10. OTHERS
16.3 PIPELINE DRUGS
16.3.1 SCM-0101
16.3.2 IMCY-MS-001
16.3.3 PIPE-307
16.3.4 ATA188
16.3.5 WP1303
16.3.6 FORALUMAB
16.3.7 OTHERS
17 EUROPE MULTIPLE SCLEROSIS TREATMENT MARKET, BY DRUG TYPE
17.1 OVERVIEW
17.2 BRANDED
17.2.1 TECFIDERA
17.2.2 GILENYA
17.2.3 AUBAGIO
17.2.4 AVONEX
17.2.5 TYSABRI
17.2.6 REBIF
17.2.7 COPAXONE
17.2.8 8 BETASERON
17.2.9 AMPYRA/FAMPYRA
17.2.10 OTHERS
17.3 GENERICS
18 EUROPE MULTIPLE SCLEROSIS TREATMENT MARKET,BY ROUTE OF ADMINISTRATION
18.1 OVERVIEW
18.2 ORAL
18.2.1 TABLETS
18.2.2 CAPSULES
18.2.3 OTHERS
18.3 PARENTERAL
18.3.1 INTRAVENOUS
18.3.2 SUBCUTANEOUS
18.3.3 OTHERS
18.4 OTHERS
19 EUROPE MULTIPLE SCLEROSIS TREATMENT MARKET,BY AGE GROUP
19.1 OVERVIEW
19.2 PEDIATRIC
19.2.1 MALE
19.2.2 FEMALE
19.3 ADULT
19.3.1 MALE
19.3.2 FEMALE
19.4 GERIATRIC
19.4.1 MALE
19.4.2 FEMALE
20 EUROPE MULTIPLE SCLEROSIS TREATMENT MARKET,BY GENDER
20.1 OVERVIEW
20.2 MALE
20.3 FEMALE
21 EUROPE MULTIPLE SCLEROSIS TREATMENT MARKET, BY END USER
21.1 OVERVIEW
21.2 HOSPITALS
21.2.1 PUBLIC
21.2.1.1. TIER 1
21.2.1.2. TIER 2
21.2.1.3. TIER 3
21.2.2 PRIVATE
21.2.2.1. TIER 1
21.2.2.2. TIER 2
21.2.2.3. TIER 3
21.3 HOME HELATHCARE
21.4 SPECIALITY CENTRE
21.5 ACADEMIC AND RESEARCH INSTITUTES
21.6 OTHERS
22 EUROPE MULTIPLE SCLEROSIS TREATMENT MARKET, BY DISTRIBUTION CHANNEL
22.1 OVERVIEW
22.2 DIRECT TENDERS
22.3 RETAIL SALES
22.3.1 ONLINE
22.3.1.1. E-STORES
22.3.1.2. COMPANY WEBSITE
22.3.1.3. OTHERS
22.3.2 OFFLINE
22.3.2.1. HOSPITAL PHARMACY
22.3.2.2. RETAIL PHARMACY
22.3.2.3. OTHERS
22.4 OTHERS
23 EUROPE MULTIPLE SCLEROSIS TREATMENT MARKET, COMPANY LANDSCAPE
23.1 COMPANY SHARE ANALYSIS: EUROPE
23.2 MERGERS & ACQUISITIONS
23.3 NEW PRODUCT DEVELOPMENT & APPROVALS
23.4 EXPANSIONS
23.5 REGULATORY CHANGES
23.6 PARTNERSHIP AND OTHER STRATEGIC DEVELOPMENTS
24 EUROPE MULTIPLE SCLEROSIS TREATMENT MARKET, SWOT AND DBMR ANALYSIS
25 EUROPE MULTIPLE SCLEROSIS TREATMENT MARKET, BY COUNTRY
EUROPE MULTIPLE SCLEROSIS TREATMENT MARKET, (ALL SEGMENTATION PROVIDED ABOVE IS REPRESENTED IN THIS CHAPTER BY COUNTRY)
25.1 EUROPE
25.1.1 GERMANY
25.1.2 FRANCE
25.1.3 U.K.
25.1.4 ITALY
25.1.5 SPAIN
25.1.6 RUSSIA
25.1.7 TURKEY
25.1.8 BELGIUM
25.1.9 DENMARK
25.1.10 NETHERLANDS
25.1.11 SWITZERLAND
25.1.12 SWEDEN
25.1.13 POLAND
25.1.14 NORWAY
25.1.15 FINLAND
25.1.16 REST OF EUROPE
25.2 KEY PRIMARY INSIGHTS: BY MAJOR COUNTRIES
26 EUROPE MULTIPLE SCLEROSIS TREATMENT MARKET, COMPANY PROFILE
26.1 MARKETED /APPROVED MANUFRACTURES
26.1.1 BIOGEN
26.1.1.1. COMPANY OVERVIEW
26.1.1.2. REVENUE ANALYSIS
26.1.1.3. GEOGRAPHIC PRESENCE
26.1.1.4. PRODUCT PORTFOLIO
26.1.1.5. RECENT DEVELOPMENTS
26.1.2 NOVARTIS AG
26.1.2.1. COMPANY OVERVIEW
26.1.2.2. REVENUE ANALYSIS
26.1.2.3. GEOGRAPHIC PRESENCE
26.1.2.4. PRODUCT PORTFOLIO
26.1.2.5. RECENT DEVELOPMENTS
26.1.3 GENZYME CORPORATION (A SUBSIDIARY OF SANOFI)
26.1.3.1. COMPANY OVERVIEW
26.1.3.2. REVENUE ANALYSIS
26.1.3.3. GEOGRAPHIC PRESENCE
26.1.3.4. PRODUCT PORTFOLIO
26.1.3.5. RECENT DEVELOPMENTS
26.1.4 MERCK KGAA
26.1.4.1. COMPANY OVERVIEW
26.1.4.2. REVENUE ANALYSIS
26.1.4.3. GEOGRAPHIC PRESENCE
26.1.4.4. PRODUCT PORTFOLIO
26.1.4.5. RECENT DEVELOPMENTS
26.1.5 TEVA PHARMACEUTICALS USA, INC. (A SUBSIDIARY OF TEVA PHARMACEUTICALS INDUSTRIES LTD.)
26.1.5.1. COMPANY OVERVIEW
26.1.5.2. REVENUE ANALYSIS
26.1.5.3. GEOGRAPHIC PRESENCE
26.1.5.4. PRODUCT PORTFOLIO
26.1.5.5. RECENT DEVELOPMENTS
26.1.6 F. HOFFMANN-LA ROCHE LTD
26.1.6.1. COMPANY OVERVIEW
26.1.6.2. REVENUE ANALYSIS
26.1.6.3. GEOGRAPHIC PRESENCE
26.1.6.4. PRODUCT PORTFOLIO
26.1.6.5. RECENT DEVELOPMENTS
26.1.7 ACORDA THERAPEUTICS, INC
26.1.7.1. COMPANY OVERVIEW
26.1.7.2. REVENUE ANALYSIS
26.1.7.3. GEOGRAPHIC PRESENCE
26.1.7.4. PRODUCT PORTFOLIO
26.1.7.5. RECENT DEVELOPMENTS
26.1.8 AMNEAL PHARMACEUTICALS LLC
26.1.8.1. COMPANY OVERVIEW
26.1.8.2. REVENUE ANALYSIS
26.1.8.3. GEOGRAPHIC PRESENCE
26.1.8.4. PRODUCT PORTFOLIO
26.1.8.5. RECENT DEVELOPMENTS
26.1.9 BAYER AG
26.1.9.1. COMPANY OVERVIEW
26.1.9.2. REVENUE ANALYSIS
26.1.9.3. GEOGRAPHIC PRESENCE
26.1.9.4. PRODUCT PORTFOLIO
26.1.9.5. RECENT DEVELOPMENTS
26.1.10 CELGENE CORPORATION (A SUBSIDIARY OF BRISTOL-MYERS SQUIBB COMPANY)
26.1.10.1. COMPANY OVERVIEW
26.1.10.2. REVENUE ANALYSIS
26.1.10.3. GEOGRAPHIC PRESENCE
26.1.10.4. PRODUCT PORTFOLIO
26.1.10.5. RECENT DEVELOPMENTS
26.1.11 HIKMA PHARMACEUTICALS PLC
26.1.11.1. COMPANY OVERVIEW
26.1.11.2. REVENUE ANALYSIS
26.1.11.3. GEOGRAPHIC PRESENCE
26.1.11.4. PRODUCT PORTFOLIO
26.1.11.5. RECENT DEVELOPMENTS
26.1.12 MALLINCKRODT PHARMACEUTICALS
26.1.12.1. COMPANY OVERVIEW
26.1.12.2. REVENUE ANALYSIS
26.1.12.3. GEOGRAPHIC PRESENCE
26.1.12.4. PRODUCT PORTFOLIO
26.1.12.5. RECENT DEVELOPMENTS
26.1.13 VITARIS INC.
26.1.13.1. COMPANY OVERVIEW
26.1.13.2. REVENUE ANALYSIS
26.1.13.3. GEOGRAPHIC PRESENCE
26.1.13.4. PRODUCT PORTFOLIO
26.1.13.5. RECENT DEVELOPMENTS
26.1.14 PAR PHARMACEUTICAL (A SUBSIDIARY OF ENDO INTERNATIONAL PLC)
26.1.14.1. COMPANY OVERVIEW
26.1.14.2. REVENUE ANALYSIS
26.1.14.3. GEOGRAPHIC PRESENCE
26.1.14.4. PRODUCT PORTFOLIO
26.1.14.5. RECENT DEVELOPMENTS
26.1.15 PFIZER INC.
26.1.15.1. COMPANY OVERVIEW
26.1.15.2. REVENUE ANALYSIS
26.1.15.3. GEOGRAPHIC PRESENCE
26.1.15.4. PRODUCT PORTFOLIO
26.1.15.5. RECENT DEVELOPMENTS
26.1.16 ZYDUS PHARMACEUTICALS, INC. (A SUBSIDIARY OF ZYDUS CADILA)
26.1.16.1. COMPANY OVERVIEW
26.1.16.2. REVENUE ANALYSIS
26.1.16.3. GEOGRAPHIC PRESENCE
26.1.16.4. PRODUCT PORTFOLIO
26.1.16.5. RECENT DEVELOPMENTS
26.1.17 JOHNSON & JOHNSON CONSUMER INC.
26.1.17.1. COMPANY OVERVIEW
26.1.17.2. PRODUCT PORTFOLIO
26.1.17.3. REVENUE ANALYSIS
26.1.17.4. GEOGRAPHIC PRESENCE
26.1.17.5. PRODUCT PORTFOLIO
26.1.18 PERRIGO COMPANY PLC,
26.1.18.1. COMPANY OVERVIEW
26.1.18.2. PRODUCT PORTFOLIO
26.1.18.3. REVENUE ANALYSIS
26.1.18.4. GEOGRAPHIC PRESENCE
26.1.18.5. PRODUCT PORTFOLIO
26.1.19 GLENMARK PHARMACEUTICAL INC., USA
26.1.19.1. COMPANY OVERVIEW
26.1.19.2. REVENUE ANALYSIS
26.1.19.3. GEOGRAPHIC PRESENCE
26.1.19.4. PRODUCT PORTFOLIO
26.1.19.5. RECENT DEVELOPMENTS
26.1.20 GLAXOSMITHKLINE PLC.
26.1.20.1. COMPANY OVERVIEW
26.1.20.2. PRODUCT PORTFOLIO
26.1.20.3. REVENUE ANALYSIS
26.1.20.4. GEOGRAPHIC PRESENCE
26.1.20.5. PRODUCT PORTFOLIO
26.2 PIPELINE COMPANIES
26.2.1 IMCYSE
26.2.1.1. COMPANY OVERVIEW
26.2.1.2. PRODUCT PORTFOLIO
26.2.1.3. REVENUE ANALYSIS
26.2.1.4. GEOGRAPHIC PRESENCE
26.2.1.5. PRODUCT PORTFOLIO
26.2.2 WORG PHARMACEUTICALS (ZHEJIANG) CO., LTD.
26.2.2.1. COMPANY OVERVIEW
26.2.2.2. PRODUCT PORTFOLIO
26.2.2.3. REVENUE ANALYSIS
26.2.2.4. GEOGRAPHIC PRESENCE
26.2.2.5. PRODUCT PORTFOLIO
26.2.3 TIZIANA LIFE SCIENCES
26.2.3.1. COMPANY OVERVIEW
26.2.3.2. PRODUCT PORTFOLIO
26.2.3.3. REVENUE ANALYSIS
26.2.3.4. GEOGRAPHIC PRESENCE
26.2.3.5. PRODUCT PORTFOLIO
27 RELATED REPORTS
28 CONCLUSION
29 QUESTIONNAIRE
30 ABOUT DATA BRIDGE MARKET RESEARCH
研究方法
数据收集和基准年分析是使用具有大样本量的数据收集模块完成的。该阶段包括通过各种来源和策略获取市场信息或相关数据。它包括提前检查和规划从过去获得的所有数据。它同样包括检查不同信息源中出现的信息不一致。使用市场统计和连贯模型分析和估计市场数据。此外,市场份额分析和关键趋势分析是市场报告中的主要成功因素。要了解更多信息,请请求分析师致电或下拉您的询问。
DBMR 研究团队使用的关键研究方法是数据三角测量,其中包括数据挖掘、数据变量对市场影响的分析和主要(行业专家)验证。数据模型包括供应商定位网格、市场时间线分析、市场概览和指南、公司定位网格、专利分析、定价分析、公司市场份额分析、测量标准、全球与区域和供应商份额分析。要了解有关研究方法的更多信息,请向我们的行业专家咨询。
可定制
Data Bridge Market Research 是高级形成性研究领域的领导者。我们为向现有和新客户提供符合其目标的数据和分析而感到自豪。报告可定制,包括目标品牌的价格趋势分析、了解其他国家的市场(索取国家列表)、临床试验结果数据、文献综述、翻新市场和产品基础分析。目标竞争对手的市场分析可以从基于技术的分析到市场组合策略进行分析。我们可以按照您所需的格式和数据样式添加您需要的任意数量的竞争对手数据。我们的分析师团队还可以为您提供原始 Excel 文件数据透视表(事实手册)中的数据,或者可以帮助您根据报告中的数据集创建演示文稿。