North America Active Medical Implantable Devices Market
市场规模(十亿美元)
CAGR :
% 
 USD
10.08 Billion
USD
18.51 Billion
2025
2033
USD
10.08 Billion
USD
18.51 Billion
2025
2033
| 2026 –2033 | |
| USD 10.08 Billion | |
| USD 18.51 Billion | |
|
|
|
|
北美主動式植入式醫療器材市場細分,依產品(心臟再同步治療裝置(CRT-D)、植入式心臟復律去顫器、植入式心臟節律器、眼科植入物、神經刺激器、主動植入式助聽器、心室輔助裝置、植入式心臟監視器/植入式循環記錄器、近距離放射治療、植入式血糖監測儀、垂足植入物、肩部植入物、植入式輸液幫浦和植入式配件)、手術類型(傳統手術方法和微創手術)、手術項目(神經血管、心血管、聽力及其他)、最終用戶(醫院、專科診所、門診手術中心和診所)劃分-產業趨勢及至2033 年的預測
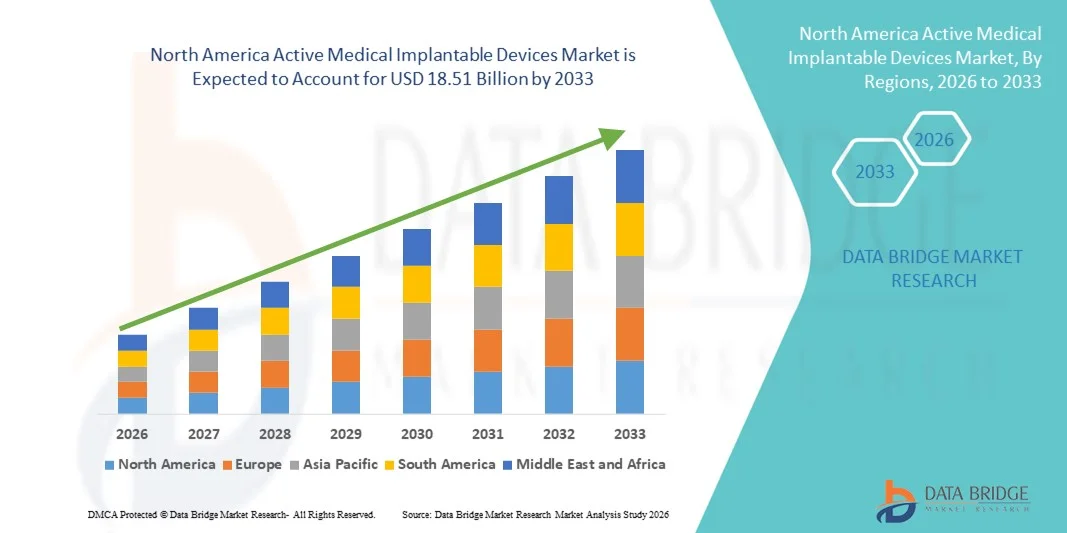
北美有源醫療植入設備市場規模
- 2025年北美有源醫療植入設備市場規模為100.8億美元 ,預計 2033年將達到185.1億美元,預測期內 複合年增長率為7.9%。
- 市場成長主要得益於先進的醫療基礎設施、慢性病患病率的上升以及植入式解決方案領域持續不斷的技術創新,這些創新能夠改善醫院和專科護理機構中患者的治療效果。
- 此外,對高精度、微創和遠端監測治療設備的需求不斷增長,加上有利的報銷政策和對尖端醫療技術的早期應用,正在鞏固北美作為領先的植入式醫療器材區域市場的地位。這些因素共同加速了先進植入式解決方案的普及,顯著推動了該產業的成長。
北美有源醫療植入設備市場分析
- 在美國,包括心臟再同步治療裝置(CRT-D)、植入式除顫器、起搏器、神經刺激器、眼科植入物和主動式助聽器在內的有源植入式醫療器械,正日益成為現代醫療保健的重要組成部分,它們在心血管、神經和感覺應用領域提供持續監測、精準治療幹預,並改善患者預後。
- 有源植入式醫療器材的需求不斷增長,主要受慢性病患病率上升、人口老化以及先進醫療技術日益普及的推動,這些技術能夠實現微創手術、遠端監測和提升患者護理水平。
- 預計到2025年,美國將以74.9%的最大市佔率主導北美主動式醫療植入設備市場。美國擁有先進的醫療基礎設施、高額的醫療支出以及強大的行業領先企業,醫院和專科診所在心臟和神經植入設備的採用方面實現了顯著增長,這主要得益於成熟醫療器械公司和專注於人工智能及數字化連接設備的初創公司不斷推出的創新產品。
- 由於醫療保健投資不斷增加、人們對植入式療法的認識不斷提高以及獲得先進醫療技術的途徑日益便利,預計加拿大將在預測期內成為北美主動式醫療植入設備市場增長最快的國家。
- 2025年,植入式心臟復律去顫器(ICD)細分市場將以45.3%的市場份額佔據主導地位,這主要得益於心血管疾病的高發生率、除顫器技術的進步以及患者對具備遠端監測功能的設備的日益增長的需求。
報告範圍及北美主動式醫療植入器材市場細分
|
屬性 |
北美主動式醫療植入器材市場關鍵洞察 |
|
涵蓋部分 |
|
|
覆蓋國家/地區 |
北美洲
|
|
主要市場參與者 |
|
|
市場機遇 |
|
|
加值資料資訊集 |
除了提供市場價值、成長率、市場細分、地理覆蓋範圍和主要參與者等市場概況外,Data Bridge Market Research 精心編制的市場報告還包括深入的專家分析、患者流行病學、產品線分析、定價分析和監管框架。 |
北美有源醫療植入設備市場趨勢
先進監測與人工智慧療法的整合
- 美國主動式醫療植入設備市場的一個顯著且加速發展的趨勢是將人工智慧賦能的監測、遠端連接和預測分析技術融入其中,這些技術能夠提升患者護理水平,並為臨床醫生提供關於設備性能和患者健康的即時資訊。
- 例如,美敦力公司的人工智慧心臟節律器可以根據患者的活動和健康數據自動調節起搏頻率,從而實現更個人化的治療並減少住院次數。同樣,雅培公司的植入式心臟監視器透過安全的雲端平台向醫師即時傳輸心律不整警報。
- 這些設備整合了人工智慧技術,能夠學習患者特定的心臟或神經系統模式,建議調整治療方案,並針對潛在併發症發出預測警報。例如,波士頓科學公司的神經刺激器可以分析患者的運動模式,以優化刺激參數,並在出現異常情況時提醒醫生。
- 遠端連接使患者和醫療服務提供者能夠在家中監測設備性能和生理指標,從而提高依從性並及早發現不良事件。透過單一平台,臨床醫生可以管理多個植入式設備以及患者的健康數據,從而創造一體化、自動化的醫療體驗。
- 這種朝向更智慧、互聯和以患者為中心的植入式裝置發展的趨勢正在重塑人們對醫療器材功能的期望。因此,像百多力(Biotronik)這樣的公司正在開發配備人工智慧的除顫器和起搏器,這些設備具有自適應治療演算法和基於雲端的監測功能。
- 隨著患者和醫護人員越來越重視便利性、精準性和改善臨床療效,醫院和專科診所對具備人工智慧監測和遠端管理功能的植入式設備的需求正在迅速增長。
- 此外,透過與穿戴式裝置和行動健康應用程式的集成,患者可以主動追蹤自身健康指標以及植入式裝置的數據,從而實現更全面的疾病管理。
北美有源醫療植入設備市場動態
司機
慢性病盛行率上升與技術應用
- 心血管、神經和感覺障礙的日益普遍,以及先進植入式技術的應用,是美國市場需求的重要驅動因素。
- 例如,2025年3月,雅培宣布擴展其植入式心臟監測系統,使其具備基於人工智慧的心律不整檢測功能,進而改善早期介入和病患管理。
- 隨著醫療機構致力於減少患者再入院率和改善治療效果,植入式設備憑藉持續監測、自動治療調整和安全數據傳輸等先進功能,為傳統幹預措施提供了極具吸引力的替代方案。
- 此外,微創手術和遠端監測日益受到青睞,使得植入式設備成為現代療法不可或缺的一部分,使臨床醫生能夠提供更精準、更個人化的照護。
- 與醫院醫療資訊科技系統、遠端監控平台和人工智慧輔助診斷的便捷整合是推動醫院和專科診所採用該技術的關鍵因素。以患者為中心的醫療服務和數位化醫療解決方案的趨勢也進一步促進了市場成長。
- 例如,採用閉環系統的神經刺激器正被越來越多地用於優化慢性疼痛和帕金森氏症的治療,從而推動了專科診所市場的擴張。
- 此外,植入式助聽器在兒童和老年人群體中的使用日益增多,也推高了市場需求,因為越來越多的患者希望透過微創手術改善聽力健康。
克制/挑戰
高昂的成本和監管合規障礙
- 高昂的設備成本、複雜的手術流程以及嚴格的監管要求,都對更廣泛的市場滲透構成了重大挑戰。由於這些設備涉及先進技術和臨床監測,因此通常價格昂貴,並且需要專業的植入技術。
- 例如,由於電池或軟體問題,某些植入式心臟設備被高調召回,這使得一些醫療機構在採用新技術時變得謹慎。
- 透過強有力的臨床驗證、醫生培訓以及遵守FDA和ISO監管標準來應對這些挑戰,對於市場拓展至關重要。美敦力(Medtronic)和波士頓科學(Boston Scientific)等公司強調嚴格的測試和合規流程,以增強臨床醫生和患者的信心。此外,與傳統療法相比,某些先進植入式設備的成本相對較高,這可能會成為預算緊張的醫院的障礙。
- 雖然價格正在逐步下降,保險覆蓋範圍也在擴大,但先進植入式醫療器材的高昂保費仍會阻礙其廣泛應用,尤其是在規模較小的診所或資金較少的醫療機構。
- 透過成本優化、報銷支援和加強監管合規來克服這些挑戰,對於美國主動式醫療植入設備市場的持續成長至關重要。
- 例如,連網植入式裝置遵守更新後的網路安全和資料隱私法規變得越來越複雜,這需要製造商和醫療保健提供者進行額外的投資。
- 此外,微創植入手術的訓練需求和術後設備監測也增加了操作上的挑戰,這可能會限制其普及,尤其是在農村或資源匱乏的醫療機構。
北美有源醫療植入設備市場範圍
市場按產品、手術類型、手術流程和最終用戶進行細分。
- 副產品
根據產品類型,北美主動式植入式醫療器材市場可細分為心臟再同步治療裝置 (CRT-D)、植入式心臟復律去顫器 (ICD)、植入式心臟節律器、眼科植入物、神經刺激器、主動植入式助聽器、心室輔助裝置、植入式心臟植入器/植入式循環記錄器、近距離植入式植入式血糖植入物、室輔助裝置、植入式心臟植入器/植入式循環記錄器、近距離輸液、肩部植入式血糖植入器、心室輔助裝置、植入式心臟植入器/植入式循環記錄器、近距離輸液、肩部植入式血糖植入物、植入式移植物、植入式植入式循環記錄器、近腳植入物植入式血糖植入物。植入式心臟復律去顫器 (ICD) 細分市場預計將佔據主導地位,到 2025 年將以 45.3% 的最大收入份額佔據市場,這主要得益於美國心血管疾病(如心律不整和心臟驟停)的高發生率。由於 ICD 在恢復正常心律方面已被證實有效,醫院和專科診所通常會優先為高風險患者植入 ICD。遠端監測和人工智慧預測警報等技術的進步進一步推動了這項需求,提高了病患安全性。 ICD(植入式心臟復律去顫器)與數位醫療平台相容,使臨床醫生能夠即時追蹤患者數據。該領域受益於完善的醫保報銷政策和臨床醫生的廣泛認可,鞏固了其市場主導地位。此外,電池壽命、小型化和無線通訊方面的持續創新也促進了市場接受度和收入成長。
植入式心臟監視器/植入式循環記錄儀領域預計將在2026年至2033年間實現最快增長,這主要得益於醫院和門診專科診所越來越多地採用此類設備進行早期心律失常和卒中風險檢測。這些設備可提供持續的長期監測,從而減少患者頻繁就醫的需求。患者越來越傾向於選擇微創監測方案,因為這些方案更方便舒適。與行動應用程式和雲端平台的整合實現了遠端資料共享和預測分析,使這些設備對臨床醫生和患者都更具吸引力。人們對早期心血管疾病檢測意識的提高以及有利的報銷政策也進一步促進了市場的快速成長。此外,該領域還湧現出許多技術創新,例如設備尺寸的縮小和電池壽命的延長,從而擴大了其在老年人和高危險群中的吸引力。
- 依手術類型
根據手術類型,市場可分為傳統手術和微創手術。由於併發症風險低、恢復快、住院時間短,微創手術預計在2025年佔據市場主導地位。醫院和專科診所越來越傾向於以微創方式植入心臟、神經血管和骨科器械,從而提高病患滿意度和臨床療效。外科醫生正在採用先進的影像技術和機器人輔助技術來提高手術精度。患者對微創手術的偏好日益增長,也推動了微創手術的普及。微創手術與大多數現代植入式器械相容,這為其持續成長提供了支持。此外,醫療報銷機制和臨床指引通常也傾向於微創方法,進一步鞏固了微創手術的市場主導地位。
預計在2026年至2033年期間,傳統手術方法細分市場將實現最快成長,尤其是在外科手術技術成熟的地區以及需要複雜植入的器械(例如心室輔助裝置或高級神經刺激器)領域。由於熟悉傳統技術且療效顯著,醫院在進行高風險或多器械植入手術時仍會繼續採用傳統技術。持續的培訓項目、流程標準化和臨床方案更新將促進該細分市場的發展。傳統方法還允許在需要時同時植入多個器械。此外,越來越多的臨床研究支持透過傳統手術方法獲得器械療效,從而推動了該方法的應用。患者意識的提高和醫生對複雜植入手術信心的增強也促進了該細分市場的預期增長。
- 按程式
根據手術類型,市場可細分為神經血管、心血管、聽力及其他領域。心血管手術領域預計在2025年佔據市場主導地位,市場份額最大,這主要得益於美國心臟衰竭、心律不整和心臟驟停的高發病率。醫院優先使用ICD、心律調節器和CRT-D等設備進行救命介入。心血管植入設備受益於完善的醫療覆蓋範圍和臨床醫生的熟悉程度。這些設備的持續監測和遠端連接功能可提高患者依從性和臨床療效。設備小型化、人工智慧監測和無線數據傳輸等方面的技術進步進一步促進了其應用。製造商也高度重視該領域的創新,致力於提高可靠性、安全性和患者體驗,從而鞏固其市場主導地位。
由於兒童和老年族群中聽力障礙盛行率不斷上升,預計2026年至2033年間,聽力手術領域將迎來最快成長。包括人工耳蝸和骨錨式助聽系統在內的主動式植入式助聽設備在醫院和專科診所的應用日益廣泛。與數位平台和行動應用程式的整合實現了遠端調音和監測,提高了患者的便利性。宣傳活動的開展和聽力保健服務可近性的提高正在推動這些設備的普及。更小的植入體尺寸和更先進的聲音處理演算法等技術進步,提升了該領域的吸引力。此外,公共和私人醫療保健系統報銷範圍的擴大也為預計的快速成長提供了支持。
- 最終用戶
根據最終用戶,市場可細分為醫院、專科診所、門診手術中心和一般診所。由於醫院擁有先進的手術基礎設施、技術精湛的臨床人員以及植入式醫療器材手術量大,預計到2025年,醫院將佔據市場主導地位。醫院負責處理複雜的心臟、神經血管和骨科植入手術,使其成為ICD和CRT-D等高價值醫療器材的主要治療場所。與醫院IT系統的整合可實現即時監測和資料管理。強有力的報銷支持和長期的患者追蹤服務進一步鞏固了醫院的市場主導地位。此外,醫院還扮演著器材試驗、培訓和新技術應用創新中心的角色,從而保持領先地位。
預計在2026年至2033年期間,專科診所領域將實現最快成長,主要受微創植入手術和門診監測解決方案需求成長的推動。專科診所專注於心臟病、神經病學和聽力障礙等領域,提供便利且個人化的治療。患者更傾向於選擇診所進行後續護理、設備調整和遠端監測。該領域受益於先進的診斷工具、行動醫療平台以及與設備製造商的合作。患者意識的提高、醫療費用的可負擔性以及專科醫療服務可近性的改善,都推動了這個終端用戶領域的快速擴張。
北美有源醫療植入設備市場區域分析
- 預計到2025年,美國將以74.9%的最大市場份額主導北美主動式醫療植入設備市場,其特點是擁有先進的醫療基礎設施、高額的醫療支出以及主要行業參與者的強大影響力。
- 該地區的患者和醫療保健提供者高度重視植入式裝置(如ICD、心臟節律器和神經刺激器)所提供的精準性、持續監測和改善的臨床效果。
- 這種廣泛應用得益於先進的醫院和專科診所、技術精湛的醫護人員以及人們對微創手術日益增長的認識,從而使植入式醫療器械成為醫院和門診環境中不可或缺的治療解決方案。
美國主動式醫療植入設備市場洞察
2025年,美國主動植入式醫療器材市場在北美地區佔據了最大的市場份額,達到74.9%,這主要得益於先進心臟、神經刺激器和骨科器械的廣泛應用。由於ICD、心律調節器和CRT-D具有挽救生命的潛力和精準的治療能力,醫院和專科診所越來越重視這些器械。患者和醫療服務提供者正在採用微創手術和人工智慧賦能的監測系統,從而改善治療效果。人們對慢性疾病的日益關注,以及有利的醫療保險報銷機制,正在推動市場擴張。此外,與遠端監測和雲端醫療平台的整合也顯著促進了器械的普及。成熟的器材製造商的強大實力和持續的技術創新也進一步支撐了市場的成長。
加拿大主動式醫療植入設備市場洞察
預計在預測期內,加拿大主動式植入式醫療器材市場將成為北美成長最快的市場,這主要得益於醫療保健投資的增加、心血管和神經系統疾病盛行率的上升以及人們對植入式療法的認知度不斷提高。醫院和專科診所正在採用微創植入手術和遠端監測解決方案來改善病患照護。政府為提高先進醫療技術的可近性而採取的舉措,以及患者對門診和專科診所服務日益增長的需求,都促進了這些技術的快速普及。此外,醫療基礎設施的改善和臨床醫生培訓計畫的加強也對市場發展起到了積極作用。人工智慧除顫器、神經刺激器和遠端患者監測設備等技術進步也推動了市場的加速成長。
墨西哥有源醫療植入設備市場洞察
墨西哥有源植入式醫療器材市場正穩定成長,這主要得益於人們對慢性心血管疾病和神經系統疾病的認識不斷提高,以及先進醫療服務的普及。城市中心的醫院和專科診所正在採用植入式心臟裝置、神經刺激器和聽力植入物來改善病患的治療效果。政府為改善醫療基礎設施和公共衛生計畫所採取的措施也促進了市場擴張。患者越來越傾向於微創手術和遠端監測解決方案,從而提高了這些設備的普及率。本地分銷網路以及與國際醫療器械製造商的合作提高了設備的供應量。此外,持續增加對醫療專業人員培訓的投入並擴大醫保報銷範圍,也推動了墨西哥在北美市場份額的成長。
北美有源醫療植入設備市場份額
北美有源醫療植入器材產業主要由一些成熟企業引領,其中包括:
- 美敦力(愛爾蘭)
- 雅培(美國)
- 波士頓科學公司(美國)
- 科利耳有限公司(澳洲)
- 百多力(德國)
- LivaNova PLC(英國)
- MED EL 醫療電子(奧地利)
- 索諾瓦(瑞士)
- Axonics公司(美國)
- NeuroPace公司(美國)
- NEVRO 公司(美國)
- 浙江紐朗生物科技有限公司(中國)
- Demant A/S(丹麥)
- 奧迪康醫療(丹麥)
- 索諾瓦控股股份公司(瑞士)
- Microson(澳洲)
- Nano Retina(以色列)
- GluSense(美國)
- 第二視線(美國)
北美主動式醫療植入設備市場近期有哪些發展動態?
- 2025 年 11 月,波士頓科學公司宣布將以約 5.33 億美元收購 Nalu Medical 的剩餘股權,透過整合 Nalu 的周邊神經刺激技術來擴展其慢性疼痛神經刺激產品組合。該技術旨在透過智慧型手機應用程式控制的無線、無電池植入物提供有針對性的疼痛緩解。
- 2025年7月,科利耳有限公司宣布其Cochlear™ Nucleus® Nexa™系統已獲得FDA核准。該系統是全球首款智慧人工耳蝸植入系統,其內部韌體可升級,無需更換硬體即可實現未來的功能更新。這項創新使用戶能夠隨著時間的推移獲得新的增強功能,減少了更換外部硬體的需求,並憑藉更小、更輕的處理器提升了整體聽力性能。
- 2025年2月,美國食品藥物管理局(FDA)批准了美敦力公司研發的首款用於治療巴金森氏症的自適應腦節律器,這標誌著腦機介面技術領域的一個重要里程碑。這款自適應裝置能夠即時回應患者的需求,減少帕金森氏症患者的不自主運動,並改善症狀管理,凸顯了FDA對先進神經植入技術的支持。
- 2024年1月,馬斯克的Neuralink公司成功為首位人體患者植入了腦機介面晶片,啟動了早期臨床試驗,旨在幫助癱瘓或神經系統疾病患者僅憑意念即可與電腦和數位工具進行互動。這標誌著植入式腦機介面在實際臨床應用中取得了重大進展。
- 2023年9月,Neuralink啟動了其首個植入式腦機介面人體臨床試驗的受試者招募工作。此前,該介面已於同年稍早獲得美國食品藥物管理局(FDA)的批准。該試驗旨在評估其安全性和功能性,標誌著植入式神經技術在美國醫療應用領域中邁出了重要的監管和臨床里程碑。
SKU-
研究方法
数据收集和基准年分析是使用具有大样本量的数据收集模块完成的。该阶段包括通过各种来源和策略获取市场信息或相关数据。它包括提前检查和规划从过去获得的所有数据。它同样包括检查不同信息源中出现的信息不一致。使用市场统计和连贯模型分析和估计市场数据。此外,市场份额分析和关键趋势分析是市场报告中的主要成功因素。要了解更多信息,请请求分析师致电或下拉您的询问。
DBMR 研究团队使用的关键研究方法是数据三角测量,其中包括数据挖掘、数据变量对市场影响的分析和主要(行业专家)验证。数据模型包括供应商定位网格、市场时间线分析、市场概览和指南、公司定位网格、专利分析、定价分析、公司市场份额分析、测量标准、全球与区域和供应商份额分析。要了解有关研究方法的更多信息,请向我们的行业专家咨询。
可定制
Data Bridge Market Research 是高级形成性研究领域的领导者。我们为向现有和新客户提供符合其目标的数据和分析而感到自豪。报告可定制,包括目标品牌的价格趋势分析、了解其他国家的市场(索取国家列表)、临床试验结果数据、文献综述、翻新市场和产品基础分析。目标竞争对手的市场分析可以从基于技术的分析到市场组合策略进行分析。我们可以按照您所需的格式和数据样式添加您需要的任意数量的竞争对手数据。我们的分析师团队还可以为您提供原始 Excel 文件数据透视表(事实手册)中的数据,或者可以帮助您根据报告中的数据集创建演示文稿。