
The outbreak of coronavirus highlights the need of rapid and effective test method in order to detect and identify the target virus.
WHO has validated two types of test for detection of COVID 19 one is molecular diagnostic test and other one is immunological assay test. But as molecular diagnostic test such as PCR among others cannot be used for monitoring of disease progressing stage, there is a great need of immunological assays.
The development of rapid test method such as immunoassay among others has become an urgent need for effective drug and vaccine development so as to curtail the spread of COVID 19.
The need of immunoassay kit for reliable COVID 19 test led the country’s government to reduce the price of these tests, so that individual belonging to any economic status can afford such tests.
Moreover some of the immunoassay kits were imported from different regions of the world, however due to ban and lockdown across borders import of testing kits also stopped. Along with this, the manufacturing of immunoassay kits in the country itself has also caused price reduction.
Immunoassay tests in order to recognize the viral antigen and related immunoglobulin are free at government laboratories, which again caused price reduction of kits.
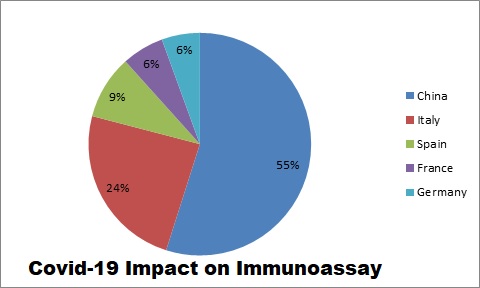
For instance:
Thus, it signifies that due to COVID 19 pandemic, the price of immunoassays kits and related products will reduce but product sales will increase.
This has been reported that RT-PCR based viral RNA detection test are widely used for diagnosis of COVID-19, however in order to monitor the progress of disease and identification of different stages there is a great need of immunological assays.
The results provided by immunological tests have an important role in vaccine development, which again increases the demand of immunoassay test.
Lateral flow immunoassay is one of the rapid diagnostic tests used for detection of viral antigens within 10-30 minutes. Moreover this is an inexpensive test and does not require any trained personnel for performing the test. All such factors again enhance the demand of immunoassay tests.
ELISA test can also be used for detection of viral components. ELISA is compatible with various kinds of samples that increase its demand for COVID 19 test. Moreover it is suitable for point of care detection.
Luminescent immunoassay has been reported in excessive use for COVID 19 diagnosis and manufacturers are also taking steps in order to fulfill such increasing demand.
For instance,
Moreover increasing prevalence of COVID-19 all around the world is also responsible for increasing demand of immunoassay kits.
Figure1: Number of Laboratory Confirmed COVID-19 Cases
Source: WHO
All these factors suggest that COVID 19 outbreak has enhanced the demand of immunoassay market.
Due to continuous COVID-19 outbreak, several restrictions have been imposed by government bodies of several countries which involves ban throughout the country’s borders among others. These restrictions led to shortage of in the supply of immunoassay kits.
While such lockdown Indian government has allowed import of certain immunoassay kits used for detection of coronavirus. As per the report published in May, 2020 Abbott Healthcare and Roche Diagnostics received approval for import of immunoassay kits. This approval involves the import of chemiluminescent immunoassay kits. This step by Indian government helped to maintain a continuous supply chain amidst the COVID-19 outbreak.
The Asian Development Bank in March 2020, decided to provide with USD 200 million for companies who are engaged in manufacturing of coronavirus related products involving immunoassay kits. This step by organization helps the company to ensure continuous supply chain of their products.
This signifies that even during the COVID-19 pandemic companies are able to maintain supply chain.
Strategic Initiatives by Manufacturers and Organizations:
Due to increasing demand of immunoassay kits across the world, the market players are launching various types of immunoassay kits.
For instance,
This signifies that several strategic initiatives by different manufacturers in order to fight against novel coronavirus, fuel up the market growth.
| Test Name | Test Type | Manufacturer | Sample Sources | Test Result Time |
|---|---|---|---|---|
| m2000 SARS-CoV-2 assay | chemiluminescent microparticle immunoassay | Abbott Core Laboratory | serum/plasma/whole blood | runs up to 100-200 tests/h |
| COVID-19 IgG/IgM LF | lateral flow immunoassay | Advagen Biotech | serum/plasma/whole blood | results in 10 min |
| COVID-19 IgM/IgG rapid test | lateral flow immunoassay | BioMedomics | serum/plasma/whole blood | results in 15 min |
| One-Step COVID-2019 test | lateral flow immunoassay | Celer Biotechnologia | serum/plasma/whole blood | results in 15 min |
| qSARS-CoV-2 IgG/IgM rapid test | lateral flow immunoassay | Cellex Inc. | serum/plasma/whole blood | results in 15-20 min |
| DEIASL019/020 SARS-CoV-2 IgG ELISA kit | ELISA | Creative Diagnostics | serum/plasma | - |
| OnSite COVID-19 IgG/IgM rapid test | lateral flow immunoassay | CTK Biotech Inc. (USA) | serum/plasma/whole blood | results in 10 min |
Source: NCBI
As per the data provided in table large number of products available in order to perform immunoassays, this paves the way for manufacturers to earn more product sales, which again boost up the market growth.
Amid the COVID -19 outbreaks, various regulations associated with immunoassay test kit launch has been relaxed by FDA and other regulatory bodies due to increasing demand of rapid test for coronavirus detection.
FDA is aware of that several manufacturers are developing immunoassay test for coronavirus with an aim of submission of Emergency Use Authorization (EUA) to FDA. As the need of such test increasing, FDA is not intended for reasonable specimen testing, which consumes a lot of time.
FDA offers 15 business days to manufacturer for preparation of EUA submission for an immunoassay test whose performance has already been validated by the manufacturer.
With the urging need of worldwide population FDA has engaged with more than 100 of test developing manufacturer since the end of January and has granted EUA to several of immunoassay manufacturers.
During this pandemic FDA ensures that these test kits distribute to every labs, hospital land to other facilities which are in need of it.
Moreover FDA is working 24*7 in order to support laboratories and test developers, so that the manufacturers can distribute immunoassay test kits throughout the several countries at this urgent time.
In April, European Commission has issued a directive related to medical devices used for coronavirus testing. According to the directive launched in April if an assay meets all the requirements mentioned in directive launched by European Commission then there is no need for manufacturer to get a CE mark for that assay.
In Asia-Pacific region, after the initial outbreak of COVID 19 South Korea’s Centers for Disease Control and Prevention coordinated with the manufacturers engaged in getting approval for assay used for COVID 19 testing. Moreover in China, Singapore and Australia regulatory guidelines have been adjusted in order to get early approval and emergency use authorization for assays.
As strict regulations have been relaxed by regulatory authorities for immunoassay test kits during the COVID-19 pandemic, thus signifies that coronavirus has paved the way for the growth of immunoassay market.
The pandemic of COVID 19 has led to an urgent need for the detection of coronavirus to progress the disease stage and to develop a novel vaccine, which can be possible by using immunoassay kits that provides rapid detection of antibodies formed against coronavirus. This led to the increasing demand for immunoassay kits and enhanced the growth of the immunoassay market. Various types of bans resulted in a shortage of product supplies that are expected to hamper the market growth, however, initiatives taken by the government related to product import and investment by some authorities helped the manufacturers to maintain the supply chain of immunoassay products. Increasing the prevalence of COVID-19 and the availability of immunoassay products is also expected to fuel up the market growth. Moreover, support by regulatory authorities related to approval and emergency use authorization for immunoassay products helping the market players to achieve a lucrative growth.