Global Catheter Stabilization Devicecatheter Securement Devices Market
Market Size in USD Billion
CAGR :
% 
 USD
1.81 Billion
USD
3.45 Billion
2024
2032
USD
1.81 Billion
USD
3.45 Billion
2024
2032
| 2025 –2032 | |
| USD 1.81 Billion | |
| USD 3.45 Billion | |
|
|
|
|
Catheter Stabilization Device/Catheter Securement Devices Market Size
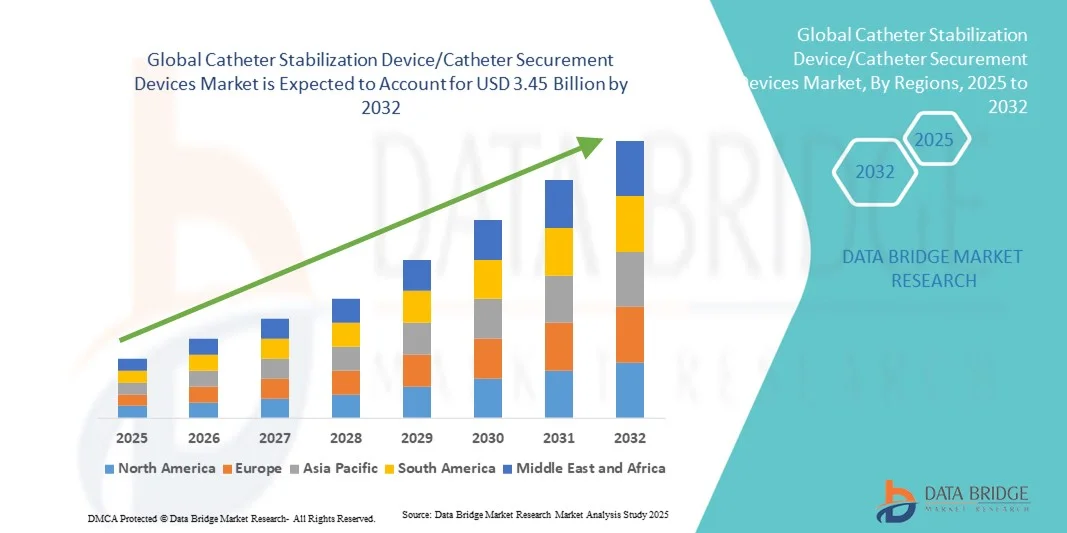
- The global catheter stabilization device/catheter securement devices market size was valued at USD 1.81 billion in 2024 and is expected to reach USD 3.45 billion by 2032, at a CAGR of 8.4% during the forecast period
- This market growth is primarily driven by the increasing prevalence of chronic diseases, the rising number of surgical procedures, and the growing demand for home-based and self-care solutions. These factors collectively enhance the need for secure and effective catheter stabilization methods
- Furthermore, advancements in medical technology and a heightened focus on infection prevention are establishing catheter stabilization devices as essential components in modern healthcare settings. These converging factors are accelerating the adoption of securement solutions, thereby significantly boosting the industry's growth
Catheter Stabilization Device/Catheter Securement Devices Market Analysis
- Catheter stabilization devices, designed to securely anchor catheters and prevent movement-related complications, are increasingly vital in modern healthcare settings, including hospitals, clinics, and home care, due to their role in reducing infection risks, improving patient comfort, and ensuring procedural efficiency
- The escalating demand for catheter stabilization devices is primarily fueled by the rising prevalence of chronic diseases requiring long-term catheter use, growing surgical procedures, and increasing focus on patient safety and infection prevention
- North America dominated the catheter stabilization device market with the largest revenue share of 37% in 2024, characterized by early adoption of advanced medical technologies, high healthcare spending, and stringent regulations promoting patient safety, with the U.S. experiencing substantial adoption of innovative securement devices across hospitals and home care settings
- Asia-Pacific is expected to be the fastest-growing region in the catheter stabilization device market during the forecast period due to expanding healthcare infrastructure, rising disposable incomes, and increasing awareness of catheter-related infection prevention
- The Peripheral Catheter Securement Devices segment dominated the catheter stabilization device market with a market share of 37.87% in 2024, driven by their broad usage in medical procedures, ease of application, and proven effectiveness in reducing catheter dislodgement and associated complications
Report Scope and Catheter Stabilization Device/Catheter Securement Devices Market Segmentation
|
Attributes |
Catheter Stabilization Device/Catheter Securement Devices Key Market Insights |
|
Segments Covered |
|
|
Countries Covered |
North America
Europe
Asia-Pacific
Middle East and Africa
South America
|
|
Key Market Players |
|
|
Market Opportunities |
|
|
Value Added Data Infosets |
In addition to the insights on market scenarios such as market value, growth rate, segmentation, geographical coverage, and major players, the market reports curated by the Data Bridge Market Research also include in-depth expert analysis, pricing analysis, brand share analysis, consumer survey, demography analysis, supply chain analysis, value chain analysis, raw material/consumables overview, vendor selection criteria, PESTLE Analysis, Porter Analysis, and regulatory framework. |
Catheter Stabilization Device/Catheter Securement Devices Market Trends
Advancements in Sutureless and Adhesive-Based Securement Solutions
- A key trend in the global catheter stabilization device market is the increasing development and adoption of sutureless, adhesive-based, and suture-free securement solutions, which reduce the risk of infection, improve patient comfort, and simplify catheter management
- For instance, StatLock and 3M Tegaderm devices offer adhesive securement options that eliminate the need for sutures, minimizing skin trauma and lowering the incidence of catheter-related bloodstream infections (CRBSIs)
- Innovations in materials and design are enabling more reliable stabilization for both short-term and long-term catheters, while maintaining ease of application and removal for healthcare providers
- These solutions also support integration with monitoring systems to track catheter position and detect accidental dislodgement, enhancing patient safety and reducing healthcare costs
- The trend towards more patient-friendly, efficient, and infection-preventive securement solutions is reshaping expectations for standard catheter care. Companies such as Becton Dickinson and ICU Medical are developing next-generation adhesive-based and sutureless devices with improved adhesion, skin compatibility, and securement reliability
- Demand for these advanced catheter stabilization devices is growing rapidly across hospitals, outpatient care, and home healthcare settings, driven by the need to improve clinical outcomes and reduce hospital-acquired complications
Catheter Stabilization Device/Catheter Securement Devices Market Dynamics
Driver
Increasing Focus on Infection Prevention and Patient Safety
- The rising prevalence of catheter-related infections and complications is a primary driver for the adoption of catheter stabilization devices across healthcare settings
- For instance, in 2024, ICU Medical expanded its portfolio of securement devices aimed at reducing catheter dislodgement and bloodstream infections, reflecting the emphasis on patient safety
- Healthcare providers are prioritizing devices that reduce infection risk, improve patient comfort, and streamline catheter management, fueling market growth
- Furthermore, the growing number of surgical procedures, long-term intravenous therapies, and home healthcare services are increasing the demand for reliable catheter securement solutions
- The increasing trend of home-based care for chronic and long-term catheter patients is driving demand for easy-to-use, reliable securement devices that allow caregivers and patients to manage catheters safely outside hospital environments
- Features such as sutureless application, adhesive-based securement, and compatibility with various catheter types are key factors propelling adoption in hospitals, clinics, and home care settings
Restraint/Challenge
Skin Irritation Issues and Regulatory Compliance Hurdle
- Challenges in the catheter stabilization device market include potential skin irritation or allergic reactions caused by adhesive materials, which can limit use in sensitive patients or require specialized product options
- For instance, reports of dermatitis or skin breakdown in long-term catheter patients have prompted manufacturers to develop hypoallergenic and skin-friendly alternatives
- Regulatory hurdles and the need for compliance with stringent medical device standards in different regions can delay product approvals and market entry, affecting growth
- In addition, high costs of advanced securement devices compared to conventional methods, such as sutures or tapes, may limit adoption in price-sensitive healthcare settings or developing regions
- Some advanced securement devices, particularly sutureless or adhesive-based systems, require proper training for healthcare providers to ensure correct application and avoid device failure, which can slow adoption in smaller clinics or home care settings
- Addressing these challenges through the development of skin-safe adhesives, regulatory alignment, and cost-effective solutions will be essential for sustained market expansion
Catheter Stabilization Device/Catheter Securement Devices Market Scope
The market is segmented on the basis of product, application, and end-user.
- By Product
On the basis of product, the catheter stabilization device market is segmented into arterial catheter securement devices, central venous catheter securement devices, peripheral catheter securement devices, urinary catheter securement devices, abdominal drainage tubes securement devices, epidural securement devices, chest drainage tube securement devices, all-site securement devices, and others. Peripheral Catheter Securement Devices dominated the market with the largest market revenue share of 37.87% in 2024, driven by their extensive use in hospitals, outpatient care, and home healthcare settings. These devices are ideal for short-term procedures, offering easy application and removal, reducing caregiver workload, and improving patient comfort. Their cost-effectiveness and broad availability make them preferred choices in both developed and developing markets. In addition, the growth of home healthcare and outpatient treatments has fueled adoption. Continuous improvements in adhesive materials and device design have enhanced stability, reliability, and overall patient safety. Peripheral catheter securement devices also benefit from widespread clinical familiarity and minimal training requirements for healthcare staff.
The Central Venous Catheter Securement Devices segment is anticipated to witness the fastest growth rate from 2025 to 2032, fueled by the rising prevalence of chronic diseases, cancer therapies, and intensive care treatments. These devices are critical for long-term treatments and complex procedures, providing enhanced stability and reducing the risk of catheter-related complications. Stricter healthcare regulations and the need for high-quality securement solutions drive adoption. Companies are investing in research and development to improve device performance, usability, and patient comfort. The global rise in surgeries and long-term intravenous therapies is further accelerating the growth of this segment. Advanced features and innovations in design make central venous securement devices increasingly essential in critical care and specialty hospitals.
- By Application
On the basis of application, the catheter stabilization device market is segmented into cardiovascular, respiratory, urological, gastric, and radiology. The Cardiovascular segment dominated the market with the largest revenue share in 2024, primarily due to the high frequency of catheterization procedures in cardiac care. Cardiovascular interventions are critical, necessitating stable and reliable catheter securement. Advanced technologies and continuous monitoring in cardiovascular care increase reliance on effective devices. Well-established cardiovascular departments in hospitals drive consistent demand. Strict regulatory standards ensure the use of safe and effective securement solutions. The integration of modern medical technologies has further reinforced the importance of these devices in cardiovascular procedures.
The Urological segment is expected to witness the fastest growth during the forecast period due to the increasing elderly population and rising prevalence of chronic urological conditions. Frequent catheterization for conditions such as urinary retention drives demand for securement solutions. The trend toward home healthcare for urological patients is expanding the market. Innovations in device design enhance patient comfort and reduce skin irritation. Specialized devices for urological applications are being developed through ongoing research. Supportive healthcare policies for homecare indirectly contribute to growth in this segment.
- By End-User
On the basis of end-user, the catheter stabilization device market is segmented into hospitals, homecare settings, diagnostic imaging centers, and others. Hospitals dominated the market in 2024, as they remain the primary settings for catheterization procedures. Hospitals have advanced equipment, skilled personnel, and high patient volumes, necessitating reliable securement devices. Compliance with regulatory standards drives adoption of effective solutions. Hospitals often participate in clinical trials, encouraging early adoption of innovative technologies. Established infrastructure ensures consistent procurement and usage. The critical nature of inpatient care and surgeries reinforces hospitals as the primary end-user segment.
The Homecare Settings segment is anticipated to witness the fastest growth rate from 2025 to 2032, driven by increasing patient preference for home-based care and the rising prevalence of chronic diseases. Homecare is more cost-effective than prolonged hospital stays, promoting adoption of user-friendly securement devices. Technological advancements allow safe and effective management of catheters outside hospitals. Manufacturers are developing easy-to-use products for patients and caregivers. Supportive healthcare policies indirectly boost market growth. The aging population and growing demand for outpatient care further drive adoption in homecare settings.
Catheter Stabilization Device/Catheter Securement Devices Market Regional Analysis
- North America dominated the catheter stabilization device market with the largest revenue share of 37% in 2024, characterized by early adoption of advanced medical technologies, high healthcare spending, and stringent regulations promoting patient safety, with the U.S. experiencing substantial adoption of innovative securement devices across hospitals and home care settings
- Healthcare providers in the region prioritize devices that reduce catheter-related complications, improve patient comfort, and streamline clinical workflows, resulting in widespread adoption across hospitals, outpatient care centers, and home healthcare settings
- The presence of major global manufacturers and continuous product innovations, including sutureless and adhesive-based securement solutions, further strengthens market penetration in North America
U.S. Catheter Stabilization Device/Catheter Securement Devices Market Insight
The U.S. catheter stabilization device market captured the largest revenue share of 38% in 2024 within North America, fueled by the growing prevalence of chronic diseases, increasing surgical procedures, and rising patient awareness about catheter-related complications. Hospitals and outpatient care centers are increasingly adopting advanced securement devices to reduce infection risks and improve patient comfort. The expanding trend of home healthcare is also contributing to market growth, as caregivers and patients require user-friendly, reliable securement solutions. In addition, technological innovations, such as sutureless and adhesive-based devices, are enhancing device efficiency and safety. Strong healthcare infrastructure, reimbursement policies, and the presence of leading global manufacturers further drive market expansion.
Europe Catheter Stabilization Device / Catheter Securement Devices Market Insight
The Europe catheter stabilization device market is projected to expand at a substantial CAGR throughout the forecast period, primarily driven by stringent medical device regulations and the rising focus on infection prevention and patient safety. The adoption of advanced securement solutions in hospitals, outpatient clinics, and homecare settings is growing due to increasing awareness of catheter-related bloodstream infections. Urbanization, rising healthcare expenditure, and the demand for technologically advanced devices are fostering market adoption. In addition, the region is witnessing growth across cardiovascular, urological, and critical care applications, with securement devices increasingly integrated into standard catheter management protocols.
U.K. Catheter Stabilization Device / Catheter Securement Devices Market Insight
The U.K. catheter stabilization device market is anticipated to grow at a noteworthy CAGR during the forecast period, driven by the rising number of surgical procedures and increasing adoption of minimally invasive therapies requiring catheterization. Healthcare providers are emphasizing the use of advanced securement devices to enhance patient safety, reduce catheter dislodgement, and prevent infections. The U.K.’s well-established healthcare system, along with growing awareness of home healthcare services, is encouraging the adoption of easy-to-use securement devices. Furthermore, ongoing research and technological innovations in catheter stabilization solutions are expected to stimulate market growth.
Germany Catheter Stabilization Device / Catheter Securement Devices Market Insight
The Germany catheter stabilization device market is expected to expand at a considerable CAGR during the forecast period, fueled by increasing awareness of catheter-related infection risks and the need for high-quality medical devices. Hospitals and specialty clinics in Germany are increasingly adopting sutureless and adhesive-based securement solutions to improve patient outcomes and reduce clinical complications. Germany’s advanced healthcare infrastructure and emphasis on technological innovation support the adoption of sophisticated securement devices. Integration of these devices with monitoring systems and enhanced patient safety protocols is becoming increasingly prevalent, particularly in critical care and cardiovascular procedures.
Asia-Pacific Catheter Stabilization Device / Catheter Securement Devices Market Insight
The Asia-Pacific catheter stabilization device market is poised to grow at the fastest CAGR of 9.8% during the forecast period of 2025 to 2032, driven by rising healthcare expenditure, increasing hospital infrastructure, and rapid urbanization in countries such as China, India, and Japan. The growing prevalence of chronic diseases and the rising number of surgical procedures are boosting demand for catheter securement devices. In addition, increasing awareness about catheter-related infections and the expansion of home healthcare services are further propelling adoption. The availability of cost-effective solutions and the presence of both domestic and global manufacturers are facilitating wider accessibility and market penetration.
Japan Catheter Stabilization Device / Catheter Securement Devices Market Insight
The Japan catheter stabilization device market is gaining momentum due to the country’s advanced healthcare infrastructure, aging population, and high adoption of minimally invasive procedures requiring catheterization. Hospitals and homecare providers are increasingly implementing advanced securement devices to reduce infection risks and enhance patient comfort. Integration with monitoring systems and focus on patient-centered care are driving adoption in both residential and commercial healthcare settings. In addition, technological innovation and government initiatives promoting healthcare safety standards are contributing to market growth.
India Catheter Stabilization Device / Catheter Securement Devices Market Insight
The India catheter stabilization device market accounted for the largest market revenue share in Asia-Pacific in 2024, attributed to the expanding middle-class population, rapid urbanization, and rising rates of surgical and chronic care procedures. Hospitals, diagnostic centers, and homecare providers are increasingly adopting catheter securement devices to improve patient outcomes and minimize catheter-related complications. Government initiatives promoting healthcare infrastructure, smart hospital projects, and increasing awareness about infection prevention are key factors propelling market growth. The availability of affordable securement solutions and a growing number of domestic manufacturers further supports the expansion of the Indian market.
Catheter Stabilization Device/Catheter Securement Devices Market Share
The Catheter Stabilization Device/Catheter Securement Devices industry is primarily led by well-established companies, including:
- BD (U.S.)
- B. Braun SE (Germany)
- Medtronic (Ireland)
- 3M (U.S.)
- Convatec Inc. (U.K.)
- Medline Industries, Inc. (U.S.)
- Merit Medical Systems (U.S.)
- Teleflex Incorporated (U.S.)
- Cook (U.S.)
- Vygon (France)
- Deltamed (Italy)
- Levity Products (U.S.)
- Argon Medical Devices (U.S.)
- TIDI Products, LLC (U.S.)
- Cardinal Health (U.S.)
- SecurePort Medical, LLC (U.S.)
- MARPAC Inc. (U.S.)
What are the Recent Developments in Global Catheter Stabilization Device/Catheter Securement Devices Market?
- In July 2025, AMSure introduced upgraded Foley kits featuring the FlexGRIP securement device, designed to improve patient comfort and minimize complications such as infection and catheter dislodgement. This development reflects a commitment to enhancing the safety and efficacy of catheterization procedures
- In May 2025, Vygon UK was announced as the new distributor for the NICE-approved, NHSE-endorsed SecurAcath catheter securement device. This partnership aims to expand the availability of this innovative device, which offers a secure and reliable solution for catheter stabilization in clinical settings
- In January 2025, B. Braun Medical Inc. introduced the Clik-FIX Epidural/PNB Catheter Securement Device, designed to enhance patient comfort and minimize catheter displacement during regional anesthesia procedures. This low-profile, secure device aims to improve both patient and healthcare professional experiences by simplifying application and ensuring reliable securement
- In September 2022, B. Braun Medical Inc. expanded its portfolio by acquiring the Clik-FIX catheter securement device line from Starboard Medical, Inc. This acquisition strengthens B. Braun’s commitment to advancing patient care in peripheral intravenous therapy by integrating innovative securement solutions
- In November 2021, CATHETRIX unveiled the Foley/Safe 2.0, the world’s first Foley catheter stabilizer designed to actively prevent accidental extractions. This innovative device aims to enhance patient safety by securely anchoring catheters, thereby reducing the risk of unintentional dislodgement
SKU-
Get online access to the report on the World's First Market Intelligence Cloud
- Interactive Data Analysis Dashboard
- Company Analysis Dashboard for high growth potential opportunities
- Research Analyst Access for customization & queries
- Competitor Analysis with Interactive dashboard
- Latest News, Updates & Trend analysis
- Harness the Power of Benchmark Analysis for Comprehensive Competitor Tracking
Research Methodology
Data collection and base year analysis are done using data collection modules with large sample sizes. The stage includes obtaining market information or related data through various sources and strategies. It includes examining and planning all the data acquired from the past in advance. It likewise envelops the examination of information inconsistencies seen across different information sources. The market data is analysed and estimated using market statistical and coherent models. Also, market share analysis and key trend analysis are the major success factors in the market report. To know more, please request an analyst call or drop down your inquiry.
The key research methodology used by DBMR research team is data triangulation which involves data mining, analysis of the impact of data variables on the market and primary (industry expert) validation. Data models include Vendor Positioning Grid, Market Time Line Analysis, Market Overview and Guide, Company Positioning Grid, Patent Analysis, Pricing Analysis, Company Market Share Analysis, Standards of Measurement, Global versus Regional and Vendor Share Analysis. To know more about the research methodology, drop in an inquiry to speak to our industry experts.
Customization Available
Data Bridge Market Research is a leader in advanced formative research. We take pride in servicing our existing and new customers with data and analysis that match and suits their goal. The report can be customized to include price trend analysis of target brands understanding the market for additional countries (ask for the list of countries), clinical trial results data, literature review, refurbished market and product base analysis. Market analysis of target competitors can be analyzed from technology-based analysis to market portfolio strategies. We can add as many competitors that you require data about in the format and data style you are looking for. Our team of analysts can also provide you data in crude raw excel files pivot tables (Fact book) or can assist you in creating presentations from the data sets available in the report.