Global Endotoxin Tests Market
Market Size in USD Billion
CAGR :
% 
 USD
1.50 Billion
USD
2.88 Billion
2025
2033
USD
1.50 Billion
USD
2.88 Billion
2025
2033
| 2026 - 2033 | |
| USD 1.50 Billion | |
| USD 2.88 Billion | |
|
|
|
|
Endotoxin Tests Market Size
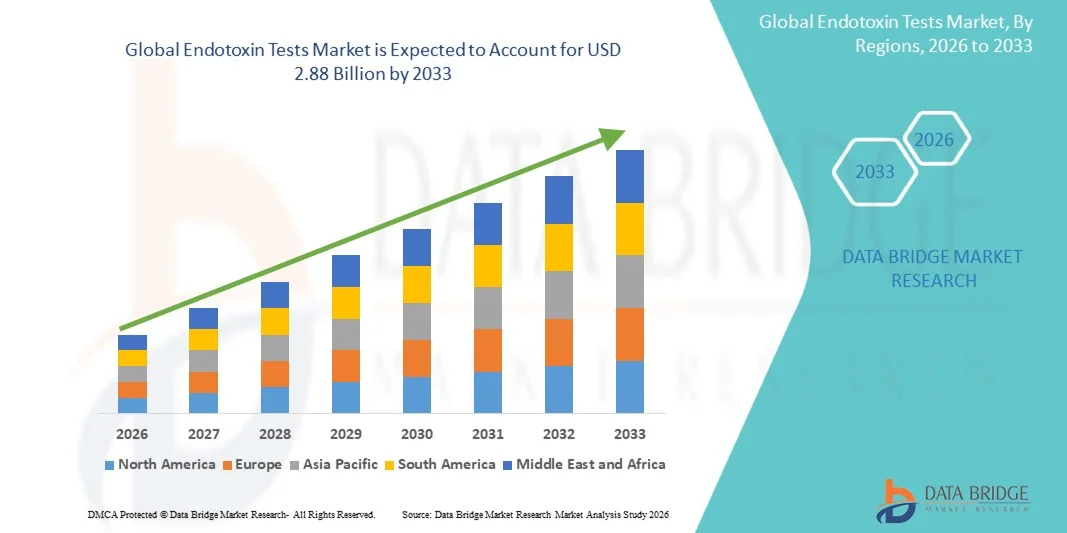
- The global Endotoxin Tests market size was valued at USD 1.50 billion in 2025and is expected to reach USD 2.88 billion by 2033, at a CAGR of8.50% during the forecast period
- The market growth is largely fueled by the increasing demand for pharmaceutical safety testing, rising biologics and vaccine production, and stringent regulatory requirements for endotoxin detection in medical products, leading to higher adoption of advanced endotoxin testing solutions across pharmaceutical and biotechnology industries
- Furthermore, growing focus on product quality assurance, increasing use of injectable drugs and implantable medical devices, and expanding application of rapid microbial testing technologies are establishing Endotoxin Tests solutions as a critical component of modern pharmaceutical manufacturing and healthcare quality control. These converging factors are accelerating the uptake of Endotoxin Tests solutions, thereby significantly boosting the industry's growth
Endotoxin Tests Market Analysis
- Endotoxin testing solutions, which include limulus amebocyte lysate (LAL) assays, recombinant factor C tests, chromogenic tests, and rapid endotoxin detection systems, are increasingly vital in pharmaceutical and biotechnology manufacturing due to their role in ensuring product sterility, patient safety, and regulatory compliance for injectable drugs and medical devices
- The escalating demand for endotoxin tests is primarily fueled by the growing production of biologics, vaccines, and injectable therapeutics, along with increasing regulatory scrutiny regarding pharmaceutical quality control and rising adoption of rapid microbial testing technologies across healthcare and life sciences industries
- North America dominated the endotoxin tests market with the largest revenue share of 38.4% in 2025, driven by strong pharmaceutical manufacturing capabilities, stringent FDA regulations, increasing biologics production, and high adoption of advanced quality assurance technologies, with the U.S. accounting for the majority of endotoxin testing activities and laboratory automation deployments
- Asia-Pacific is expected to be the fastest growing region in the endotoxin tests market during the forecast period due to expanding biopharmaceutical manufacturing, increasing vaccine production capacity, rising healthcare investments, and growing regulatory compliance requirements across countries such as China, India, South Korea, and Japan
- The LAL test segment accounted for the largest market revenue share of 61.3% in 2025, driven by its widespread acceptance as the standard method for bacterial endotoxin detection in pharmaceutical and medical device industries
Report Scope and Endotoxin Tests Market Segmentation
|
Attributes |
Endotoxin Tests Key Market Insights |
|
Segments Covered |
|
|
Countries Covered |
North America · U.S. · Canada · Mexico Europe · Germany · France · U.K. · Netherlands · Switzerland · Belgium · Russia · Italy · Spain · Turkey · Rest of Europe Asia-Pacific · China · Japan · India · South Korea · Singapore · Malaysia · Australia · Thailand · Indonesia · Philippines · Rest of Asia-Pacific Middle East and Africa · Saudi Arabia · U.A.E. · South Africa · Egypt · Israel · Rest of Middle East and Africa South America · Brazil · Argentina · Rest of South America |
|
Key Market Players |
· Charles River Laboratories (U.S.) · Lonza Group AG (Switzerland) · Thermo Fisher Scientific Inc. (U.S.) · Merck KGaA (Germany) · Associates of Cape Cod, Inc. (U.S.) · GenScript Biotech Corporation (China) · FUJIFILM Wako Pure Chemical Corporation (Japan) · BioMérieux SA (France) · WuXi AppTec (China) · Seikagaku Corporation (Japan) · Ellab A/S (Denmark) · Pacific BioLabs (U.S.) · Cambrex Corporation (U.S.) · Nelson Laboratories, LLC (U.S.) · Microcoat Biotechnologie GmbH (Germany) · Baker Hughes Company (U.S.) · Sanquin (Netherlands) · Hyglos GmbH (Germany) · Creative Bioarray (U.S.) · ToxinSensor (China) |
|
Market Opportunities |
· Rising Adoption of Recombinant Factor C (rFC) and Animal-Free Endotoxin Testing Methods · Expanding Biologics, Vaccine, and Injectable Drug Manufacturing Globally |
|
Value Added Data Infosets |
In addition to the insights on market scenarios such as market value, growth rate, segmentation, geographical coverage, and major players, the market reports curated by the Data Bridge Market Research also include in-depth expert analysis, patient epidemiology, pipeline analysis, pricing analysis, and regulatory framework. |
Endotoxin Tests Market Trends
“Growing Adoption of Rapid Microbial Detection and Automated Quality Control Technologies”
- A significant and accelerating trend in the global Endotoxin Tests market is the increasing adoption of rapid microbial detection technologies and automated quality control systems across pharmaceutical, biotechnology, and medical device manufacturing industries
- For instance, pharmaceutical manufacturers are increasingly utilizing rapid endotoxin testing methods, automated chromogenic assays, and recombinant factor C (rFC) technologies to improve testing efficiency, accuracy, and regulatory compliance
- The growing demand for biologics, injectable drugs, vaccines, and sterile medical products is driving increased implementation of advanced endotoxin detection systems to ensure product safety and contamination control
- Furthermore, the transition toward animal-free testing alternatives and sustainable laboratory practices is accelerating the adoption of recombinant endotoxin testing technologies as alternatives to traditional Limulus Amebocyte Lysate (LAL)-based assays
- The increasing integration of automated laboratory workflows and digital quality assurance systems is also improving testing throughput, reducing manual errors, and enhancing operational efficiency in pharmaceutical manufacturing facilities
- This trend toward rapid microbial testing, automation, and sustainable quality control solutions is reshaping pharmaceutical safety and compliance practices. Consequently, companies such as Charles River Laboratories and Lonza are expanding their endotoxin testing technology portfolios and laboratory automation capabilities
- The demand for advanced endotoxin testing solutions is increasing steadily due to stricter pharmaceutical safety regulations and growing global biologics production
Endotoxin Tests Market Dynamics
Driver
“Increasing Pharmaceutical Manufacturing and Stringent Regulatory Requirements”
- The expansion of pharmaceutical and biotechnology manufacturing activities is a major driver supporting the growth of the global Endotoxin Tests market
- For instance, the increasing production of injectable drugs, vaccines, biologics, and implantable medical devices is creating strong demand for reliable endotoxin testing procedures to ensure product sterility and patient safety
- Stringent regulatory guidelines from global health authorities regarding microbial contamination testing and pharmaceutical quality assurance are encouraging manufacturers to adopt advanced endotoxin detection technologies
- Furthermore, the rapid growth of biologics and cell & gene therapy development is increasing the need for sensitive and high-throughput endotoxin testing solutions throughout the drug development and manufacturing process
- The rising outsourcing of pharmaceutical manufacturing and laboratory testing services to contract development and manufacturing organizations (CDMOs) is also contributing to greater demand for standardized endotoxin testing systems
- In addition, increasing investments in healthcare infrastructure, pharmaceutical R&D, and vaccine manufacturing capacity are further supporting market expansion globally
Restraint/Challenge
“High Testing Costs and Technical Limitations of Traditional Assays”
- The high cost associated with advanced endotoxin testing systems and laboratory automation infrastructure remains a significant challenge affecting broader market adoption
- For instance, implementation of high-sensitivity testing instruments, automated analyzers, and recombinant testing platforms may require substantial capital investment, particularly for small and mid-sized pharmaceutical manufacturers
- In addition, traditional Limulus Amebocyte Lysate (LAL)-based assays face limitations such as sensitivity variability, interference from certain sample formulations, and dependence on horseshoe crab-derived biological materials
- The complexity of validating endotoxin testing procedures across diverse pharmaceutical products and manufacturing environments can also increase operational challenges and compliance burdens
- Furthermore, shortages of skilled laboratory professionals and stringent regulatory validation requirements may slow the adoption of newer endotoxin testing technologies in some regions
- Overcoming these challenges through development of cost-effective rapid testing platforms, expansion of recombinant testing technologies, improved laboratory automation, and stronger regulatory harmonization will be essential for sustaining long-term growth in the global Endotoxin Tests market
Endotoxin Tests Market Scope
The market is segmented on the basis of product type, test type, and application.
- By Product Type
On the basis of product type, the Endotoxin Tests market is segmented into endotoxin detection kits and reagents, instruments and systems, and consumables and accessories. The endotoxin detection kits and reagents segment dominated the largest market revenue share of 52.8% in 2025, driven by their extensive use in pharmaceutical quality control and biopharmaceutical manufacturing processes. These kits and reagents are essential for detecting bacterial endotoxins in injectable drugs, vaccines, and medical devices to ensure product safety and regulatory compliance. The segment benefits from increasing global pharmaceutical production and rising biologics manufacturing activities. Healthcare manufacturers are increasingly adopting rapid endotoxin detection solutions to reduce production downtime and improve testing efficiency. Strict regulatory requirements from agencies such as the FDA and EMA are significantly supporting market demand. Growing investment in sterile drug manufacturing infrastructure is further contributing to segment expansion. Technological advancements in chromogenic and turbidimetric assays are improving sensitivity and accuracy. Increasing outsourcing of pharmaceutical testing services is also strengthening product adoption. Furthermore, rising focus on contamination prevention and patient safety is expected to sustain segment dominance during the forecast period.
The instruments and systems segment is expected to witness the fastest CAGR of 11.7% from 2026 to 2033, driven by increasing laboratory automation and demand for high-throughput endotoxin testing platforms. Advanced instruments improve workflow efficiency, reduce human error, and enhance compliance with stringent quality standards. The segment benefits from growing adoption of automated endotoxin analyzers in pharmaceutical and biotechnology laboratories. Rising investment in modern laboratory infrastructure is significantly supporting market growth. Manufacturers are increasingly integrating digital monitoring and AI-enabled analytics into testing systems for improved precision and traceability. Expansion of biologics and vaccine production facilities is further accelerating demand for advanced testing equipment. Technological innovations in portable and rapid endotoxin testing devices are also contributing to market expansion. Increasing preference for automated quality assurance systems is strengthening adoption across regulated industries. Furthermore, rising focus on operational efficiency and contamination control is expected to drive strong segment growth during the forecast period.
- By Test Type
On the basis of test type, the Endotoxin Tests market is segmented into LAL test, MAT test, rabbit pyrogen test, and recombinant factor C (rFC) assay. The LAL test segment accounted for the largest market revenue share of 61.3% in 2025, driven by its widespread acceptance as the standard method for bacterial endotoxin detection in pharmaceutical and medical device industries. Limulus Amebocyte Lysate (LAL) testing is extensively utilized due to its high sensitivity, rapid results, and strong regulatory validation. The segment benefits from increasing production of injectable drugs, vaccines, and biologics requiring stringent endotoxin monitoring. Pharmaceutical manufacturers heavily rely on LAL testing for batch release and quality assurance procedures. Regulatory agencies continue to recommend LAL-based assays for endotoxin detection, further supporting adoption. Technological advancements in kinetic chromogenic and turbidimetric LAL assays are improving testing efficiency. Rising global healthcare expenditure and expansion of sterile manufacturing facilities are also contributing to market growth. Increasing focus on patient safety and contamination prevention is further strengthening demand. Furthermore, continued reliance on validated endotoxin detection standards is expected to sustain segment dominance during the forecast period.
The recombinant factor C (rFC) assay segment is expected to witness the fastest CAGR of 13.5% from 2026 to 2033, driven by increasing demand for sustainable and animal-free endotoxin testing alternatives. Recombinant factor C assays eliminate the need for horseshoe crab blood harvesting, making them environmentally sustainable and ethically favorable. The segment benefits from growing regulatory acceptance of alternative endotoxin testing methods. Pharmaceutical companies are increasingly adopting rFC assays to align with sustainability and ESG initiatives. Technological advancements in recombinant protein engineering are improving assay sensitivity and reliability. Rising investment in next-generation quality control technologies is significantly supporting market growth. Biopharmaceutical manufacturers are increasingly utilizing rFC platforms for rapid and accurate endotoxin detection. Expansion of biologics manufacturing and advanced therapy production is also driving demand. Research organizations are actively validating rFC assays for broader industrial applications. Furthermore, increasing emphasis on sustainable laboratory practices is expected to drive strong segment growth during the forecast period.
- By Application
On the basis of application, the Endotoxin Tests market is segmented into pharmaceutical manufacturing, medical device manufacturing, raw materials production, and packaging manufacture. The pharmaceutical manufacturing segment dominated the largest market revenue share of 57.6% in 2025, driven by stringent regulatory requirements for endotoxin testing during drug production and quality assurance processes. Pharmaceutical companies conduct extensive endotoxin testing to ensure the safety of injectable drugs, biologics, vaccines, and intravenous products. The segment benefits from increasing global pharmaceutical production and rising biologics development activities. Healthcare manufacturers are heavily investing in advanced contamination control and quality management systems. Growing prevalence of chronic diseases and expanding vaccine production programs are significantly supporting market demand. Regulatory agencies worldwide mandate rigorous endotoxin screening before product commercialization, further driving adoption. Technological advancements in rapid endotoxin testing methods are improving manufacturing efficiency and reducing batch release timelines. Increasing outsourcing of pharmaceutical manufacturing and quality testing services is also contributing to market expansion. Furthermore, rising focus on patient safety and compliance with GMP standards is expected to sustain segment dominance during the forecast period.
The medical device manufacturing segment is expected to witness the fastest CAGR of 12.1% from 2026 to 2033, driven by increasing production of implantable and sterile medical devices requiring stringent endotoxin control. Medical device manufacturers are increasingly adopting advanced endotoxin testing solutions to ensure compliance with international safety regulations. The segment benefits from rising demand for minimally invasive surgical devices and implantable technologies. Healthcare companies are investing heavily in contamination monitoring systems and automated quality assurance workflows. Expansion of global medical device manufacturing infrastructure is significantly supporting market growth. Technological advancements in rapid endotoxin detection platforms are improving operational efficiency and product safety. Regulatory agencies are strengthening guidelines for endotoxin testing in high-risk medical devices, further accelerating adoption. Increasing prevalence of hospital-acquired infections is also driving demand for safer and contamination-free devices. Research and development investments in next-generation medical technologies are contributing to segment expansion. Furthermore, increasing emphasis on sterile manufacturing practices and patient safety is expected to drive strong segment growth during the forecast period.
Endotoxin Tests Market Regional Analysis
- North America dominated the endotoxin tests market with the largest revenue share of 38.4% in 2025, driven by strong pharmaceutical manufacturing capabilities, stringent FDA regulations, increasing biologics production, and high adoption of advanced quality assurance technologies. The region benefits from the presence of leading pharmaceutical and biotechnology companies, along with rising demand for rapid microbial detection and contamination control solutions across drug manufacturing facilities and research laboratories
- The growing emphasis on product safety, sterility assurance, and compliance with regulatory standards has significantly accelerated the adoption of endotoxin testing technologies across pharmaceutical, medical device, and biotechnology sectors.
- This widespread adoption is further supported by increasing investments in laboratory automation, expanding biologics and vaccine manufacturing activities, and continuous advancements in endotoxin detection systems, establishing endotoxin testing as a critical component of pharmaceutical quality control and patient safety assurance
U.S. Endotoxin Tests Market Insight
The U.S. endotoxin tests market captured the largest revenue share in 2025 within North America, fueled by robust pharmaceutical and biopharmaceutical manufacturing activities, strong FDA compliance requirements, and increasing production of injectable drugs and biologics. The country continues to witness substantial adoption of advanced endotoxin testing technologies across pharmaceutical manufacturing facilities, contract research organizations, and quality control laboratories. Moreover, the increasing implementation of automated laboratory systems, rapid endotoxin detection platforms, and stringent sterility testing protocols is significantly contributing to the expansion of the Endotoxin Tests industry in the U.S.
Europe Endotoxin Tests Market Insight
The Europe endotoxin tests market is projected to expand at a substantial CAGR throughout the forecast period, primarily driven by stringent pharmaceutical safety regulations, growing biologics manufacturing activities, and increasing focus on contamination prevention in healthcare products. The region is witnessing rising adoption of endotoxin testing technologies across pharmaceutical production facilities, medical device manufacturing units, and research institutions. European countries are also emphasizing quality assurance and regulatory compliance, further supporting market growth across healthcare and life sciences sectors.
U.K. Endotoxin Tests Market Insight
The U.K. endotoxin tests market is anticipated to grow at a noteworthy CAGR during the forecast period, driven by increasing pharmaceutical R&D activities, rising biologics and vaccine production, and growing emphasis on healthcare product safety. In addition, strong regulatory oversight and the expansion of clinical research and laboratory testing infrastructure are encouraging the adoption of advanced endotoxin testing solutions across pharmaceutical and biotechnology industries. The country’s well-established healthcare and life sciences ecosystem is expected to continue stimulating market growth.
Germany Endotoxin Tests Market Insight
The Germany endotoxin tests market is expected to expand at a considerable CAGR during the forecast period, fueled by strong pharmaceutical manufacturing infrastructure, increasing adoption of laboratory automation technologies, and rising investments in biologics and biosimilar production. Germany’s emphasis on precision manufacturing, quality control, and regulatory compliance supports the widespread integration of endotoxin testing systems across pharmaceutical and medical device industries. The increasing focus on innovative and efficient microbial testing solutions is also contributing to market expansion.
Asia-Pacific Endotoxin Tests Market Insight
The Asia-Pacific endotoxin tests market is poised to grow at the fastest CAGR during the forecast period of 2026 to 2033, driven by expanding biopharmaceutical manufacturing, increasing vaccine production capacity, rising healthcare investments, and growing regulatory compliance requirements across countries such as China, India, South Korea, and Japan. The region’s rapidly developing pharmaceutical industry, coupled with increasing focus on product quality and safety standards, is accelerating the adoption of endotoxin testing technologies. Furthermore, the growing presence of contract manufacturing organizations and biotechnology companies is expanding market opportunities across APAC.
Japan Endotoxin Tests Market Insight
The Japan endotoxin tests market is gaining momentum due to the country’s advanced pharmaceutical manufacturing sector, high healthcare quality standards, and increasing focus on biologics and regenerative medicine development. The Japanese market places significant emphasis on precision diagnostics and contamination control, driving the adoption of rapid and automated endotoxin testing systems. Moreover, increasing investments in pharmaceutical innovation and laboratory modernization are further fueling market growth in Japan.
China Endotoxin Tests Market Insight
The China endotoxin tests market accounted for the largest market revenue share in Asia Pacific in 2025, attributed to the country’s rapidly expanding pharmaceutical and biotechnology industries, increasing vaccine manufacturing capacity, and rising implementation of stringent quality assurance protocols. China has emerged as a major manufacturing hub for biologics and pharmaceutical products, significantly boosting demand for endotoxin testing technologies across production and research facilities. The growing adoption of advanced laboratory automation systems, along with increasing government support for healthcare and biopharmaceutical development, continues to propel the market in China.
Endotoxin Tests Market Share
The Endotoxin Tests industry is primarily led by well-established companies, including:
- Charles River Laboratories (U.S.)
- Lonza Group AG (Switzerland)
- Thermo Fisher Scientific Inc. (U.S.)
- Merck KGaA (Germany)
- Associates of Cape Cod, Inc. (U.S.)
- GenScript Biotech Corporation (China)
- FUJIFILM Wako Pure Chemical Corporation (Japan)
- BioMérieux SA (France)
- WuXi AppTec (China)
- Seikagaku Corporation (Japan)
- Ellab A/S (Denmark)
- Pacific BioLabs (U.S.)
- Cambrex Corporation (U.S.)
- Nelson Laboratories, LLC (U.S.)
- Microcoat Biotechnologie GmbH (Germany)
- Baker Hughes Company (U.S.)
- Sanquin (Netherlands)
- Hyglos GmbH (Germany)
- Creative Bioarray (U.S.)
- ToxinSensor (China)
Latest Developments in Global Endotoxin Tests Market
- In March 2022, Bioendo Technology announced the launch of its new Micro Kinetic Chromogenic Endotoxin Test Kit, designed to provide higher sensitivity, broader linear detection range, and compatibility with vaccines, antibodies, and biologic products. The development strengthened rapid endotoxin detection capabilities for pharmaceutical quality control and biologics manufacturing
- In January 2024, Charles River Laboratories launched the Endosafe Trillium recombinant cascade reagent (rCR) cartridge, marking one of the company’s first major recombinant endotoxin testing platforms designed to reduce reliance on horseshoe crab-derived lysate reagents. The launch reflected the growing industry transition toward sustainable, animal-free endotoxin testing technologies
- In June 2024, FUJIFILM Wako Pure Chemical Corporation launched two new pyrogen and endotoxin testing products: the LumiMAT Pyrogen Detection Kit and PYROSTAR™ Neo+, a recombinant protein reagent for bacterial endotoxin detection. These launches expanded next-generation non-animal-derived testing solutions for pharmaceutical and medical device manufacturers globally
- In July 2024, the U.S. Pharmacopeia (USP) Microbiology Expert Committee approved USP Chapter <86> “Bacterial Endotoxins Test Using Recombinant Reagents,” formally permitting the use of non-animal-derived recombinant reagents for endotoxin testing. The decision represented a major regulatory milestone accelerating adoption of recombinant Factor C (rFC) and recombinant cascade reagent (rCR) technologies across the pharmaceutical industry
- In September 2024, Lonza Group began expansion of its endotoxin assay manufacturing facility in Walkersville, Maryland, adding approximately 18,000 square feet of production space to support rising global demand for endotoxin testing assays used in injectable drug and medical device safety testing
- In May 2025, Minerva Biolabs GmbH announced the launch of the NAT-MAT system, an advanced pyrogen detection platform combining Monocyte Activation Test (MAT) technology with digital PCR analysis. The system was designed to improve sensitivity and speed in detecting endotoxin and non-endotoxin pyrogens compared with conventional ELISA-based MAT assays
- In May 2025, Associates of Cape Cod (ACC) issued a formal industry position statement supporting USP Chapter <86> and recombinant bacterial endotoxin testing reagents, reaffirming PyroSmart NextGen as one of the first commercially available recombinant cascade reagent (rCR) platforms. The announcement reinforced increasing regulatory and commercial acceptance of sustainable endotoxin testing technologies
SKU-
Get online access to the report on the World's First Market Intelligence Cloud
- Interactive Data Analysis Dashboard
- Company Analysis Dashboard for high growth potential opportunities
- Research Analyst Access for customization & queries
- Competitor Analysis with Interactive dashboard
- Latest News, Updates & Trend analysis
- Harness the Power of Benchmark Analysis for Comprehensive Competitor Tracking
Research Methodology
Data collection and base year analysis are done using data collection modules with large sample sizes. The stage includes obtaining market information or related data through various sources and strategies. It includes examining and planning all the data acquired from the past in advance. It likewise envelops the examination of information inconsistencies seen across different information sources. The market data is analysed and estimated using market statistical and coherent models. Also, market share analysis and key trend analysis are the major success factors in the market report. To know more, please request an analyst call or drop down your inquiry.
The key research methodology used by DBMR research team is data triangulation which involves data mining, analysis of the impact of data variables on the market and primary (industry expert) validation. Data models include Vendor Positioning Grid, Market Time Line Analysis, Market Overview and Guide, Company Positioning Grid, Patent Analysis, Pricing Analysis, Company Market Share Analysis, Standards of Measurement, Global versus Regional and Vendor Share Analysis. To know more about the research methodology, drop in an inquiry to speak to our industry experts.
Customization Available
Data Bridge Market Research is a leader in advanced formative research. We take pride in servicing our existing and new customers with data and analysis that match and suits their goal. The report can be customized to include price trend analysis of target brands understanding the market for additional countries (ask for the list of countries), clinical trial results data, literature review, refurbished market and product base analysis. Market analysis of target competitors can be analyzed from technology-based analysis to market portfolio strategies. We can add as many competitors that you require data about in the format and data style you are looking for. Our team of analysts can also provide you data in crude raw excel files pivot tables (Fact book) or can assist you in creating presentations from the data sets available in the report.