Global Multiplex Molecular Diagnosis Technology Market
Market Size in USD Billion
CAGR :
% 
 USD
37.04 Billion
USD
76.00 Billion
2021
2029
USD
37.04 Billion
USD
76.00 Billion
2021
2029
| 2022 –2029 | |
| USD 37.04 Billion | |
| USD 76.00 Billion | |
|
|
|
|
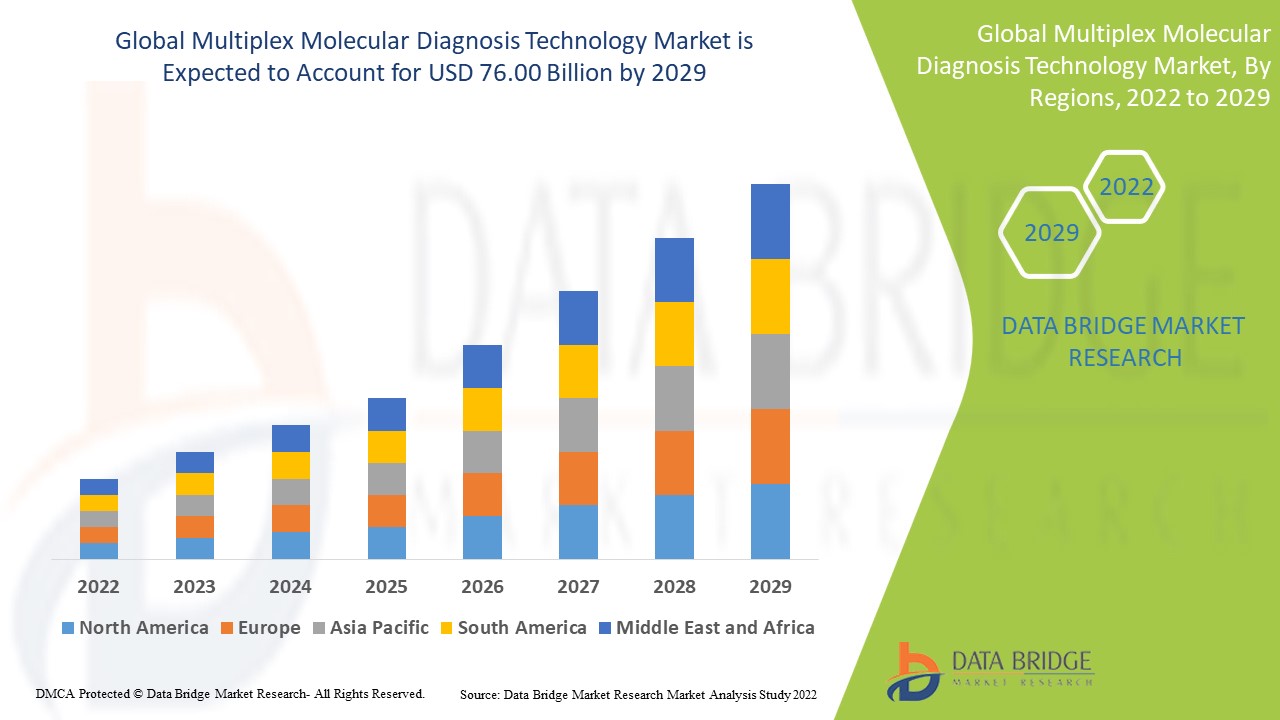
Multiplex Molecular Diagnosis Technology Market Analysis and Size
The market for multiplex molecular diagnosis technology is expected to grow during forecast period. Multiplex molecular diagnosis is used to find diseases such as H1N1 virus, chlamydia, and other infections. End users demand for multiplex molecular diagnosis is projected to rise as a result of the discovery of such diseases. For single-analytic testing, enzyme-linked immunosorbent assays (ELISA) are widely used to quantitatively assess the presence of protein in a sample.
Data Bridge Market Research analyses that the multiplex molecular diagnosis technology market which was USD 37.04 billion in 2021, would rocket up to USD 76.00 billion by 2029, and is expected to undergo a CAGR of 9.40% during the forecast period 2022 to 2029. In addition to the market insights such as market value, growth rate, market segments, geographical coverage, market players, and market scenario, the market report curated by the Data Bridge Market Research team also includes in-depth expert analysis, patient epidemiology, pipeline analysis, pricing analysis, and regulatory framework.
Multiplex Molecular Diagnosis Technology Market Scope and Segmentation
|
Report Metric |
Details |
|
Forecast Period |
2022 to 2029 |
|
Base Year |
2021 |
|
Historic Years |
2020 (Customizable to 2014 - 2019) |
|
Quantitative Units |
Revenue in USD Billion, Volumes in Units, Pricing in USD |
|
Segments Covered |
Product Type (Analyzers, Software, Instruments, Centrifuge, Counting Chambers, Incubators), Method (Real-time PCR, Nested PCR with Melt Curve Analysis, PCR with Low density Microarray, PCR with Liquid Phase Bead Array, PCR with Electrochemical Detection) |
|
Countries Covered |
U.S., Canada and Mexico in North America, Germany, France, U.K., Netherlands, Switzerland, Belgium, Russia, Italy, Spain, Turkey, Rest of Europe in Europe, China, Japan, India, South Korea, Singapore, Malaysia, Australia, Thailand, Indonesia, Philippines, Rest of Asia-Pacific (APAC) in the Asia-Pacific (APAC), Saudi Arabia, U.A.E, South Africa, Egypt, Israel, Rest of Middle East and Africa (MEA) as a part of Middle East and Africa (MEA), Brazil, Argentina and Rest of South America as part of South America. |
|
Market Players Covered |
Siemens (Germany), Hologic, Inc. (U.S.), Shenzhen Mindray Bio-Medical Electronics Co., Ltd. (China), Abbott (U.S.), BD (U.S.), F. Hoffmann-La Roche Ltd (Switzerland), Thermo Fisher Scientific Inc. (U.S.), Koninklijke Philips N.V. (Netherlands), NeuroLogica Corp. (U.S.), Shimadzu Medical (India) pvt. Ltd. (Japan), GENERAL ELECTRIC (U.S.), Quest Diagnostics Incorporated (U.S.), Sysmex India Pvt. Ltd. (Japan), Hitachi, Ltd. (Japan), Canon Inc. (Japan), FUJIFILM Holdings Corporation (U.K.) |
|
Market Opportunities |
|
Market Definition
Multiplexing allows for the identification of a wide range of conditions as opposed to carrying out separate individual procedures, when doing a molecular diagnostic test method. It makes it possible to detect multiple traits, diseases, or genetic sequences at once, including those associated with cancer genes.
Global Multiplex Molecular Diagnosis Technology Market Dynamics
Drivers
- Rise in rapid testing procedures
Globally, the novel coronavirus epidemic has caused tremendous loss to humanity. Rapid testing procedures are required to combat the COVID-19 virus's rising rate of transmission. As RT-PCR testing kits growing as the industry's gold standard, there is an enormous global demand for them. This element might encourage producers in the multiplex molecular diagnosis technology market to boost.
- Increase in demand of multiplex molecular diagnostics
Multiplex molecular diagnosis are expected to be in high demand during the forecast period. Increased economic stability results in better healthcare and research funding, which boosts demand for multiplex molecular diagnosis technology.
The market for multiplex molecular diagnosis is expanding as gastrointestinal, respiratory, and CNS illnesses become more common.
Opportunities
- Technological developments
The market for multiplex molecular diagnosis technology is gaining significant traction to novel technological breakthroughs. For instance, Bosch Healthcare Solutions developed one of the first multiplex molecular diagnostics tests in the world that can both detect COVID-19 and distinguish it from more than 8 respiratory illnesses. This product will remove pointless confusion and aid in patient differentiation.
Restraints/Challenges
On the other hand, the limited budgets for R&D and economic slowdown will obstruct the market's growth rate. The dearth of skilled professionals and lack of healthcare infrastructure in developing economies will challenge the for multiplex molecular diagnosis technology market.
This multiplex molecular diagnosis technology market report provides details of new recent developments, trade regulations, import-export analysis, production analysis, value chain optimization, market share, impact of domestic and localized market players, analyses opportunities in terms of emerging revenue pockets, changes in market regulations, strategic market growth analysis, market size, category market growths, application niches and dominance, product approvals, product launches, geographic expansions, technological innovations in the market. To gain more info on the multiplex molecular diagnosis technology market contact Data Bridge Market Research for an Analyst Brief, our team will help you take an informed market decision to achieve market growth.
COVID-19 Impact on Multiplex Molecular Diagnosis Technology Market
The impact of COVID-19 on multiplex molecular diagnosis technology is positive during the pandemic. US FDA has been approved many instruments in the diagnosis of COVID-19. The surge in demand for polymerase chain reaction tests, next-generation sequencing (NGS), serology-based rapid-test products, and a dramatic increase in the target patient population are all predicted to contribute to an increase in the demand for diagnostics as a result of COVID-19 in Brazil. For instance, Seegene Inc., a pioneer in multiplex PCR technology development, provided Brazil with five million Allplex 2019-nCOV assay tests in May 2020 through its subsidiary Seegene do Brasil Diagnosticos Ltda.
Recent Development
- In July 2020, For the purpose of assisting with SARS CoV-2 diagnostic testing, Roche Diagnostics India launched the Cobas 8800 instrument at the National Institute of Cholera and Enteric Diseases in Kolkata and the Cobas 6800 at the National Institute for Research in Reproductive Health in Mumbai. The Cobas 6800/8800 systems from Roche offer faster turnaround times, increased flexibility, and test results in three and a half hours.
- In January 2020, The Japanese Pharmaceuticals and Medical Devices Agency (PMDA) granted Illumina product approval for the MiSeqDx equipment. The MiSeqDx can now be sold in Japan as a Class I medical equipment, which will have a favourable effect on the market.
Global Multiplex Molecular Diagnosis Technology Market Scope
The multiplex molecular diagnosis technology market is segmented on the basis of product type and method. The growth amongst these segments will help you analyze meagre growth segments in the industries and provide the users with a valuable market overview and market insights to help them make strategic decisions for identifying core market applications.
Product Type
- Analyzers
- Software
- Instruments
- Centrifuge
- Counting Chambers
- Incubators
Method
- Real-time PCR
- Nested PCR with Melt Curve Analysis
- PCR with Low Density Microarray
- PCR with Liquid Phase Bead Array
- PCR with Electrochemical Detection
Multiplex Molecular Diagnosis Technology Market Regional Analysis/Insights
The multiplex molecular diagnosis technology market is analysed and market size insights and trends are provided by country, product type and method as referenced above.
The countries covered in the multiplex molecular diagnosis technology market report are U.S., Canada and Mexico in North America, Germany, France, U.K., Netherlands, Switzerland, Belgium, Russia, Italy, Spain, Turkey, Rest of Europe in Europe, China, Japan, India, South Korea, Singapore, Malaysia, Australia, Thailand, Indonesia, Philippines, Rest of Asia-Pacific (APAC) in the Asia-Pacific (APAC), Saudi Arabia, U.A.E, South Africa, Egypt, Israel, Rest of Middle East and Africa (MEA) as a part of Middle East and Africa (MEA), Brazil, Argentina and Rest of South America as part of South America.
North America dominates the multiplex molecular diagnosis technology market due to the well- equipped infrastructure, patient awareness, advancement of techniques among others.
Asia-Pacific is expected to grow at the highest growth rate in the forecast period of 2022 to 2029 due to the rising demand for improved multiplex molecular diagnosis technology.
The country section of the report also provides individual market impacting factors and changes in regulation in the market domestically that impacts the current and future trends of the market. Data points like down-stream and upstream value chain analysis, technical trends and porter's five forces analysis, case studies are some of the pointers used to forecast the market scenario for individual countries. Also, the presence and availability of global brands and their challenges faced due to large or scarce competition from local and domestic brands, impact of domestic tariffs and trade routes are considered while providing forecast analysis of the country data.
Competitive Landscape and Multiplex Molecular Diagnosis Technology Market Share Analysis
The multiplex molecular diagnosis technology market competitive landscape provides details by competitor. Details included are company overview, company financials, revenue generated, market potential, investment in research and development, new market initiatives, global presence, production sites and facilities, production capacities, company strengths and weaknesses, product launch, product width and breadth, application dominance. The above data points provided are only related to the companies' focus related to multiplex molecular diagnosis technology market.
Some of the major players operating in the multiplex molecular diagnosis technology market are:
- Siemens (Germany)
- Hologic, Inc. (U.S.)
- Shenzhen Mindray Bio-Medical Electronics Co., Ltd. (China)
- Abbott (U.S.)
- BD (U.S.)
- F. Hoffmann-La Roche Ltd (Switzerland)
- Thermo Fisher Scientific Inc. (U.S.)
- Koninklijke Philips N.V. (Netherlands)
- NeuroLogica Corp. (U.S.)
- Shimadzu Medical (India) pvt. Ltd. (Japan)
- GENERAL ELECTRIC (U.S.)
- Quest Diagnostics Incorporated (U.S.)
- Sysmex India Pvt. Ltd. (Japan)
- Hitachi, Ltd. (Japan)
- Canon Inc. (Japan)
- FUJIFILM Holdings Corporation (U.K)
SKU-
Get online access to the report on the World's First Market Intelligence Cloud
- Interactive Data Analysis Dashboard
- Company Analysis Dashboard for high growth potential opportunities
- Research Analyst Access for customization & queries
- Competitor Analysis with Interactive dashboard
- Latest News, Updates & Trend analysis
- Harness the Power of Benchmark Analysis for Comprehensive Competitor Tracking
Table of Content
1 INTRODUCTION
1.1 OBJECTIVES OF THE STUDY
1.2 MARKET DEFINITION
1.3 OVERVIEW OF GLOBAL MULTIPLEX MOLECULAR DIAGNOSIS TECHNOLOGY MARKET
1.4 CURRENCY AND PRICING
1.5 LIMITATION
1.6 MARKETS COVERED
2 MARKET SEGMENTATION
2.1 KEY TAKEAWAYS
2.2 ARRIVING AT THE GLOBAL MULTIPLEX MOLECULAR DIAGNOSIS TECHNOLOGY MARKET SIZE
2.2.1 VENDOR POSITIONING GRID
2.2.2 TECHNOLOGY LIFE LINE CURVE
2.2.3 TRIPOD DATA VALIDATION MODEL
2.2.4 MARKET GUIDE
2.2.5 MULTIVARIATE MODELLING
2.2.6 TOP TO BOTTOM ANALYSIS
2.2.7 CHALLENGE MATRIX
2.2.8 APPLICATION COVERAGE GRID
2.2.9 STANDARDS OF MEASUREMENT
2.2.10 VENDOR SHARE ANALYSIS
2.2.11 DATA POINTS FROM KEY PRIMARY INTERVIEWS
2.2.12 DATA POINTS FROM KEY SECONDARY DATABASES
2.3 GLOBAL MULTIPLEX MOLECULAR DIAGNOSIS TECHNOLOGY MARKET : RESEARCH SNAPSHOT
2.4 ASSUMPTIONS
3 MARKET OVERVIEW
3.1 DRIVERS
3.2 RESTRAINTS
3.3 OPPORTUNITIES
3.4 CHALLENGES
4 EXECUTIVE SUMMARY
5 PREMIUM INSIGHTS
5.1 PESTEL ANALYSIS
5.2 PORTER’S FIVE FORCES MODEL
6 INDUSTRY INSIGHTS
6.1 MICRO AND MACRO ECONOMIC FACTORS
6.2 PENETRATION AND GROWTH PROSPECT MAPPING
6.3 KEY PRICING STRATEGIES
6.4 INTERVIEWS WITH SPECIALIST
6.5 ANALYIS AND RECOMMENDATION
7 INTELLECTUAL PROPERTY (IP) PORTFOLIO
7.1 PATENT QUALITY AND STRENGTH
7.2 PATENT FAMILIES
7.3 LICENSING AND COLLABORATIONS
7.4 COMPETITIVE LANDSCAPE
7.5 IP STRATEGY AND MANAGEMENT
7.6 OTHER
8 COST ANALYSIS BREAKDOWN
9 TECHNONLOGY ROADMAP
10 INNOVATION TRACKER AND STRATEGIC ANALYSIS
10.1 MAJOR DEALS AND STRATEGIC ALLIANCES ANALYSIS
10.1.1 JOINT VENTURES
10.1.2 MERGERS AND ACQUISITIONS
10.1.3 LICENSING AND PARTNERSHIP
10.1.4 TECHNOLOGY COLLABORATIONS
10.1.5 STRATEGIC DIVESTMENTS
10.2 NUMBER OF PRODUCTS IN DEVELOPMENT
10.3 STAGE OF DEVELOPMENT
10.4 TIMELINES AND MILESTONES
10.5 INNOVATION STRATEGIES AND METHODOLOGIES
10.6 RISK ASSESSMENT AND MITIGATION
10.7 FUTURE OUTLOOK
11 REGULATORY COMPLIANCE
11.1 REGULATORY AUTHORITIES
11.2 REGULATORY CLASSIFICATIONS
11.2.1 CLASS I
11.2.2 CLASS II
11.2.3 CLASS III
11.3 REGULATORY SUBMISSIONS
11.4 INTERNATIONAL HARMONIZATION
11.5 COMPLIANCE AND QUALITY MANAGEMENT SYSTEMS
11.6 REGULATORY CHALLENGES AND STRATEGIES
12 REIMBURSEMENT FRAMEWORK
13 OPPUTUNITY MAP ANALYSIS
14 VALUE CHAIN ANALYSIS
15 HEALTHCARE ECONOMY
15.1 HEALTHCARE EXPENDITURE
15.2 CAPITAL EXPENDITURE
15.3 CAPEX TRENDS
15.4 CAPEX ALLOCATION
15.5 FUNDING SOURCES
15.6 INDUSTRY BENCHMARKS
15.7 GDP RATION IN OVERALL GDP
15.8 HEALTHCARE SYSTEM STRUCTURE
15.9 GOVERNMENT POLICIES
15.1 ECONOMIC DEVELOPMENT
16 GLOBAL MULTIPLEX MOLECULAR DIAGNOSIS TECHNOLOGY MARKET, BY PRODUCT TYPE
16.1 OVERVIEW
16.2 INSTRUMENT/SYSTEMS
16.2.1 ANALYZERS
16.2.1.1. BENCHTOP
16.2.1.2. PORTABLE
16.2.2 CENTRIFUGE
16.2.2.1. BY TYPE
16.2.2.1.1. FIXED ANGLE
16.2.2.1.2. SWINGING BUCKET
16.2.2.1.3. VERTICAL ROTORS
16.2.2.2. BY MODALITY
16.2.2.2.1. BENCHTOP
16.2.2.2.2. PORTABLE
16.2.2.3. BY SPEED OF ROTOR
16.2.2.3.1. HIGH SPEED
16.2.2.3.2. LOW SPEED
16.2.2.4. OTHERS
16.2.3 COUNTING CHAMBERS
16.2.3.1. V-SLASH
16.2.3.2. SPECIAL DEPTH
16.2.3.3. 3 GRIDS
16.2.3.4. OTHERS
16.2.4 MULTIPLEX PCR SYSTEMS
16.2.5 FLOW CYTOMETRY-BASED INSTRUMENTS
16.2.6 INCUBATORS
16.2.7 OTHERS
16.3 CONSUMABLES
16.3.1 REAGENTS, ASSAYS & KITS
16.3.1.1. PCR REAGENTS AND QPCR REAGENTS
16.3.1.2. NGS REAGENTS AND SEQUENCING KITS
16.3.1.3. MICROARRAY KITS AND REAGENTS
16.3.1.4. MULTIPLEX REAGENT AND KITS
16.3.1.5. CELL LYSIS RT-QPCR KITS
16.3.1.6. CDNA SYNTHESIS KITS
16.3.1.7. RT-QPCR REAGENTS
16.3.1.8. ELISA KITS, CONTROLS
16.3.1.9. BEADS
16.3.1.9.1. MAGNETIC
16.3.1.9.2. NON-MAGNETIC
16.3.1.10. ASSAYS
16.3.1.10.1. PREMIXED PANELS
16.3.1.10.2. CUSTOM MIXES
16.3.1.10.3. SINGLEPLEX SETS
16.3.2 HANDHELD MAGNETIC WASHER
16.3.3 AIR INTAKE FILTER
16.3.4 SHEATH FLUID BOTTLE
16.3.5 SHEATH FLUID
16.3.6 COMMUNICATION CABLE
16.3.7 PCR PLATES
16.3.8 PCR TUBES AND CAPS
16.3.9 PCR PLATE SEALS
16.3.10 PCR PLASTICS ACCESSORIES
16.3.11 OTHERS
16.4 SOFTWARE
17 GLOBAL MULTIPLEX MOLECULAR DIAGNOSIS TECHNOLOGY MARKET, BY TECHNOLOGY
17.1 OVERVIEW
17.2 PCR
17.2.1 REAL-TIME PCR
17.2.2 NESTED PCR WITH MELT CURVE ANALYSIS
17.2.3 PCR WITH LOW DENSITY MICROARRAY
17.2.4 PCR WITH LIQUID PHASE BEAD ARRAY
17.2.5 PCR WITH ELECTROCHEMICAL DETECTION
17.2.6 OTHERS
17.3 NEXT GENERATION SEQUENCING
17.3.1 WHOLE GENOME SEQUENCING
17.3.2 WHOLE EXOME SEQUENCING
17.3.3 TARGETED SEQUENCING
17.3.4 RNA SEQUENCING (RNA-SEQ)
17.3.5 OTHERS
17.3.6 OTHERS
18 GLOBAL MULTIPLEX MOLECULAR DIAGNOSIS TECHNOLOGY MARKET, BY SITE
18.1 OVERVIEW
18.2 LABORATORY BASED
18.3 POINT OF CARE
18.4 SELF-TEST/OTC
19 GLOBAL MULTIPLEX MOLECULAR DIAGNOSIS TECHNOLOGY MARKET, BY ANALYTES
19.1 OVERVIEW
19.2 NUCLEIC ACID ASSAYS
19.3 IMMUNOASSAYS
19.4 ENZYME ASSAYS
19.5 OTHERS
20 GLOBAL MULTIPLEX MOLECULAR DIAGNOSIS TECHNOLOGY MARKET, BY APPLICATION
20.1 OVERVIEW
20.2 CLINICAL
20.2.1 CNS INFECTION
20.2.2 RESPIRATORY DISEASE
20.2.3 ACUTE RESPIRATORY TRACT INFECTIONS
20.2.4 AUTOIMMUNE INFLAMMATORY DISEASES
20.2.5 ACUTE VIRAL GASTROENTERITIS
20.2.6 OTHERS
20.3 RESEARCH
20.3.1 DRUG DISCOVERY AND DEVELOPMENT
20.3.2 GENETIC ANALYSIS
20.3.3 BIODEFENSE
20.3.4 FOOD SAFETY
20.3.5 BIOMEDICAL RESEARCH
20.3.6 OTHERS
21 GLOBAL MULTIPLEX MOLECULAR DIAGNOSIS TECHNOLOGY MARKET, BY END USER
21.1 OVERVIEW
21.2 HOSPITALS
21.2.1 BY TYPE
21.2.1.1. PUBLIC
21.2.1.2. PRIVATE
21.2.2 BY CARE
21.2.2.1. ACUTE CARE HOSPITALS
21.2.2.2. LONG-TERM CARE HOSPITALS
21.3 CLINICAL LABORATORIES
21.4 PHARMACEUTICAL & BIOTECHNOLOGY COMPANIES
21.5 ACADEMIC AND RESEARCH INSTITUTES
21.6 FORENSIC LABORATORIES
21.7 CONTRACT ORGANISATIONS
21.8 ENVIRONMENTAL LABORATORIES
21.9 OTHERS
22 GLOBAL MULTIPLEX MOLECULAR DIAGNOSIS TECHNOLOGY MARKET, BY DISTRIBUTION CHANNEL
22.1 OVERVIEW
22.2 DIRECT TENDERS
22.3 RETAIL SALES
22.3.1 OFFLINE
22.3.2 ONLINE
22.4 OTHERS
23 GLOBAL MULTIPLEX MOLECULAR DIAGNOSIS TECHNOLOGY MARKET, BY COUNTRY
GLOBAL MULTIPLEX MOLECULAR DIAGNOSIS TECHNOLOGY MARKET (ALL SEGMENTATION PROVIDED ABOVE IS REPRESENTED IN THIS CHAPTER BY COUNTRY)
23.1.1 NORTH AMERICA
23.1.1.1. U.S.
23.1.1.2. CANADA
23.1.1.3. MEXICO
23.1.2 EUROPE
23.1.2.1. GERMANY
23.1.2.2. FRANCE
23.1.2.3. U.K.
23.1.2.4. ITALY
23.1.2.5. SPAIN
23.1.2.6. RUSSIA
23.1.2.7. TURKEY
23.1.2.8. HUNGARY
23.1.2.9. BELGIUM
23.1.2.10. NETHERLANDS
23.1.2.11. SWITZERLAND
23.1.2.12. REST OF EUROPE
23.1.3 ASIA-PACIFIC
23.1.3.1. JAPAN
23.1.3.2. CHINA
23.1.3.3. SOUTH KOREA
23.1.3.4. INDIA
23.1.3.5. AUSTRALIA
23.1.3.6. SINGAPORE
23.1.3.7. THAILAND
23.1.3.8. MALAYSIA
23.1.3.9. INDONESIA
23.1.3.10. PHILIPPINES
23.1.3.11. VIETNAM
23.1.3.12. REST OF ASIA-PACIFIC
23.1.4 SOUTH AMERICA
23.1.4.1. BRAZIL
23.1.4.2. ARGENTINA
23.1.4.3. PERU
23.1.4.4. REST OF SOUTH AMERICA
23.1.5 MIDDLE EAST AND AFRICA
23.1.5.1. SOUTH AFRICA
23.1.5.2. SAUDI ARABIA
23.1.5.3. UAE
23.1.5.4. EGYPT
23.1.5.5. KUWAIT
23.1.5.6. ISRAEL
23.1.5.7. REST OF MIDDLE EAST AND AFRICA
23.1.6 KEY PRIMARY INSIGHTS: BY MAJOR COUNTRIES
24 GLOBAL MULTIPLEX MOLECULAR DIAGNOSIS TECHNOLOGY MARKET, COMPANY LANDSCAPE
24.1 COMPANY SHARE ANALYSIS: GLOBAL
24.2 COMPANY SHARE ANALYSIS: NORTH AMERICA
24.3 COMPANY SHARE ANALYSIS: EUROPE
24.4 COMPANY SHARE ANALYSIS: ASIA-PACIFIC
24.5 MERGERS & ACQUISITIONS
24.6 NEW PRODUCT DEVELOPMENT & APPROVALS
24.7 EXPANSIONS
24.8 REGULATORY CHANGES
24.9 PARTNERSHIP AND OTHER STRATEGIC DEVELOPMENTS
25 GLOBAL MULTIPLEX MOLECULAR DIAGNOSIS TECHNOLOGY MARKET, SWOT AND DBR ANALYSIS
26 GLOBAL MULTIPLEX MOLECULAR DIAGNOSIS TECHNOLOGY MARKET, COMPANY PROFILE
26.1 BD
26.1.1 COMPANY OVERVIEW
26.1.2 REVENUE ANALYSIS
26.1.3 GEOGRAPHIC PRESENCE
26.1.4 PRODUCT PORTFOLIO
26.1.5 RECENT DEVELOPMENTS
26.2 BIOMÉRIEUX SA
26.2.1 COMPANY OVERVIEW
26.2.2 REVENUE ANALYSIS
26.2.3 GEOGRAPHIC PRESENCE
26.2.4 PRODUCT PORTFOLIO
26.2.5 RECENT DEVELOPMENTS
26.3 BIO-RAD LABORATORIES, INC.
26.3.1 COMPANY OVERVIEW
26.3.2 REVENUE ANALYSIS
26.3.3 GEOGRAPHIC PRESENCE
26.3.4 PRODUCT PORTFOLIO
26.3.5 RECENT DEVELOPMENTS
26.4 THERMO FISCHER SCIENTIFIC INC.
26.4.1 COMPANY OVERVIEW
26.4.2 REVENUE ANALYSIS
26.4.3 GEOGRAPHIC PRESENCE
26.4.4 PRODUCT PORTFOLIO
26.4.5 RECENT DEVELOPMENTS
26.5 MERCK KGAA
26.5.1 COMPANY OVERVIEW
26.5.2 REVENUE ANALYSIS
26.5.3 GEOGRAPHIC PRESENCE
26.5.4 PRODUCT PORTFOLIO
26.5.5 RECENT DEVELOPMENTS
26.6 ILLUMINA, INC.
26.6.1 COMPANY OVERVIEW
26.6.2 REVENUE ANALYSIS
26.6.3 GEOGRAPHIC PRESENCE
26.6.4 PRODUCT PORTFOLIO
26.6.5 RECENT DEVELOPMENTS
26.7 QIAGEN
26.7.1 COMPANY OVERVIEW
26.7.2 REVENUE ANALYSIS
26.7.3 GEOGRAPHIC PRESENCE
26.7.4 PRODUCT PORTFOLIO
26.7.5 RECENT DEVELOPMENTS
26.8 ABCAM PLC.
26.8.1 COMPANY OVERVIEW
26.8.2 REVENUE ANALYSIS
26.8.3 GEOGRAPHIC PRESENCE
26.8.4 PRODUCT PORTFOLIO
26.8.5 RECENT DEVELOPMENTS
26.9 AGILENT TECHNOLOGIES, INC.
26.9.1 COMPANY OVERVIEW
26.9.2 REVENUE ANALYSIS
26.9.3 GEOGRAPHIC PRESENCE
26.9.4 PRODUCT PORTFOLIO
26.9.5 RECENT DEVELOPMENTS
26.1 MESO SCALE DIAGNOSTICS, LLC.
26.10.1 COMPANY OVERVIEW
26.10.2 REVENUE ANALYSIS
26.10.3 GEOGRAPHIC PRESENCE
26.10.4 PRODUCT PORTFOLIO
26.10.5 RECENT DEVELOPMENTS
26.11 RANDOX LABORATORIES LTD.
26.11.1 COMPANY OVERVIEW
26.11.2 REVENUE ANALYSIS
26.11.3 GEOGRAPHIC PRESENCE
26.11.4 PRODUCT PORTFOLIO
26.11.5 RECENT DEVELOPMENTS
26.12 QUANTERIX
26.12.1 COMPANY OVERVIEW
26.12.2 REVENUE ANALYSIS
26.12.3 GEOGRAPHIC PRESENCE
26.12.4 PRODUCT PORTFOLIO
26.12.5 RECENT DEVELOPMENTS
26.13 BIO-TECHNE
26.13.1 COMPANY OVERVIEW
26.13.2 REVENUE ANALYSIS
26.13.3 GEOGRAPHIC PRESENCE
26.13.4 PRODUCT PORTFOLIO
26.13.5 RECENT DEVELOPMENTS
26.14 OLINK
26.14.1 COMPANY OVERVIEW
26.14.2 REVENUE ANALYSIS
26.14.3 GEOGRAPHIC PRESENCE
26.14.4 PRODUCT PORTFOLIO
26.14.5 RECENT DEVELOPMENTS
26.15 SEEGENE INC. + HOLOGIC INC.
26.15.1 COMPANY OVERVIEW
26.15.2 REVENUE ANALYSIS
26.15.3 GEOGRAPHIC PRESENCE
26.15.4 PRODUCT PORTFOLIO
26.15.5 RECENT DEVELOPMENTS
26.16 F. HOFFMANN-LA ROCHE LTD
26.16.1 COMPANY OVERVIEW
26.16.2 REVENUE ANALYSIS
26.16.3 GEOGRAPHIC PRESENCE
26.16.4 PRODUCT PORTFOLIO
26.16.5 RECENT DEVELOPMENTS
26.17 MERIDIAN BIOSCIENCE
26.17.1 COMPANY OVERVIEW
26.17.2 REVENUE ANALYSIS
26.17.3 GEOGRAPHIC PRESENCE
26.17.4 PRODUCT PORTFOLIO
26.17.5 RECENT DEVELOPMENTS
26.18 BIOGX
26.18.1 COMPANY OVERVIEW
26.18.2 REVENUE ANALYSIS
26.18.3 GEOGRAPHIC PRESENCE
26.18.4 PRODUCT PORTFOLIO
26.18.5 RECENT DEVELOPMENTS
26.19 PROMEGA CORPORATION.
26.19.1 COMPANY OVERVIEW
26.19.2 REVENUE ANALYSIS
26.19.3 GEOGRAPHIC PRESENCE
26.19.4 PRODUCT PORTFOLIO
26.19.5 RECENT DEVELOPMENTS
26.2 DIASORIN S.P.A
26.20.1 COMPANY OVERVIEW
26.20.2 REVENUE ANALYSIS
26.20.3 GEOGRAPHIC PRESENCE
26.20.4 PRODUCT PORTFOLIO
26.20.5 RECENT DEVELOPMENTS
26.21 SIEMENS HEALTHCARE DIAGNOSTICS INC.
26.21.1 COMPANY OVERVIEW
26.21.2 REVENUE ANALYSIS
26.21.3 GEOGRAPHIC PRESENCE
26.21.4 PRODUCT PORTFOLIO
26.21.5 RECENT DEVELOPMENTS
26.22 SOLIS BIODYNE
26.22.1 COMPANY OVERVIEW
26.22.2 REVENUE ANALYSIS
26.22.3 GEOGRAPHIC PRESENCE
26.22.4 PRODUCT PORTFOLIO
26.22.5 RECENT DEVELOPMENTS
26.23 JANT PHARMACAL CORPORATION
26.23.1 COMPANY OVERVIEW
26.23.2 REVENUE ANALYSIS
26.23.3 GEOGRAPHIC PRESENCE
26.23.4 PRODUCT PORTFOLIO
26.23.5 RECENT DEVELOPMENTS
26.24 SHIMADZU SCIENTIFIC INSTRUMENTS
26.24.1 COMPANY OVERVIEW
26.24.2 REVENUE ANALYSIS
26.24.3 GEOGRAPHIC PRESENCE
26.24.4 PRODUCT PORTFOLIO
26.24.5 RECENT DEVELOPMENTS
26.25 ADALTIS S.R.L.
26.25.1 COMPANY OVERVIEW
26.25.2 REVENUE ANALYSIS
26.25.3 GEOGRAPHIC PRESENCE
26.25.4 PRODUCT PORTFOLIO
26.25.5 RECENT DEVELOPMENTS
26.26 MULTIPLEXDX INTERNATIONAL
26.26.1 COMPANY OVERVIEW
26.26.2 REVENUE ANALYSIS
26.26.3 GEOGRAPHIC PRESENCE
26.26.4 PRODUCT PORTFOLIO
26.26.5 RECENT DEVELOPMENTS
26.27 BRUKER
26.27.1 COMPANY OVERVIEW
26.27.2 REVENUE ANALYSIS
26.27.3 GEOGRAPHIC PRESENCE
26.27.4 PRODUCT PORTFOLIO
26.27.5 RECENT DEVELOPMENTS
NOTE: THE COMPANIES PROFILED IS NOT EXHAUSTIVE LIST AND IS AS PER OUR PREVIOUS CLIENT REQUIREMENT. WE PROFILE MORE THAN 100 COMPANIES IN OUR STUDY AND HENCE THE LIST OF COMPANIES CAN BE MODIFIED OR REPLACED ON REQUEST RELATED REPORTS
27 CONCLUSION
28 QUESTIONNAIRE
29 ABOUT DATA BRIDGE MARKET RESEARCH

Research Methodology
Data collection and base year analysis are done using data collection modules with large sample sizes. The stage includes obtaining market information or related data through various sources and strategies. It includes examining and planning all the data acquired from the past in advance. It likewise envelops the examination of information inconsistencies seen across different information sources. The market data is analysed and estimated using market statistical and coherent models. Also, market share analysis and key trend analysis are the major success factors in the market report. To know more, please request an analyst call or drop down your inquiry.
The key research methodology used by DBMR research team is data triangulation which involves data mining, analysis of the impact of data variables on the market and primary (industry expert) validation. Data models include Vendor Positioning Grid, Market Time Line Analysis, Market Overview and Guide, Company Positioning Grid, Patent Analysis, Pricing Analysis, Company Market Share Analysis, Standards of Measurement, Global versus Regional and Vendor Share Analysis. To know more about the research methodology, drop in an inquiry to speak to our industry experts.
Customization Available
Data Bridge Market Research is a leader in advanced formative research. We take pride in servicing our existing and new customers with data and analysis that match and suits their goal. The report can be customized to include price trend analysis of target brands understanding the market for additional countries (ask for the list of countries), clinical trial results data, literature review, refurbished market and product base analysis. Market analysis of target competitors can be analyzed from technology-based analysis to market portfolio strategies. We can add as many competitors that you require data about in the format and data style you are looking for. Our team of analysts can also provide you data in crude raw excel files pivot tables (Fact book) or can assist you in creating presentations from the data sets available in the report.














