The rapid spread of COVID-19 around the world with over 18.2 million cases and over 6,92,000 deaths which is creating new challenges and forcing healthcare professionals to develop new and innovative ways to explore new things to safeguard millions of lives. The Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) virus has spread around the world in all regions starting from Asia-Pacific, North America, South America, Europe up to the Middle East and Africa. Coronavirus disease (COVID-19) pandemic outbreak has influenced, inflicted & affected millions of human lives across all regions ranging from Asia-Pacific, Europe, North America, Middle East, and Africa and South America.
World Health Organization (WHO) has been declared the Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) virus spread as a public health emergency of international concern (PHEIC) as to the virus is spreading rapidly and is contagious kind of nature that has life treated millions live and resulted over 6,92,000 deaths from the virus around the world.
Coronavirus (COVID 19) virus pandemic outbreak has inflicted & impacted all the countries either in a smaller or larger way and compiled governments and authorities to impose unprecedented actions in the order to curb the Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) virus spread. The governments in various counties have imposed isolation & lockdowns, close borders, businesses in the order to safeguard lives and wide-spread closures in the limit and slow down the wide-spread of the Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2). Countries are facing crises at multiple layers which are composed of international and domestic economic disruptions in demand and supply, the collapse of prices, and straining health systems.
The Coronavirus disease (COVID-19) pandemic outbreak by Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) virus spread has left unprecedented strains on the healthcare and clinical laboratories and the virus has completely changed clinical chemistry market dynamics worldwide.
Clinical chemistry is the area of chemistry that helps with bodily fluids analysis and measurement of amounts of biologically important substances in the bodily fluids for therapeutic and diagnostic purposes. Clinical chemistry is also known as medical biochemistry; chemical pathology is applied biochemistry. Clinical chemistry uses various methods to measure substances that are carefully designed for providing accurate assessments of the concentrations.
Coronavirus disease (COVID-19) outbreak has majorly impacted clinical laboratories in the past few months. Clinical laboratory diagnosis of COVID-19 is required to confirm the presence of the virus in the human. Clinical laboratories are using molecular testing and antibody testing recognizes the genetic material of the Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) virus (RNA), from samples such as throat/nasal or sputum swabs. While respiratory secretions are having a maximum chance of containing a virus; it might be at an insufficient level for detection.
Clinical laboratories are rapidly developing a new test for the specific identification of COVID-19; SARS-CoV-2 is a truly new virus. Clinical laboratories are using the most common type of molecular test for SARS-CoV-2 identifications uses a unique technique called RT-PCR (reverse transcription-polymerase chain reaction).
Various clinical laboratories and companies are now offering tests for SARS-CoV-2 identifications. Most of the diagnostic tests are required to be analyzed in a Clinical lab, though a few diagnostic tests can be conducted near the patient ("point of care" testing) though trained clinicians. In certain cases, diagnosis tests outcomes require hours or day for the sample to be transported, the RNA isolated and amplified, and a diagnosis test outcomes obtained. Point-of-care (POCT) diagnose testing might be more quick and rapid, however, typically can only run one test to some patients samples at a time.
Unfortunately, the supply chain has created some restrictions and limited the ability to perform a molecular test when needed. Challenges have included both test reagent and consumables shortages as a various reagent and consumables manufacturers are struggling to keep up with demand due to affected supply chain Supply of raw materials has been critically disrupted due to the quarantine and lack of raw materials, labor shortage, and international borders. The warehouses do not remain as same as previously pre-Coronavirus Disease (COVID-19) pandemic outbreak, raw materials the order between companies cannot be performed; shortage of raw material and components affected the product supply chain. Further, isolation & lockdowns, close borders are also limiting the manufacturer’s transportation capacity in other supplies such as swabs and media transport to the lab. The coronavirus disease (COVID-19) pandemic outbreak has forced manufacturers to find new contract manufacturers and suppliers to move manufacturing activities to different or new facilities locations that are less impacted by the outbreak. In current situations manufactures are facing difficulties with the existing component supplier or contract manufacturer which are at risk or closed or not being able to fulfill supply requirements due to challenges such as extended periods of shut down or workforce.
Also, clinical laboratories are facing a shortage of lab professionals due to some of the lab professionals are quitting due to an increased risk of contracting the infection. While on the other end clinical laboratories have to deal with the various challenges during the Coronavirus disease (COVID-19) outbreak such as the availability of PPE kits.
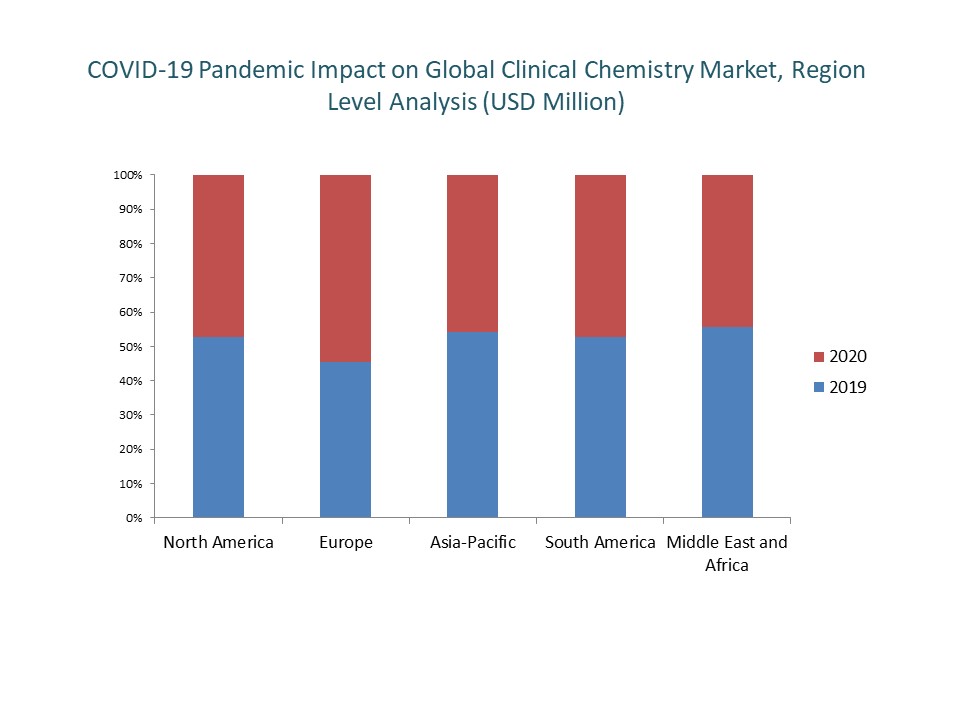 Various reports published by the market player and regulatory authorities have depicted that the clinical laboratory industry is facing very high demand around the world and data collected from various countries depicts that only a few products are categorized as non-essential products in the clinical laboratory.
Various reports published by the market player and regulatory authorities have depicted that the clinical laboratory industry is facing very high demand around the world and data collected from various countries depicts that only a few products are categorized as non-essential products in the clinical laboratory.
Several governments, self-financing organizations, regulatory authorities from all over the world and including World Health Organisation, European Medicines Agency, Food and Drug Administration, have published new regulations and adopted new standards for to full fill the demand of clinical chemistry products during the Coronavirus disease (COVID-19) pandemic outbreak.
Authorities and governments all over the world have stressed the policy measures and adopted various policy measures to curb the disease transmission. The disease transmission rate has increased the demand for clinical chemistry analysers, POC test kits, clinical chemistry analyser kits & reagents in hospitals, pathology laboratories, maternity centres, outpatient centres, and clinics. Governments have asked clinicians’ and manufacturers to facilitate patients with the emergency clinical chemistry market procedure to save life.
Coronavirus disease (COVID-19) pandemic outbreak is pushing clinical chemistry market stakeholders, manufactures, and researchers to push boundaries to find new ways of manufacturing new devices, equipment and accessories with new innovative & unique design, new technologies, and new functionality.
DBMR Insights will provide case study based analysis of changes in market dynamics in terms of market share, new entrance, new developments, impact on domestic and localised market players, country-level analysis, emerging market, PESTLE analysis, revenue pockets analysis, changes in market regulations, strategic decisions, product approvals, product launches, geographic expansions in the markets.
 To understand the analysis and the market scenario contact us for an Analyst Brief, our team will help you create an impact solution for COVID-19 pandemic.
To understand the analysis and the market scenario contact us for an Analyst Brief, our team will help you create an impact solution for COVID-19 pandemic.