
Coronavirus disease (COVID-19) has expeditiously spread across the various countries of the globe, with large number of cases having been reported worldwide. This pandemic has first imprinted in the city of Wuhan, China from where it has badly affected almost every region of the world. The COVID-19 pandemic is not only creating health crisis but also it is affecting healthcare companies, economies and pharmaceutical sector as well as at their core.
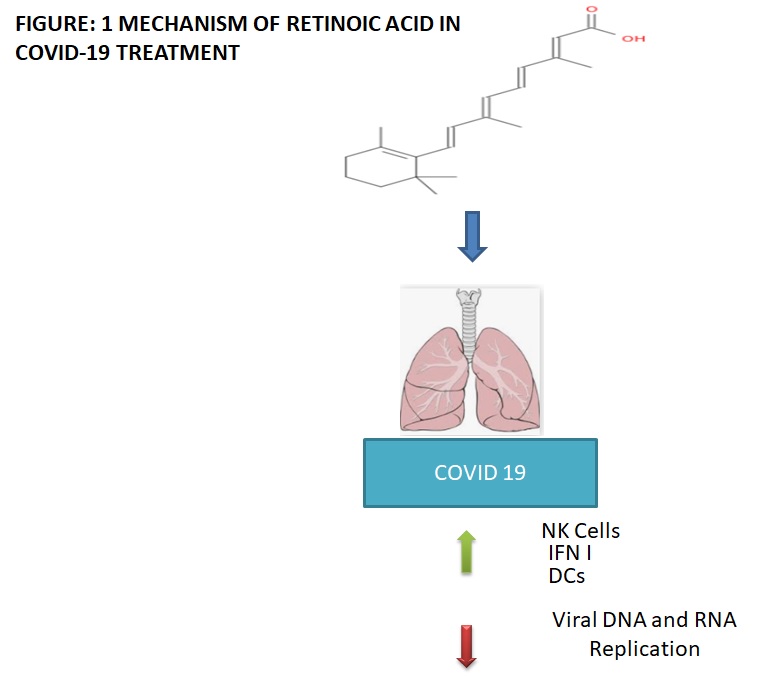
As COVID-19 is an infectious disease caused by SARS-CoV-2, its spread is endless, moreover, there is no specific vaccine and treatment solution approved yet for the coronavirus disease (COVID-19). During the pandemic of COVID-19, people are much more concerned about their health and are using retinoic acid in order to prevent both inflammation and viral entry which causes respiratory tract infections. Moreover, this has also been proved that isotretinoin can be given in the form of aerosol spray via respiratory route and hence acts as recommended treatment option for COVID-19.
However, retinoic acid manufacturing companies are finding it very much difficult to manufacture and distribute the drugs due to continuous locked down, disruption of supply chains and social distancing restrictions.
 PRICE IMPACT
PRICE IMPACTThe coronavirus disease (COVID-19) pandemic has enormous impact on global pharmaceutical market involving retinoic acid drugs among others. The demand for retinoic acid drugs is on surge across the globe because of increasing prevalence of coronavirus disease (COVID-19) all over the world and demand of effective treatment options. Various studies conducted demonstrated the efficacy of retinoic acid in the treatment of COVID-19.
For instance,
As the usage of retinoic acid is increasing all over the world in order to fulfill the need of COVID-19 treatment, the price of retinoic acid is expected to increase.
For instance,
If the COVID-19 continues to spread for a long interval of time, then it is estimated that demand of retinoic acid will highly upsurge. Moreover, the Australian Commission has also put major emphasis on the use of Roaccutane to treat coronavirus 2 (SARS-CoV-2) infections which is an isotretinoin drug.
Owing to sudden lift in demand of retinoic acid, the manufacturers are struggling to supply the requirements. Additionally, if the lockdown continues to stretch for long period then import of retinoic acid drug estimated to be banned which may leads to rise in price of retinoic acid. Resulting, it is predicted that price of the retinoic acid drugs is anticipated to bolster in the forecasted period.
The COVID-19 pandemic has transformed many organizations’ business environment, supply availability of various drugs has been hampered by a number of issues, including travel and export restrictions by countries and lockdowns that have forced suppliers to (temporarily) shut down the manufacturing and supply capabilities. China has a predominant performance in the global retinoic acid market. China has secured a considerable market share of vitamin market at global level. China exports about 17.87% of vitamin APIs including retinoic acids among others.
Moreover, this has been reported that European pharmaceutical companies also relies on India for manufacturing of their products and India is largely dependent on China for 70% of APIs including vitamin A APIs among others. Moreover, retinoic acid is extracted from the plants and pesky animals, however the activities involving in extraction of retinoic acids from animal source are not performed on upscale which has dropped the level of manufacturing and drug supply containing retinoic acids.
Since 1990, the U.S. is heavily relied on India and China for manufacturing of pharmaceutical products involving isotretinoin drugs among others because China provides cheaper ingredients and those are not subjected to that much high regulatory rules and laws.
Moreover, this has been projected out that UNICEF procures most of the pharmaceutical products from China and India and few of them from Europe as well. For Vitamin A capsules containing retinol, UNICEF and CIK depends on same supplier and also procures from Canada.
Moreover, as the market players are totally aware of the loss that they may likely to suffer due to disruption of supply chain, they are continuously monitoring their product supplies and manufacturing capabilities in order to prevent the loss to their overall business.
For instance,
This signifies that COVID-19 is disrupting the supply chain of APIs, it is also restricting the procurement procedure of retinoic acids from different sources among other factors but several initiatives are taken by the manufacturers and government organizations is creating a hope for the improvement of supply chain of retinoic acids.
Coronavirus disease (COVID-19) is one of the infectious diseases caused by SARS-CoV-2 virus and has affected and still affecting a large pool of the population worldwide. Coronavirus disease (COVID-19) causes acute respiratory illness ranges from mild to severe pneumonia along with respiratory failure. As there is no specific and approved treatment to treat the coronavirus disease (COVID-19), the need of effective treatment is at high demand, which can be catered by prescribing the retinoic acid drug which has the efficacy to enhance immune response by boosting up the activity of immune cells. Owing to all these affirmative factors, there is direct requirement of retinoic acid drugs indicating the future demand of retinoic acids.
By recognizing the actual as well as prompt increase in customer demand or anticipated prospective increase in customer demand, healthcare organization as well as government bodies’ calls on industry to increase manufacturing of retinoic acids.
Moreover, several initiatives are taken by the government in order to meet the demand of drug manufacturing.
For instance,
The department of pharmaceuticals (DoP, India) has proposed the creation of new manufacturing facility of rupees 3,000 crore in order to allow companies to set up the new manufacturing facilities for APIs involving retinoic acid among others.
TABLE 1. CLINICAL TRIALS IN ORDER TO PROVE THE EFFICACY OF RETINOIC ACID FOR COVID-19 TREATMENT
|
Study Title |
Interventions |
NCT Number |
|
Assessment the Activity Value of Isotretinoin (13- Cis-Retinoic Acid ) in the Treatment of COVID-19 (Randomized) |
Drug: Drug Isotretinoin (13 Cis Retinoic Acid) Capsules+Standard Treatment |
NCT04353180 |
|
Efficacy of Aerosol Combination Therapy of 13 Cis Retinoic Acid and Captopril for Treating Covid-19 Patients via Indirect Inhibition of Transmembrane Protease, Serine 2 (TMPRSS2) |
Combination Product: Aerosolized 13 cis retinoic acid plus Inhalation administration by nebulization captopril 25mg |
NCT04578236 |
|
Combination of Chemopreventive Agents (All- Trans Retinoic Acid and Tamoxifen) as Potential Treatment for the Lung Complication of COVID-19 |
Combination Product: Aerosolized All-Trans Retinoic acid plus oral Tamoxifen |
NCT04568096 |
|
Aerosol Combination Therapy of All-trans Retinoic Acid and Isotretinoin as A Novel Treatment for Inducing Neutralizing Antibodies in COVID -19 Infected Patients Better than Vaccine : An Innovative Treatment (Antibodies) |
|
NCT04396067 |
|
Efficacy and Safety of Drug Combination Therapy of Isotretinoin and Some Antifungal Drugs as A Potential Aerosol Therapy for COVID-19 : An Innovative Therapeutic Approach COVID-19 (Isotretinoin) |
Drug: Drug Isotretinoin (Aerosolized 13 Cis Retinoic Acid) plus Aerosolized Itraconazole |
NCT04577378 |
|
Combination of Recombinant Bacterial ACE2 Receptors -Like Enzyme of B38-CAP and Isotretinoin could be Promising COVID-19 Infection- and Lung Injury Preventing Drug Better than Recombinant Human ACE2 |
Combination Product: Recombinant Bacterial ACE2 receptors -like enzyme of B38-CAP (rbACE2) plus Aerosolized 13 cis retinoic acid |
NCT04382950 |
|
Combination Therapy With Isotretinoin and Tamoxifen Expected to Provide Complete Protection against Severe Acute Respiratory Syndrome Coronavirus (Combination) |
|
NCT04389580 |
|
Isotretinoin in Treatment of COVID-19 (Randomized) |
Drug: Isotretinoin Only Product in Oral Dose Form |
NCT04361422 |
Disproportionately high COVID-19 patient volumes putting strain on scarce drugs and therapies. Henceforth, it is estimated that global retinoic acid market has been anticipated to boom up in the forecasted period.
Sudden voluminous demand for retinoic acid drugs in order to fight against the COVID-19 has led the manufacturers to take several strategic initiatives. Large number of population affected with this COVID-19 virus and expanding spread across the world from last few months has enhanced the need of effective treatment options. To deal with the COVID-19 outbreak challenges, many retinoic acid manufacturers are creating new strategies. To meet an actual as well as prompt increase in customer demand or anticipated prospective increase in customer demand, the retinoic acid market players are exclusively concentrating on gaining the production capacity. It is predicted that these strategies are anticipated to escalate up the company revenue and its growth in the forecasted period.
For instance,
This thus signifies that various strategic initiatives adopted by the companies allow them to attain a lucrative growth even during the COVID-19 pandemic.
It is predicted that the COVID-19 pandemic outbreak have significant impact on the growth of global retinoic acid market owing to huge elevation in the demand of retinoic acid drugs. Moreover, many governments and healthcare organizations (United Nations Children's Fund (UNICEF) and partners, World Health Organization) are exclusively focusing on the supply of retinoic acid and its manufacturing owing to priorities and essential requirement in this crucial period. Moreover, several clinical trials are also expecting to provide market with a lucrative growth.