Asia-Pacific CRISPR Gene Detection and Diagnostic Market, By Class (Class 1- Multiple Effector Proteins and Class 2 -Single CrRNA-Binding Protein), Products & Services (Products and Services), Application (Biomedical Diagnostics, Genome Engineering, Drug Discovery, Agricultural Applications and Others), Workflow (Sample Preparation, Pre-Amplification, CrRNA, Cas Enzymes and Sensing), End User (Hospitals, Diagnostic Centers, Biotechnology Companies, Academic and Research Institutes and Others), Distribution Channel (Direct Tender, Retail Sales) Industry Trends and Forecast to 2029
Market Definition and Insights
CRISPR is clustered regularly interspaced short palindromic repeats and is a tool for genome editing, it allows researchers to alter DNA sequences and modify gene function easily. It has many potential applications, including correcting genetic defects and treating and preventing the spread of diseases. CRISPR-based diagnostics have been used for many biomedical applications, such as sensing nucleic-acid-based biomarkers of infectious and non-infectious diseases and detecting genetic diseases. The assay kits in CRISPR are composed of two components: a protein called Cas9 and a guide RNA, a string of nucleic acid molecules with a certain genetic code.
This CRISPR-Cas9 system has been modified for use in mammalian cells. We can either knock out specific genes by introducing a guide sequence (sgRNA) specific to our gene of interest by introducing frameshift mutations via Non-Homologous End Joining (NHEJ) or generate knock-in mutations.
CRISPR-Cas 9 systems have extended the scope of diagnostics and services in gene and cell therapies. Pharmaceutical companies invest heavily in R&D to develop new products, with a surge of gene and cell therapy agents entering early development. The market players investing would allow producing safe and effective treatments for patients in serious need.
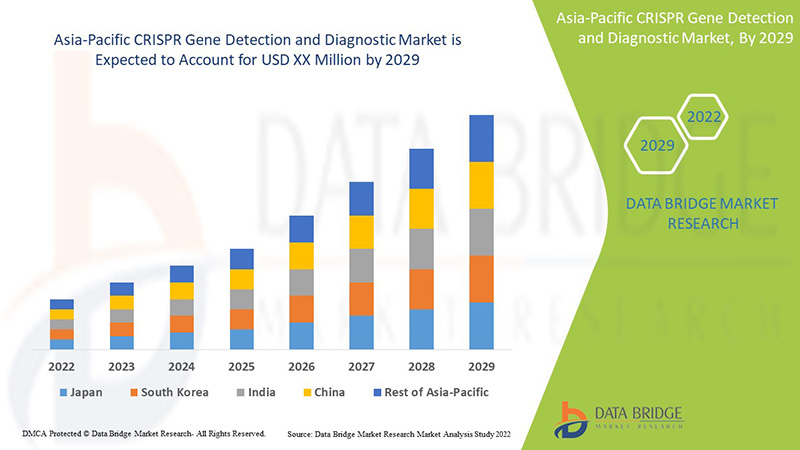
The Asia-Pacific CRISPR gene detection and diagnostic is supportive and aims to reduce the severity of the symptoms. Data Bridge Market Research analyses that the CRISPR gene detection and diagnostic market will grow at a CAGR of 21.6% during the forecast period of 2022 to 2029.
|
Report Metric |
Details |
|
Forecast Period |
2022 to 2029 |
|
Base Year |
2021 |
|
Historic Years |
2020 (Customizable to 2019 - 2014) |
|
Quantitative Units |
Revenue in USD Million, Pricing in USD |
|
Segments Covered |
Por clase (Clase 1: proteínas efectoras múltiples y Clase 2: proteína de unión a ARNcr simple), Productos y servicios (Productos y servicios), Aplicación (Diagnóstico biomédico, Ingeniería genómica, Descubrimiento de fármacos, Aplicaciones agrícolas y otros), Flujo de trabajo (Preparación de muestras, Preamplificación, ARNcr, Enzimas Cas y detección), Usuario final (Hospitales, Centros de diagnóstico, Empresas de biotecnología, Institutos académicos y de investigación y otros), Canal de distribución (Licitación directa, Ventas minoristas), |
|
Países cubiertos |
China, Japón, Corea del Sur, India, Australia, Singapur, Tailandia, Malasia, Indonesia, Filipinas y el resto de Asia-Pacífico |
|
Actores del mercado cubiertos |
GenScript, Takara Bio Inc., OriGene Technologies, Inc., Agilent Technologies, Inc., Synthego, Merck KGaA, Integrated DNA Technologies, Inc. (una subsidiaria de Danaher) Thermo Fisher Scientific Inc., entre otros. |
Dinámica del mercado de detección y diagnóstico de genes mediante CRISPR en Asia y el Pacífico
Conductores
- Aumento de la prevalencia e incidencia de enfermedades crónicas
Las enfermedades crónicas son afecciones de salud comunes, y uno de cada tres adultos padece alguna de ellas. Las enfermedades crónicas han afectado la salud y la calidad de vida de muchos ciudadanos.
CRISPR es la abreviatura de repeticiones palindrómicas cortas agrupadas y regularmente interespaciadas. En los últimos años, CRISPR se ha convertido en una herramienta de gran utilidad para la edición genética, que se utiliza para alterar las secuencias específicas de ADN en una célula. CRISPR tiene un uso importante en la investigación y el tratamiento de la enfermedad de Huntington, la distrofia muscular, el cáncer y el colesterol alto.
Por ejemplo,
- En 2021, los datos de NORD - National Organization for Rare Disorders, Inc. indicaron la incidencia diagnosticada de distrofia muscular de Duchenne (DMD). La distrofia muscular de Duchenne (DMD) es una enfermedad genética frecuente que afecta a 1 de cada 3500 nacimientos de varones en todo el mundo.
- Aumento de la inversión en investigación y desarrollo
Las tecnologías de edición genética, como el sistema CRISPR-Cas 9, han ampliado el alcance de los diagnósticos y servicios en terapias génicas y celulares. Las compañías farmacéuticas invierten mucho en I+D para desarrollar nuevos productos, y se ha producido un aumento de agentes de terapia génica y celular en las primeras etapas de desarrollo. La inversión de los actores del mercado permitiría alcanzar el objetivo de producir tratamientos seguros y eficaces para pacientes con necesidades graves.
Por ejemplo,
- En febrero de 2022, Synthego había recaudado 200 millones de dólares como inversión para investigación y desarrollo con el fin de impulsar el desarrollo de medicamentos basados en CRISPR desde la fase inicial de investigación hasta la fase clínica. Synthego utilizará el monto de inversión de la financiación de la Serie E para acelerar la creación de diagnósticos y servicios CRISPR.
Disponibilidad de financiación para el diagnóstico genético CRISPR
El Instituto Nacional de Salud (NIH) financia el diagnóstico y la investigación de genes mediante CRISPR. El sector privado también financia la detección e investigación de genes mediante CRISPR, pero esa inversión generalmente ocurre más tarde, durante la fase de prueba y desarrollo, y luego durante la investigación básica inicial. Como la edición genómica es un campo tan nuevo, un organismo gubernamental imparcial debe supervisarlos; la FDA es cautelosa y minuciosa, pero lucha sin descanso por obtener fondos, lo que hace una inversión a largo plazo que alinea el pago con los posibles beneficiarios futuros., mejorará aún más el crecimiento del mercado de detección y diagnóstico de genes mediante CRISPR.
Además, el avance en el diagnóstico genético CRISPR, las crecientes iniciativas de organizaciones públicas y privadas para difundir la conciencia y el aumento de la financiación gubernamental son los factores que expandirán el mercado de detección genética CRISPR en Asia y el Pacífico. Otros factores, como el aumento de la demanda de terapias efectivas y la creciente conciencia sobre el diagnóstico oportuno, tendrán un impacto positivo en la tasa de crecimiento del mercado de detección y diagnóstico genético CRISPR. Además, los altos ingresos disponibles, el creciente número de enfermedades crónicas y el cambio en el estilo de vida darán como resultado la expansión del mercado de detección y diagnóstico genético CRISPR.
Oportunidades
- El aumento del gasto sanitario
Además, el aumento de las actividades de investigación y desarrollo y el aumento de las inversiones por parte del gobierno y de organizaciones privadas impulsarán nuevas oportunidades para la tasa de crecimiento del mercado.
- Iniciativa estratégica de los actores del mercado
La demanda de detección y diagnóstico de genes mediante CRISPR ha aumentado en los EE. UU. y la región de Asia y el Pacífico debido al tratamiento oportuno de enfermedades crónicas. Estos factores favorables aumentan la necesidad de medicamentos y, para satisfacer la demanda del mercado, los actores principales y secundarios del mercado están utilizando diversas estrategias.
Los principales actores también están tratando de diseñar estrategias específicas, como lanzamientos de productos, adquisiciones, aprobaciones, expansiones y asociaciones, para garantizar el buen funcionamiento del negocio, evitar riesgos y aumentar el crecimiento a largo plazo de las ventas del mercado.
Por ejemplo,
- En mayo de 2021, Horizon Discovery Ltd. amplió su cartera de modulación genética con el primer ARN guía sintético único y el represor dcas9 pendiente de patente para la interferencia CRISPR en Waltham. La expansión de la cartera había aumentado las ventas y los ingresos de la cartera de ARN guía sintético en la región de EE. UU. y el Reino Unido y había aumentado la colaboración con los actores del mercado.
Además, el lanzamiento de terapias efectivas y ensayos clínicos continuos brindarán oportunidades beneficiosas para el mercado de detección y diagnóstico de genes CRISPR en el período de pronóstico de 2022 a 2029. Además, la gran necesidad insatisfecha de tecnología actual y los avances en el campo de la atención médica aumentarán la tasa de crecimiento del mercado de detección y diagnóstico de genes CRISPR en el futuro.
Restricciones/Desafíos
Sin embargo, el alto costo de los diagnósticos CRISPR y los riesgos que se enfrentan al utilizarlos impedirán la tasa de crecimiento del mercado de detección y diagnóstico de genes CRISPR. Además, los riesgos que se corren al utilizar los dispositivos de resonancia magnética obstaculizarán el crecimiento del mercado de detección y diagnóstico de genes CRISPR. La falta de conocimientos especializados y regulaciones supondrán un desafío adicional para el mercado en el período de pronóstico mencionado anteriormente.
- Aumento del coste de los diagnósticos basados en CRISPR
El enorme potencial de las terapias basadas en CRISPR tiene un coste. Las terapias de edición genómica máxima requieren una mayor cantidad de tiempo para su desarrollo y producción, y de ahí el aumento de los costes. Además, los kits de ensayo y los medicamentos relacionados con la detección y el diagnóstico de genes mediante CRISPR son aplicables a un gran sector de la población. Estos costes recaen sobre los pacientes, por lo que se espera que los elevados costes actuales muestren una tendencia descendente en el futuro.
Por ejemplo,
- En julio de 2021, según Integrated DNA Technologies, Inc., el primer ensayo de diagnóstico basado en CRISPR disponible comercialmente para el SARS-CoV-2 que incluye LAMP de transcripción inversa (RT-LAMP) como preamplificación está actualmente disponible a USD 30,15 por reacción.
El informe de mercado de detección y diagnóstico de genes CRISPR proporciona detalles de nuevos desarrollos recientes, regulaciones comerciales, análisis de importación y exportación, análisis de producción, optimización de la cadena de valor, participación de mercado, impacto de los actores del mercado nacional y localizado, analiza oportunidades en términos de bolsillos de ingresos emergentes, cambios en las regulaciones del mercado, análisis estratégico del crecimiento del mercado, tamaño del mercado, crecimientos del mercado de categorías, nichos de aplicación y dominio, aprobaciones de productos, lanzamientos de productos, expansiones geográficas, innovaciones tecnológicas en el mercado. Para obtener más información sobre el mercado de detección y diagnóstico de genes CRISPR, comuníquese con Data Bridge Market Research para obtener un informe de analista, nuestro equipo lo ayudará a tomar una decisión de mercado informada para lograr el crecimiento del mercado.
Análisis de la epidemiología de los pacientes
Según un estudio de Globocan, en 2020, el cáncer de mama tuvo una alta incidencia de casos, alrededor del 11,7%, seguido del cáncer de pulmón con un 11,40%, el cáncer colorrectal con un 10,00% y el cáncer de cuello uterino y esófago con un menor número de casos incidentes.
El mercado de detección y diagnóstico de genes CRISPR también le proporciona un análisis detallado del mercado para el análisis de pacientes, el pronóstico y las curas. La prevalencia, la incidencia, la mortalidad y las tasas de adherencia son algunas de las variables de datos que están disponibles en el informe. Se analizan los análisis de impacto directo o indirecto de la epidemiología en el crecimiento del mercado para crear un modelo estadístico multivariado de cohorte más sólido para pronosticar el mercado en el período de crecimiento.
Impacto de la COVID-19 en el mercado de detección y diagnóstico de genes mediante CRISPR
El COVID-19 ha afectado negativamente al mercado. Los confinamientos y el aislamiento durante las pandemias complican el diagnóstico, la gestión y el tratamiento. La falta de acceso a los centros sanitarios para la administración rutinaria y de medicamentos afectará aún más al mercado. El aislamiento social aumenta el estrés, la desesperación y el apoyo social, todo lo cual puede provocar una reducción de la adherencia a la medicación anticonvulsiva durante la pandemia.
Desarrollo reciente
- En agosto de 2020, SHERLOCK BIOSCIENCES anunció una colaboración con Dartmouth-Hitchcock Health para llevar a cabo el ensayo clínico del kit de diagnóstico SHERLOCK para el SARS-CoV-2. El kit recibió la aprobación de emergencia de la Autorización de uso de emergencia (EUA) de la Administración de Alimentos y Medicamentos de los Estados Unidos (FDA).
Alcance del mercado de detección y diagnóstico de genes mediante CRISPR en Asia y el Pacífico
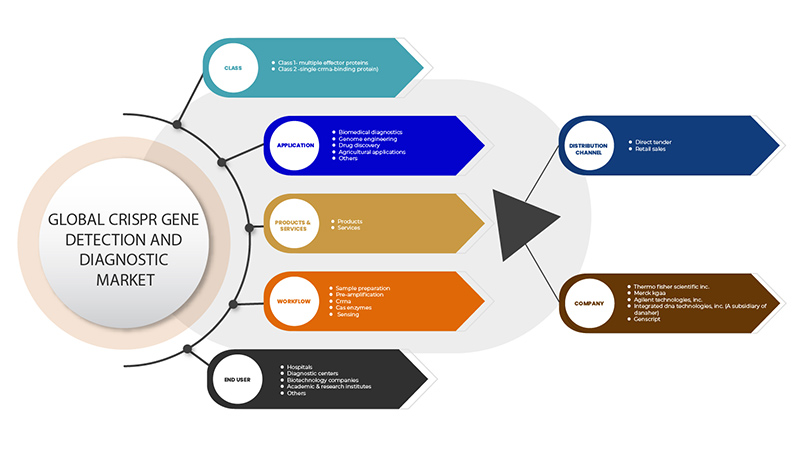
El mercado de detección y diagnóstico de genes mediante CRISPR está segmentado en base a seis segmentos: clase, productos y servicios, aplicación, flujo de trabajo, usuario final y canal de distribución. El crecimiento entre estos segmentos le ayudará a analizar los segmentos de crecimiento reducidos en las industrias y brindará a los usuarios una valiosa descripción general del mercado y conocimientos del mercado para ayudarlos a tomar decisiones estratégicas para identificar las principales aplicaciones del mercado.
Clase
- Clase 1: Proteínas efectoras múltiples
- Clase 2: proteína de unión a ARNcr simple
Sobre la base de la clase, el mercado de detección y diagnóstico de genes CRISPR de Asia y el Pacífico está segmentado en clase 1 (proteínas efectoras múltiples) y clase 2 (proteína de unión de ARN Cr único).
Productos y servicios
- Productos
- Servicios
Sobre la base de productos y servicios, el mercado de detección y diagnóstico de genes CRISPR de Asia-Pacífico está segmentado en productos y servicios.
Solicitud
- Diagnóstico biomédico
- Ingeniería Genómica
- Descubrimiento de fármacos
- Aplicaciones agrícolas
- Otros
Sobre la base de la aplicación, el mercado de detección y diagnóstico de genes CRISPR de Asia-Pacífico se segmenta en diagnóstico biomédico, ingeniería genómica, descubrimiento de fármacos, aplicaciones agrícolas y otros.
Flujo de trabajo
- Preparación de la muestra
- Preamplificación
- ARNcr
- Enzimas Cas
- Detección
Sobre la base del flujo de trabajo, el mercado de detección y diagnóstico de genes CRISPR de Asia-Pacífico está segmentado en preparación de muestras, preamplificación, CrRNA, enzimas Cas y detección.
Usuario final
- Hospitales
- Centros de diagnóstico
- Empresas de biotecnología
- Institutos académicos y de investigación
- Otros
Sobre la base del usuario final, el mercado de detección y diagnóstico de genes CRISPR de Asia-Pacífico está segmentado en hospitales, centros de diagnóstico, empresas de biotecnología, institutos académicos y de investigación y otros.
Canal de distribución
- Licitaciones directas
- Ventas al por menor
Sobre la base del canal de distribución, el mercado de detección y diagnóstico de genes CRISPR de Asia-Pacífico está segmentado en licitaciones directas y ventas minoristas.
Análisis y perspectivas regionales del mercado de detección y diagnóstico de genes mediante CRISPR
The Asia-Pacific CRISPR gene detection and diagnostic market is analysed and market size insights and trends are provided by regions, class, products & services, application, workflow, end user, and distribution channel as referenced above.
The countries covered in the CRISPR gene detection and diagnostic market report are China, Japan, South Korea, India, Australia, Singapore, Thailand, Malaysia, Indonesia, Philippines And Rest of Asia-Pacific.
The China dominates the CRISPR gene detection and diagnostic market due to the rise in clinical trials for CRISPR based diagnostics.
The country section of the report also provides individual market impacting factors and changes in regulations in the market domestically that impacts the current and future trends of the market. Data points such as new sales, replacement sales, country demographics, disease epidemiology and import-export tariffs are some of the major pointers used to forecast the market scenario for individual countries. Also, presence and availability of Asia-Pacific brands and their challenges faced due to large or scarce competition from local and domestic brands, impact of sales channels are considered while providing forecast analysis of the country data.
Competitive Landscape and CRISPR Gene Detection and Diagnostic Market Share Analysis
The Asia-Pacific CRISPR gene detection and diagnostic market competitive landscape provides details by competitor. Details included are company overview, company financials, revenue generated, market potential, investment in research and development, new market initiatives, the Asia-Pacific presence, production sites and facilities, production capacities, company strengths and weaknesses, product launch, product width and breadth, application dominance. The above data points provided are only related to the companies' focus related to the CRISPR gene detection and diagnostic market.
Some of the major players operating in the CRISPR gene detection and diagnostic market are GenScript, Takara Bio Inc., OriGene Technologies, Inc., Agilent Technologies, Inc., Synthego, Merck KGaA, Integrated DNA Technologies, Inc. (A subsidiary of Danaher) Thermo Fisher Scientific Inc. among others.
SKU-
Obtenga acceso en línea al informe sobre la primera nube de inteligencia de mercado del mundo
- Panel de análisis de datos interactivo
- Panel de análisis de empresas para oportunidades con alto potencial de crecimiento
- Acceso de analista de investigación para personalización y consultas
- Análisis de la competencia con panel interactivo
- Últimas noticias, actualizaciones y análisis de tendencias
- Aproveche el poder del análisis de referencia para un seguimiento integral de la competencia
Metodología de investigación
La recopilación de datos y el análisis del año base se realizan utilizando módulos de recopilación de datos con muestras de gran tamaño. La etapa incluye la obtención de información de mercado o datos relacionados a través de varias fuentes y estrategias. Incluye el examen y la planificación de todos los datos adquiridos del pasado con antelación. Asimismo, abarca el examen de las inconsistencias de información observadas en diferentes fuentes de información. Los datos de mercado se analizan y estiman utilizando modelos estadísticos y coherentes de mercado. Además, el análisis de la participación de mercado y el análisis de tendencias clave son los principales factores de éxito en el informe de mercado. Para obtener más información, solicite una llamada de un analista o envíe su consulta.
La metodología de investigación clave utilizada por el equipo de investigación de DBMR es la triangulación de datos, que implica la extracción de datos, el análisis del impacto de las variables de datos en el mercado y la validación primaria (experto en la industria). Los modelos de datos incluyen cuadrícula de posicionamiento de proveedores, análisis de línea de tiempo de mercado, descripción general y guía del mercado, cuadrícula de posicionamiento de la empresa, análisis de patentes, análisis de precios, análisis de participación de mercado de la empresa, estándares de medición, análisis global versus regional y de participación de proveedores. Para obtener más información sobre la metodología de investigación, envíe una consulta para hablar con nuestros expertos de la industria.
Personalización disponible
Data Bridge Market Research es líder en investigación formativa avanzada. Nos enorgullecemos de brindar servicios a nuestros clientes existentes y nuevos con datos y análisis que coinciden y se adaptan a sus objetivos. El informe se puede personalizar para incluir análisis de tendencias de precios de marcas objetivo, comprensión del mercado de países adicionales (solicite la lista de países), datos de resultados de ensayos clínicos, revisión de literatura, análisis de mercado renovado y base de productos. El análisis de mercado de competidores objetivo se puede analizar desde análisis basados en tecnología hasta estrategias de cartera de mercado. Podemos agregar tantos competidores sobre los que necesite datos en el formato y estilo de datos que esté buscando. Nuestro equipo de analistas también puede proporcionarle datos en archivos de Excel sin procesar, tablas dinámicas (libro de datos) o puede ayudarlo a crear presentaciones a partir de los conjuntos de datos disponibles en el informe.