يعكس الاستخدام المتزايد لخلايا CHO في الدراسات الجينية مزاياها الفريدة في مجال البحث الجيني. تشتهر خلايا CHO باستقرارها الجيني. يدرس الباحثون جينات أو تعديلات جينية محددة دون القلق من تداخل التغيرات الجينية غير المتوقعة مع تجاربهم. يُعد هذا الاستقرار بالغ الأهمية عند إجراء تعديلات جينية دقيقة أو دراسة الآثار طويلة المدى للتعديلات الجينية.
تُعدّ كفاءة خلايا CHO في التعبير عن البروتينات المُعاد تركيبها سببًا قويًا آخر لزيادة استخدامها في الأبحاث الجينية. يُمكن للباحثين إدخال جينات مُحددة تُشفّر البروتينات المُهمة في خلايا CHO. تعمل هذه الخلايا بعد ذلك كمفاعلات حيوية مُصغّرة تُنتج وتُفرز البروتينات المطلوبة. تُعد هذه القدرة أساسية لدراسة وظائف جينات مُحددة، ودراسة آثار الطفرات الجينية، وإنتاج بروتينات علاجية للتطبيقات البحثية والصيدلانية الحيوية.
يمكنك الوصول إلى التقرير الكامل على الرابط التالي: https://www.databridgemarketresearch.com/reports/north-america-chinese-hamster-ovary-cells-cho-market
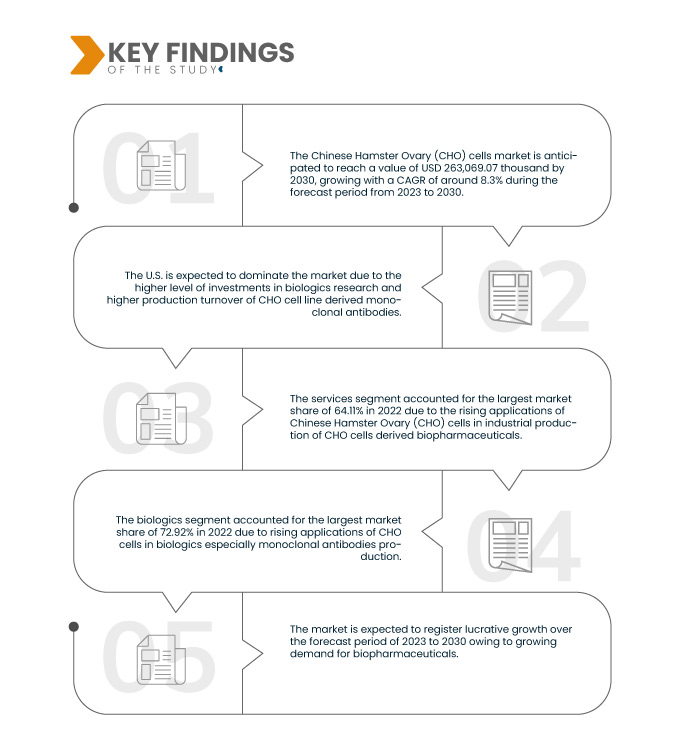
تحلل شركة Data Bridge Market Research أن سوق خلايا مبيض الهامستر الصيني (CHO) في أمريكا الشمالية من المتوقع أن ينمو بمعدل نمو سنوي مركب قدره 8.3٪ في الفترة المتوقعة من 2023 إلى 2030 ومن المتوقع أن يصل إلى 263،069.07 ألف دولار أمريكي بحلول عام 2030 من 143،106.10 ألف دولار أمريكي في عام 2022.
النتائج الرئيسية للدراسة
الطلب المتزايد على المستحضرات الصيدلانية الحيوية
أظهرت المستحضرات الصيدلانية الحيوية، المعروفة أيضًا باسم المواد البيولوجية ، نتائج علاجية ملحوظة في مجالات مثل علم الأورام، وأمراض المناعة الذاتية، والاضطرابات الوراثية النادرة. على سبيل المثال، أحدثت الأجسام المضادة وحيدة النسيلة ، وهي نوع شائع من المستحضرات الصيدلانية الحيوية، نقلة نوعية في علاج السرطان من خلال استهداف الخلايا السرطانية مباشرةً مع الحفاظ على الخلايا السليمة. كما تُقدم المستحضرات الصيدلانية الحيوية مناهج واعدة لإدارة حالات كانت مستعصية على العلاج سابقًا وأمراض نادرة، والتي غالبًا ما أهملتها شركات تطوير الأدوية التقليدية نظرًا لقلة أعداد مرضاها.
للطلب المتزايد على المستحضرات الصيدلانية الحيوية تأثير إيجابي على السوق، إذ تُعدّ خلايا CHO أداةً أساسيةً لإنتاج المستحضرات الصيدلانية الحيوية. ويتزامن ذلك مع ازدياد الطلب على خلايا CHO لإنتاج هذه الأدوية البيولوجية المعقدة، مع توسع صناعة المستحضرات الصيدلانية الحيوية لتلبية الحاجة المتزايدة إلى علاجات مبتكرة وفعالة.
نطاق التقرير وتقسيم السوق
مقياس التقرير
|
تفاصيل
|
فترة التنبؤ
|
من 2023 إلى 2030
|
سنة الأساس
|
2022
|
السنوات التاريخية
|
2021 (قابلة للتخصيص حتى 2015-2020)
|
الوحدات الكمية
|
الإيرادات بالألف دولار أمريكي، ومتوسط سعر البيع بالدولار الأمريكي، والحجم بالوحدات
|
القطاعات المغطاة
|
النوع (الخدمات والمنتج)، النظام (نظام اختيار الأيض، نظام اختيار المضادات الحيوية، وغيرها)، التطبيق (البيولوجيا والأبحاث الطبية)، المستخدم النهائي (شركات الأدوية الحيوية، شركات التكنولوجيا الحيوية، منظمات التطوير والتصنيع السريري، منظمات الأبحاث السريرية، المعاهد الأكاديمية ومنظمات البحث، وغيرها)، قناة التوزيع (العطاءات المباشرة، مبيعات التجزئة، وغيرها)
|
الدول المغطاة
|
الولايات المتحدة وكندا والمكسيك
|
الجهات الفاعلة في السوق المغطاة
|
Thermo Fisher Scientific Inc. (الولايات المتحدة)، AcceGen (الولايات المتحدة)، RayBiotech Life, Inc. (الولايات المتحدة)، CLS Cell Lines Service GmbH (ألمانيا)، BPS Bioscience, Inc. (الولايات المتحدة)، GenTarget Inc. (الولايات المتحدة)، Merck KGaA (ألمانيا)، Promega Corporation (الولايات المتحدة)، Abeomics (الولايات المتحدة)، Applied Biological Materials Inc. (abm) (كندا)، ATCC (الولايات المتحدة)، Sartorius AG (ألمانيا)، Lonza (سويسرا)، Horizon Discovery Ltd. (المملكة المتحدة)، Cytiva (الولايات المتحدة) و Curia Global, Inc. (الولايات المتحدة)، من بين شركات أخرى.
|
نقاط البيانات التي يغطيها التقرير
|
بالإضافة إلى الرؤى حول سيناريوهات السوق مثل القيمة السوقية ومعدل النمو والتجزئة والتغطية الجغرافية واللاعبين الرئيسيين، فإن تقارير السوق التي تم تنظيمها بواسطة Data Bridge Market Research تتضمن أيضًا تحليلًا متعمقًا من الخبراء وعلم الأوبئة للمرضى وتحليل خطوط الأنابيب وتحليل التسعير والإطار التنظيمي.
|
تحليل القطاعات
يتم تقسيم سوق خلايا مبيض الهامستر الصيني (CHO) في أمريكا الشمالية إلى خمسة قطاعات بارزة بناءً على النوع والنظام والتطبيق والمستخدم النهائي وقناة التوزيع.
- على أساس النوع، يتم تقسيم السوق إلى خدمات ومنتجات.
من المتوقع أن يهيمن قطاع الخدمات على سوق خلايا مبيض الهامستر الصيني (CHO) في أمريكا الشمالية في عام 2023
ومن المتوقع أن تهيمن شريحة الخدمات على السوق في عام 2023 بحصة سوقية تبلغ 64.46% بسبب الطلب المتزايد على الخدمات المتخصصة المتعلقة بتطوير خطوط خلايا CHO والمعالجة الحيوية والتصنيع التعاقدي.
- على أساس النظام، يتم تقسيم السوق إلى نظام الاختيار الأيضي، ونظام اختيار المضادات الحيوية، وغيرها.
في عام 2023، من المتوقع أن يهيمن قطاع نظام الاختيار الأيضي على سوق خلايا مبيض الهامستر الصيني (CHO) في أمريكا الشمالية
ومن المتوقع أن تهيمن شريحة نظام الاختيار الأيضي على السوق في عام 2023 بحصة سوقية تبلغ 73.17% بسبب كفاءتها في توليد خطوط خلايا CHO عالية الإنتاج لإنتاج المستحضرات الصيدلانية الحيوية.
- بناءً على التطبيق، يُقسّم السوق إلى قسمين: قسم المواد البيولوجية وقسم الأبحاث الطبية. ومن المتوقع أن يُهيمن قطاع المواد البيولوجية على السوق بحصة سوقية تبلغ 73.02% في عام 2023.
- بناءً على المستخدم النهائي، يُقسّم السوق إلى شركات الأدوية الحيوية، وشركات التكنولوجيا الحيوية، ومؤسسات التطوير والتصنيع السريري، ومؤسسات الأبحاث السريرية، والمعاهد الأكاديمية ومراكز البحث، وغيرها. في عام 2023، من المتوقع أن تهيمن شركات الأدوية الحيوية على السوق بحصة سوقية تبلغ 43.02%.
- بناءً على قنوات التوزيع، يُقسّم السوق إلى مناقصات مباشرة، ومبيعات تجزئة، وغيرها. وفي عام ٢٠٢٣، من المتوقع أن تهيمن المناقصات المباشرة على السوق بحصة سوقية تبلغ ٦٦.٧٨٪.
اللاعبون الرئيسيون
تحلل شركة Data Bridge Market Research أن شركات Lonza (سويسرا)، وSartorius AG (ألمانيا)، وMerck KGaA (ألمانيا)، وCytiva (الولايات المتحدة)، وThermo Fisher Scientific Inc. (الولايات المتحدة) هي اللاعبين الرئيسيين في سوق خلايا مبيض الهامستر الصيني (CHO) في أمريكا الشمالية.
تطورات السوق
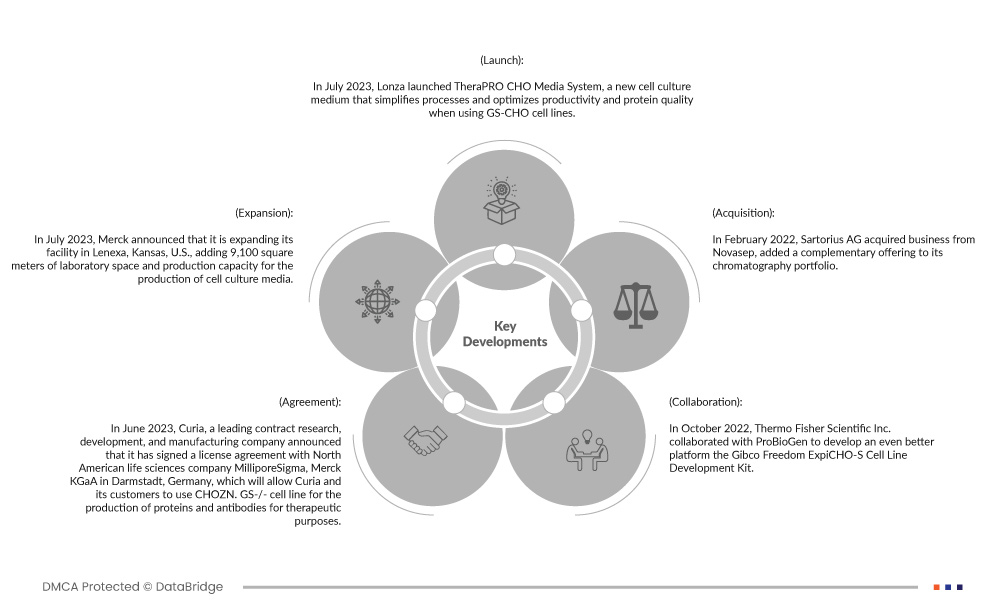
- في يوليو 2023، أطلقت شركة لونزا نظام TheraPRO CHO Media، وهو وسط زراعة خلايا جديد يُبسّط العمليات ويُحسّن الإنتاجية وجودة البروتين عند استخدام سلالات خلايا GS-CHO. تدعم هذه الشركة الناشئة شركات الأدوية والتكنولوجيا الحيوية التي تُنتج بروتينات علاجية لتحسين جودة المنتجات بشكل أكبر. يوفر نظام TheraPRO CHO Media أداءً فعالاً، مُحققًا تركيزات عالية من الخلايا الحيوية ومستويات بروتين تتجاوز 5 جم/لتر خلال فترة زراعة مدتها 15 يومًا. يُمثل هذا أكثر من ضعف مستوى البروتين الذي يُمكن إنتاجه باستخدام الحلول المتاحة تجاريًا. وقد ساعد هذا الإطلاق الشركة على توسيع محفظة منتجاتها في السوق.
- في يوليو 2023، أعلنت شركة ميرك عن توسعة منشأتها في لينيكسا، كانساس، الولايات المتحدة الأمريكية، بإضافة 9100 متر مربع من مساحة المختبرات وطاقة إنتاجية لإنتاج وسائط زراعة الخلايا. يجعل هذا التوسع لينيكسا أكبر منشأة لزراعة الخلايا بالمسحوق الجاف تابعة للشركة، ومركزًا للتميز في أمريكا الشمالية. يعكس هذا الاستثمار في المنطقة استراتيجية الشركة لتوسيع وتنويع سلسلة التوريد لتلبية الطلب الحالي والمستقبلي على منصات زراعة الخلايا.
- في يونيو 2023، أعلنت شركة كوريا، الشركة الرائدة في مجال البحث والتطوير والتصنيع التعاقدي، عن توقيعها اتفاقية ترخيص مع شركة ميليبور سيجما، وهي شركة أمريكية شمالية متخصصة في علوم الحياة، ومقرها دارمشتات بألمانيا. تتيح هذه الاتفاقية لشركة كوريا وعملائها استخدام خط خلايا CHOZN.GS-/- لإنتاج البروتينات والأجسام المضادة لأغراض علاجية. وعقب هذه الاتفاقية، سيتم دمج خط خلايا CHOZN GS-/- في سير عمل تطوير خطوط الخلايا في شركة كوريا، وسيكون متاحًا فورًا لعملائها.
- في أكتوبر 2022، تعاونت شركة Thermo Fisher Scientific Inc. مع ProBioGen لتطوير منصة أفضل، وهي مجموعة تطوير سلالات الخلايا Gibco Freedom ExpiCHO-S. تتيح هذه المجموعة للمستخدمين توليد سلالات خلوية مناسبة للتطوير السريري دون الحاجة إلى خلاياهم الأصلية أو نواقلهم أو خبرتهم السابقة في هذا المجال. ساهمت ProBioGen بشكل كبير في أداء مجموعة Freedom ExpiCHO-S من خلال الاستفادة من خبرتها الواسعة في تطوير سلالات الخلايا والعمليات. تستخدم السلسلة الجديدة سلالة خلايا ExpiCHO-S من Thermo Fisher، مما يوسع محفظة منتجات الشركة في سلسلة تطوير سلالات الخلايا CHO.
- في فبراير 2022، استحوذت شركة سارتوريوس إيه جي على أعمال شركة نوفاسيب، وأضافت عرضًا مكملًا إلى محفظة منتجاتها الكروماتوغرافية. تشمل المحفظة المستحوذة أنظمة كروماتوغرافيا مُصممة أساسًا للجزيئات الحيوية الصغيرة، مثل الأوليغونوكليوتيدات والببتيدات والأنسولين، بالإضافة إلى أنظمة مبتكرة للإنتاج المستمر للمستحضرات الصيدلانية الحيوية.
التحليل الإقليمي
من الناحية الجغرافية، المناطق التي يغطيها تقرير السوق هي الولايات المتحدة وكندا والمكسيك.
وفقًا لتحليل Data Bridge Market Research:
الولايات المتحدة هي الدولة المهيمنة والأسرع نموًا في سوق خلايا مبيض الهامستر الصيني (CHO) في أمريكا الشمالية
تُهيمن الولايات المتحدة على سوق خلايا مبيض الهامستر الصيني (CHO) بفضل ارتفاع مستوى الاستثمارات في صناعة الأدوية الحيوية. وستظل الولايات المتحدة أسرع الدول نموًا في سوق خلايا مبيض الهامستر الصيني (CHO) في أمريكا الشمالية، وذلك بفضل التطبيقات المتنامية لمزارع خلايا الثدييات، وخاصةً خلايا CHO، في الأبحاث الطبية والتكنولوجية الحيوية .
للحصول على معلومات أكثر تفصيلاً حول تقرير سوق خلايا مبيض الهامستر الصيني (CHO) في أمريكا الشمالية، انقر هنا - https://www.databridgemarketresearch.com/reports/north-america-chinese-hamster-ovary-cells-cho-market