Market Analysis and Insights
Glioblastoma multiforme (GBM) is a grade IV WHO malignant tumor with astrocytic differentiation. As one of the most common clinically diagnosed central nervous system (CNS) oncological entries, there have been a wide variety of historical reports of the description and evolution of ideas regarding these tumors. The first recorded reports of gliomas were given in British scientific reports, by Berns in 1800 and in 1804 by Abernety, with the first comprehensive histomorphological description being given in 1865 by Rudolf Virchow. In 1926 Percival Bailey and Harvey Cushing gave the base for the modern classification of gliomas. Between 1934 and 1941 the most prolific researcher in glioma research was Hans-Joachim Scherer, who postulated some of the clinico-morphological aspects of GBM. With the introduction of molecular and genetic tests the true multifomity of GBM has been established, with different genotypes bearing the same histomorphological and IHC picture, as well as some of the aspects of gliomagenesis. For a GBM to develop, a specific trigger mutation needs to occur in a GBM stem cell - primary GBM, or a slow aggregation of individual mutations, without a distinct trigger mutation - secondary GBM. Knowledge of GBM has been closely related to general medical knowledge of the CNS since these malignancies were first described more than 200 years ago. Several great leaps have been made in that time, in the footsteps of both CNS and advancements in general medical knowledge. The demand for glioblastoma multiforme treatment is increasing, for which manufacturers are involved in the new product launches, increasing pipeline products and event participation in the market. These decisions are ultimately enhancing the growth of the market.

The glioblastoma multiforme treatment market report provides details of market share, new developments, impact of domestic and localized market players, analyses opportunities in terms of emerging revenue pockets, changes in market regulations, product approvals, strategic decisions, product launches, geographic expansions, and technological innovations in the market. To understand the analysis and the market scenario contact us for an analyst brief, our team will help you create a revenue impact solution to achieve your desired goal. Strategic initiatives such as collaboration, agreement, and signing of sales agreements to invent and innovate pharmacological treatments are the major drivers which propelled the demand of the market in the forecast period.
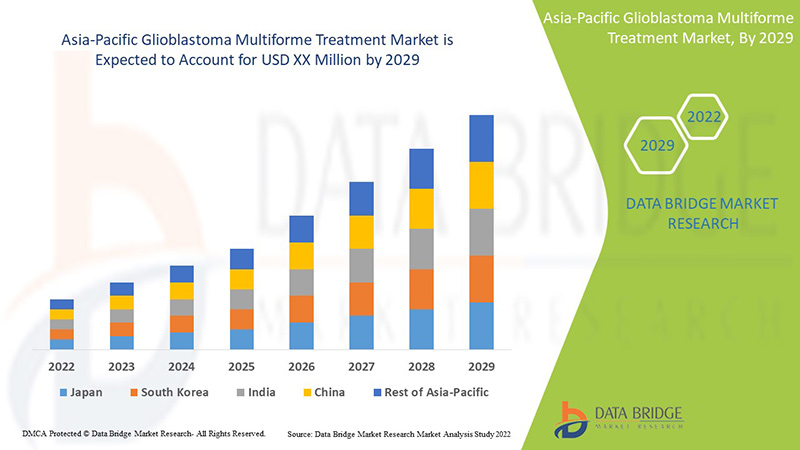
The glioblastoma multiforme treatment market is supportive and aims to reduce the progression of the disease. Data Bridge Market Research analyses that glioblastoma multiforme treatment market will grow at a CAGR of 9.2% during the forecast period of 2022 to 2029.
|
Report Metric |
Details |
|
Forecast Period |
2022 to 2029 |
|
Base Year |
2021 |
|
Historic Years |
2020 (Customizable to 2019 - 2014) |
|
Quantitative Units |
Revenue in USD Million, Pricing in USD |
|
Segments Covered |
By Type (Primary (De Novo), Secondary), Treatment (Surgery, Radiotherapy, Medications), Patient Type (Adult, Geriatric, Child), Drug Type (Generics, Branded), Route of Administration (Parenteral, Oral, Others), End User (Hospitals, Clinics, Home Healthcare, Others), Distribution Channel (Hospital Pharmacy, Retail Pharmacy, Online Pharmacy, Others) |
|
Countries Covered |
China, Japan, India, South Korea, Singapore, Malaysia, Australia, Thailand, Indonesia, Philippines, Vietnam, Rest of Asia-Pacific. |
|
Market Players Covered |
F. Hoffmann-La Roche AG, Amgen Inc., Merck & Co., Inc., Pfizer Inc., Varian Medical Systems, Inc. (A subsidiary of Siemens Healthcare), ZEISS International, Amneal Pharmaceuticals LLC, Elekta, Sun Pharmaceutical Industries Ltd, Teva Pharmaceutical Industries Ltd., Eckert & Ziegler, Accord Healthcare, Angiochem, ANI Pharmaceuticals, Inc., Arbor Pharmaceuticals, LLC. (A Subsidiary of Azurity Pharmaceuticals, Inc.), AstraZeneca, Cantex Pharmaceuticals, Inc., CELON LABS, Diffusion Pharmaceuticals Inc., EnGeneIC, ERC.SA., Genenta science, Jazz Pharmaceuticals, Inc., Loxo Oncology (A Subsidiary of Eli Lilly), Novartis AG, VBL THERAPEUTICS, Viatris Inc., and Zydus Pharmaceuticals, Inc., among others. |
MARKET DEFINITION
Glioblastoma multiforme (GBM) is the most common and aggressive primary malignant brain tumor and accounts for 60% of brain tumors in adults. GBMs can arise in the brain de novo or evolve from lower-grade astrocytoma. In adults, GBM occurs most often in the cerebral hemispheres, especially in the frontal and temporal lobes of the brain. Many genetic and environmental factors have been studied in glioblastoma multiforme, but no risk factor that accounts for a large proportion of GBM has been identified. So like many other cancers, GBM is sporadic, although some studies indicate a high prevalence (17%) of prior therapeutic irradiation among patients with GBM. The latency between irradiation and the development of GBM varies from a few years to several decades. There is no substantial evidence of GBM association with lifestyle factors like smoking, alcohol consumption, drug use, or exposure to N- Nitroso compounds. Studies have shown that the use of mobile phones doesn't increase the risk of development of GBM; however, its association with long term use needs further confirmation.
Glioblastoma Multiforme Treatment Market Dynamics
Drivers
- Growing prevalence of glioblastoma multiforme
Glioblastoma multiforme (GBM) is the most commonly occurring malignant primary brain tumor, representing 77%-81% of all central nervous system (CNS) primary malignant tumors. The World Health Organization classified it as a grade IV diffuse astrocytic and oligodendroglial tumor. The mean age of primary GBM presentation is 62 years, and the median survival is approximately 14.6 months. The poor prognosis associated with GBM is well documented, while survival rates remain disappointingly low despite medical and surgical advances. As per the study, International studies reveal an approximate annual incidence rate of 0.59 to 5 per 100,000 persons; however, studies indicate a rise in incidence. Miranda-Filho et al. in 2017 described increasing rates of CNS and brain cancers in countries in South America, Eastern Europe, and Southern Europe, while decreasing rates were only reported in Japan. Dobes et al. in 2011 have also noted an increasing incidence of GBM tumors in two of their multicentered Australian studies, with a particular increase in frontal and temporal lobe GBM tumors. The increased incidence of glioblastoma multiforme raises the demand for early detection and diagnosis via utilizing the latest technology, propelling the global glioblastoma multiforme treatment market. The rising incidence of glioblastoma across the globe is expected to accelerate the demand for glioblastoma multiforme treatment. Thus, the increased incidence rates of glioblastoma multiforme are expected to boost the market's growth.
- Increasing research and development (r&d)
Increasing research and development (R&D) activities in molecular biotechnology and gene therapy for cancer and related diseases have facilitated the development of various biological drugs. These drugs assist in diminishing the side effects of the existing treatment methods, thus creating broader acceptance amongst patients. Tumor heterogeneity and variation in the patient-to-patient treatment approach are expected to increase the demand for a personalized treatment approach to manage glioblastoma multiforme. The approval of new treatments is expected to increase the life expectancy of patients living with glioblastoma multiforme. Furthermore, a special designation granted to investigational drugs by the FDA is expected to expedite novel therapy's approval process and commercialization. An increase in collaborations between researchers and market players is expected to boost the development of novel and effective treatment options for glioblastoma multiforme. The increasing approval for novel therapy and combination therapy is expected to drive theglioblastoma multiforme treatment market.
Opportunity
- Increasing drug approvals
The escalating demand for glioblastoma multiforme treatment should yield more regulatory approvals for associated drugs. Increasing regulatory endorsements for related drugs and recombinant products will constitute an increase in glioblastoma multiforme treatment market value in the coming years. In the framework of a Pan American Health Organization (PAHO) initiative to promote the recognition of Drug Regulatory Authorities, ANMAT´s evaluation process finished on December 11, 2009. The glioblastoma multiforme treatment industry has witnessed numerous drug approvals in recent years, driven by the increasing fatality rate of the disease. Increasing drug approvals will ramp up the glioblastoma multiforme treatment market demand.
Restraints/Challenges
High cost of glioblastoma multiforme treatment
The diagnostic tests for glioblastoma multiforme include highly technologically advanced products. Developing those products involves rigorous research and development by the developing player. Thus, the cost of the product remains high, which proportionally increases the cost of testing.
The diagnostic tools and techniques used for the diagnosis of glioblastoma multiforme include
radiation therapy, chemotherapy, among others. Early stages of GBM typically present with minimal or no symptoms; therefore, GBM is frequently diagnosed at advanced stages, resulting in a poor prognosis. Thus, the high cost of treating the glioblastoma multiforme using the advanced modalities and technology products will act as a major restraining factor for the growth of the global glioblastoma multiforme treatment market.
Recent Developments
- In April 2022, Elekta and GE Healthcare announced that they had signed a global commercial collaboration agreement in the field of radiation oncology, enabling them to provide hospitals a comprehensive offering across imaging and treatment for cancer patients requiring radiation therapy. This partnership will allow the companies to jointly promote solutions for each cancer center’s needs
- In July 2019, Amgen and Allergan plc announced that MVASI (bevacizumab-awwb), a biosimilar to Avastin (bevacizumab) is available in the United States (U.S.). This launch will enhance the sales of the product in the region
Glioblastoma Multiforme Treatment Market Scope
The glioblastoma multiforme treatment market is categorized into seven notable segments which are based on type, treatment, patient type, drug type, route of administration, end user and distribution channel. The growth among segments helps you analyse niche pockets of growth and strategies to approach the market and determine your core application areas and the difference in your target markets.
Type
- Primary (De Novo)
- Secondary
On the basis of type, the glioblastoma multiforme treatment market is segmented into primary (De Novo) and secondary.
Treatment
- Surgery
- Radiotherapy
- Medications
On the basis of treatment, the glioblastoma multiforme treatment market is segmented into surgery, radiotherapy and medications.
Patient Type
- Adult
- Geriatric
- Child
On the basis of patient type, the glioblastoma multiforme treatment market is segmented into adult, geriatric and child.
Drug Type
- Branded
- Generics
On the basis of drug type, the glioblastoma multiforme treatment market is segmented into generics and branded.
Route of Administration
- Oral
- Parenteral
- Others
On the basis of route of administration, the glioblastoma multiforme treatment market is segmented into parenteral, oral and others.
End User
- Hospitals
- Clinics
- Home Healthcare
- Others
On the basis of end user, the glioblastoma multiforme treatment market is segmented into hospitals, clinics, home healthcare and others.
Distribution Channel
- Hospital Pharmacy
- Retail Pharmacy
- Online Pharmacy
- Others
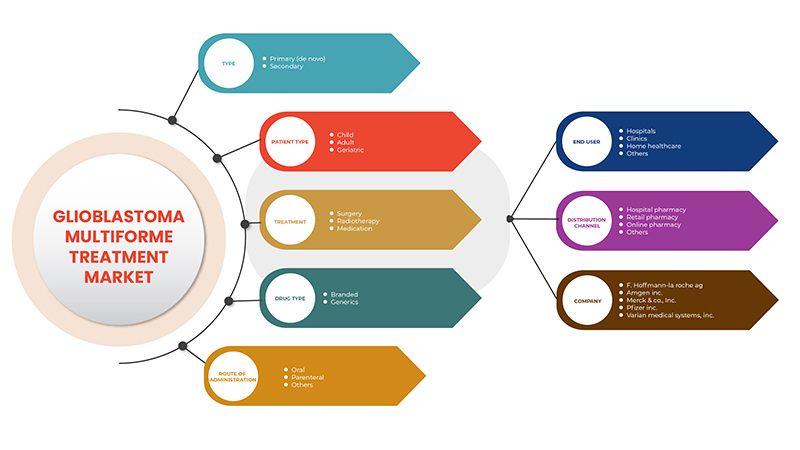
On the basis of distribution channel, the glioblastoma multiforme treatment market is segmented into hospital pharmacy, retail pharmacy and others.
Glioblastoma Multiforme Treatment Market Regional Analysis/Insights
The glioblastoma multiforme treatment market is analysed and market size insights and trends are provided by type, treatment, patient type, drug type, route of administration, end user and distribution channel as referenced above.
The regions covered in the glioblastoma multiforme treatment market report are China, Japan, India, South Korea, Singapore, Malaysia, Australia, Thailand, Indonesia, Philippines, Vietnam, Rest of Asia-Pacific.
Japan is expected to dominate the Asia-Pacific market due to the increased research and development activities in the country..
The country section of the report also provides individual market impacting factors and changes in market regulation that impact the current and future trends of the market. Data points such as down-stream and upstream value chain analysis, technical trends and porter's five forces analysis, case studies are some of the pointers used to forecast the market scenario for individual countries. Also, the presence and availability of global brands and their challenges faced due to large or scarce competition from local and domestic brands, impact of domestic tariffs and trade routes are considered while providing forecast analysis of the country data.
Competitive Landscape and Glioblastoma Multiforme Treatment Market Share Analysis
The glioblastoma multiforme treatment market competitive landscape provides details by the competitors. Details included are company overview, company financials, revenue generated, market potential, investment in research and development, new market initiatives, global presence, production sites and facilities, production capacities, company strengths and weaknesses, product launch, product width and breadth, and application dominance. The above data points provided are only related to the companies' focus on glioblastoma multiforme treatment market.
Some of the key players in the market are F. Hoffmann-La Roche AG, Amgen Inc., Merck & Co., Inc., Pfizer Inc., Varian Medical Systems, Inc. (A subsidiary of Siemens Healthcare), ZEISS International, Amneal Pharmaceuticals LLC, Elekta, Sun Pharmaceutical Industries Ltd, Teva Pharmaceutical Industries Ltd., Eckert & Ziegler, Accord Healthcare, Angiochem, ANI Pharmaceuticals, Inc., Arbor Pharmaceuticals, LLC. (A Subsidiary of Azurity Pharmaceuticals, Inc.), AstraZeneca, Cantex Pharmaceuticals, Inc., CELON LABS, Diffusion Pharmaceuticals Inc., EnGeneIC, ERC.SA., Genenta science, Jazz Pharmaceuticals, Inc., Loxo Oncology (A Subsidiary of Eli Lilly), Novartis AG, VBL THERAPEUTICS, Viatris Inc., and Zydus Pharmaceuticals, Inc., among others.
Research Methodology
Data collection and base year analysis are done using data collection modules with large sample sizes. The market data is analyzed and estimated using market statistical and coherent models. In addition, market share analysis and key trend analysis are the major success factors in the market report. The key research methodology used by DBMR research team is data triangulation which involves data mining, analysis of the impact of data variables on the market, and primary (industry expert) validation. Apart from this, data models include Vendor Positioning Grid, Market Time Line Analysis, Market Overview and Guide, Company Positioning Grid, Company Market Share Analysis, Standards of Measurement, Global Vs Regional and Vendor Share Analysis. Please request analyst call in case of further inquiry.
SKU-
Get online access to the report on the World's First Market Intelligence Cloud
- Interactive Data Analysis Dashboard
- Company Analysis Dashboard for high growth potential opportunities
- Research Analyst Access for customization & queries
- Competitor Analysis with Interactive dashboard
- Latest News, Updates & Trend analysis
- Harness the Power of Benchmark Analysis for Comprehensive Competitor Tracking
Table of Content
1 INTRODUCTION
1.1 OBJECTIVES OF THE STUDY
1.2 MARKET DEFINITION
1.3 OVERVIEW OF ASIA PACIFIC GLIOBLASTOMA MULTIFORME TREATMENT MARKET
1.4 LIMITATIONS
1.5 MARKETS COVERED
2 MARKET SEGMENTATION
2.1 MARKETS COVERED
2.2 GEOGRAPHICAL SCOPE
2.3 YEARS CONSIDERED FOR THE STUDY
2.4 CURRENCY AND PRICING
2.5 DBMR TRIPOD DATA VALIDATION MODEL
2.6 MULTIVARIATE MODELLING
2.7 TYPE LIFELINE CURVE
2.8 PRIMARY INTERVIEWS WITH KEY OPINION LEADERS
2.9 DBMR MARKET POSITION GRID
2.1 MARKET END USER COVERAGE GRID
2.11 VENDOR SHARE ANALYSIS
2.12 SECONDARY SOURCES
2.13 ASSUMPTIONS
3 EXECUTIVE SUMMARY
4 PREMIUM INSIGHTS
4.1 PESTEL
4.2 PORTER'S FIVE FORCES MODEL
5 EPIDEMIOLOGY
6 PIPELINE ANALYSIS
7 ASIA PACIFIC GLIOBLASTOMA MULTIFORME TREATMENT MARKET: REGULATORY SCENARIO
8 MARKET OVERVIEW
8.1 DRIVERS
8.1.1 GROWING PREVALENCE OF GLIOBLASTOMA MULTIFORME
8.1.2 INCREASING RESEARCH AND DEVELOPMENT (R&D)
8.1.3 PRESENCE OF A STRONG PIPELINE
8.1.4 GROWING GERIATRIC POPULATION
8.2 RESTRAINTS
8.2.1 HIGH COST OF GLIOBLASTOMA MULTIFORME TREATMENT
8.2.2 ADVERSE SIDE-EFFECTS OF GLIOBLASTOMA MULTIFORME TREATMENT
8.3 OPPORTUNITIES
8.3.1 INCREASING DRUG APPROVALS
8.3.2 PARTNERSHIP AND AGREEMENT BY MAJOR PLAYERS
8.3.3 INCREASING SUPPORT OF PRIVATE AND GOVERNMENT AGENCIES FOR TREATMENT
8.4 CHALLENGES
8.4.1 LACK OF NEW TREATMENT
8.4.2 ADVERSE EFFECTS AND RISKS ASSOCIATED WITH CANCER TREATMENT DRUGS
8.4.3 LACK OF EARLY DETECTION
9 ASIA PACIFIC GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TYPE
9.1 OVERVIEW
9.2 PRIMARY (DE NOVO)
9.3 SECONDARY
10 ASIA PACIFIC GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT
10.1 OVERVIEW
10.2 SURGERY
10.3 RADIOTHERAPY
10.3.1 BRACHYTHERAPY
10.3.2 FRACTIONATED STEREOTACTIC RT (FSRT)
10.3.3 CONFORMAL OR INTENSITY-MODULATED RT
10.3.4 RADIOSURGERY
10.4 MEDICATIONS
10.4.1 TEMOZOLOMIDE
10.4.1.1 ORAL
10.4.1.1 INTRAVENOUS
10.4.2 NITROSOUREAS DRUGS
10.4.2.1 CARMUSTINE
10.4.2.1.1 PARENTERAL
10.4.2.1.2 IMPLANTABLE WAFERS
10.4.2.2 LOMUSTINE
10.4.2.3 NIMUSTINE
10.4.2.4 FOTEMUSTINE
10.4.3 TARGETED THERAPY
10.4.3.1 BEVACIZUMAB
10.4.3.2 OTHERS
10.4.4 ANTI-EPILEPTICS
10.4.4.1 LEVETIRACETAM
10.4.4.2 PHENYTOIN
10.4.4.3 CARBAMAZEPINE
10.4.5 CORTICOSTEROIDS
10.4.5.1 METHYLPREDNISOLONE
10.4.5.2 PREDNISONE
10.4.5.3 OTHERS
11 ASIA PACIFIC GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY PATIENT TYPE
11.1 OVERVIEW
11.2 ADULT
11.2.1 MALE
11.2.2 FEMALE
11.3 GERIATRIC
11.3.1 MALE
11.3.2 FEMALE
11.4 CHILD
12 ASIA PACIFIC GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY DRUG TYPE
12.1 OVERVIEW
12.2 GENERICS
12.3 BRANDED
13 ASIA PACIFIC GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY ROUTE OF ADMINISTRATION
13.1 OVERVIEW
13.2 PARENTERAL
13.3 ORAL
13.3.1 CAPSULES
13.3.2 TABLETS
13.3.3 POWDERS
13.4 OTHERS
14 ASIA PACIFIC GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY END USER
14.1 OVERVIEW
14.2 HOSPITAL
14.3 CLINICS
14.4 HOME HEALTHCARE
14.5 OTHERS
15 ASIA PACIFIC GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY DISTRIBUTION CHANNEL
15.1 OVERVIEW
15.2 HOSPITAL PHARMACY
15.3 RETAIL PHARMACY
15.4 ONLINE PHARMACY
15.5 OTHERS
16 ASIA PACIFIC GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY REGION
16.1 ASIA-PACIFIC
16.1.1 JAPAN
16.1.2 CHINA
16.1.3 INDIA
16.1.4 AUSTRALIA
16.1.5 SOUTH KOREA
16.1.6 SINGAPORE
16.1.7 THAILAND
16.1.8 INDONESIA
16.1.9 PHILIPPINES
16.1.10 MALAYSIA
16.1.11 VIETNAM
16.1.12 REST OF ASIA-PACIFIC
17 ASIA PACIFIC GLIOBLASTOMA MULTIFORME TREATMENT MARKET: COMPANY LANDSCAPE
17.1 COMPANY SHARE ANALYSIS: ASIA PACIFIC
18 SWOT ANALYSIS
19 COMPANY PROFILE
19.1 F.HOFFMAN-LA ROCHE
19.1.1 COMPANY SNAPSHOT
19.1.2 REVENUE ANALYSIS
19.1.3 COMPANY SHARE ANALYSIS
19.1.4 PRODUCT PORTFOLIO
19.1.5 RECENT DEVELOPMENT
19.2 AMGEN INC.
19.2.1 COMPANY SNAPSHOT
19.2.2 REVENUE ANALYSIS
19.2.3 COMPANY SHARE ANALYSIS
19.2.4 PRODUCT PORTFOLIO
19.2.5 RECENT DEVELOPMENTS
19.2.5.1 PRODUCT APPROVAL
19.3 MERCK & CO., INC
19.3.1 COMPANY SNAPSHOT
19.3.2 REVENUE ANALYSIS
19.3.3 COMPANY SHARE ANALYSIS
19.3.4 PRODUCT PORTFOLIO
19.3.5 RECENT DEVELOPMENTS
19.3.5.1 STRATETIC COLLABORATION
19.3.5.2 EVENTS
19.4 PFIZER INC.
19.4.1 COMPANY SNAPSHOT
19.4.2 REVENUE ANALYSIS
19.4.3 COMPANY SHARE ANALYSIS
19.4.4 PRODUCT PORTFOLIO
19.4.5 RECENT DEVELOPMENT
19.4.5.1 MERGER
19.5 VARIAN MEDICAL SYSTEMS, INC. (A SUBSIDIARY OF SIEMENS HEALTHCARE)
19.5.1 COMPANY SNAPSHOT
19.5.2 REVENUE ANALYSIS
19.5.3 COMPANY SHARE ANALYSIS
19.5.4 PRODUCT PORTFOLIO
19.5.5 RECENT DEVELOPMENT
19.5.5.1 PARTNERSHIP
19.5.5.2 ACQUISITION
19.6 ZEISS INTERNATIONAL
19.6.1 COMPANY SNAPSHOT
19.6.2 REVENUE ANALYSIS
19.6.3 PRODUCT PORTFOLIO
19.6.4 RECENT DEVELOPMENTS
19.6.4.1 PRODUCT EXPANSION
19.7 AMNEAL PHARMACEUTICALS LLC
19.7.1 COMPANY SNAPSHOT
19.7.2 REVENUE ANALYSIS
19.7.3 PRODUCT PORTFOLIO
19.7.4 RECENT DEVELOPMENTS
19.7.4.1 EVENT
19.7.4.2 LAUNCH
19.7.4.3 ACQUISITION
19.8 ELEKTA
19.8.1 COMPANY SNAPSHOT
19.8.2 REVENUE ANALYSIS
19.8.3 PRODUCT PORTFOLIO
19.8.4 RECENT DEVELOPMENTS
19.8.4.1 PARTNERSHIP
19.9 SUN PHARMACEUTICAL INDUSTRIES LTD
19.9.1 COMPANY SNAPSHOT
19.9.2 REVENUE ANALYSIS
19.9.3 PRODUCT PORTFOLIO
19.9.4 RECENT DEVELOPMENT
19.9.4.1 AGREEMENT
19.1 TEVA PHARMACEUTICAL INDUSTRIES LTD
19.10.1 COMPANY SNAPSHOT
19.10.2 REVENUE ANALYSIS
19.10.3 PRODUCT PORTFOLIO
19.10.4 RECENT DEVELOPMENT
19.11 ECKERT & ZIEGLER
19.11.1 COMPANY SNAPSHOT
19.11.2 REVENUE ANALYSIS
19.11.3 PRODUCT PORTFOLIO
19.11.4 RECENT DEVELOPMENT
19.12 ACCORD HEALTHCARE
19.12.1 COMPANY SNAPSHOT
19.12.2 PRODUCT PORTFOLIO
19.12.3 RECENT DEVELOPMENT
19.13 ANGIOCHEM
19.13.1 COMPANY SNAPSHOT
19.13.2 PRODUCT PORTFOLIO
19.13.3 RECENT DEVELOPMENT
19.13.3.1 AGREMEENT
19.14 ANI PHARMACEUTICALS, INC.
19.14.1 COMPANY SNAPSHOT
19.14.2 REVENUE ANALYSIS
19.14.3 PRODUCT PORTFOLIO
19.14.4 RECENT DEVELOPMENTS
19.14.4.1 ACQUISITION
19.15 ARBOR PHARMACEUTICALS, LLC. A SUBSIDIARY OF AZURITY PHARMACEUTICALS, INC.
19.15.1 COMPANY SNAPSHOT
19.15.2 PRODUCT PORTFOLIO
19.15.3 RECENT DEVELOPMENT
19.15.3.1 ACQUISITION
19.15.3.2 PRODUCT APPROVAL
19.16 ASTRAZENECA
19.16.1 COMPANY SNAPSHOT
19.16.2 REVENUE ANALYSIS
19.16.3 PRODUCT PORTFOLIO
19.16.4 RECENT DEVELOPMENT
19.16.4.1 AGREEMENT
19.17 CANTEX PHARMACEUTICALS, INC.
19.17.1 COMPANY SNAPSHOT
19.17.2 PRODUCT PORTFOLIO
19.17.3 RECENT DEVELOPMENT
19.18 CELON LABS
19.18.1 COMPANY SNAPSHOT
19.18.2 PRODUCT PORTFOLIO
19.18.3 RECENT DEVELOPMENT
19.19 DIFFUSION PHARMACEUTICAL
19.19.1 COMPANY SNAPSHOT
19.19.2 SERVICES PORTFOLIO
19.19.3 RECENT DEVELOPMENT
19.2 ERC.SA
19.20.1 COMPANY SNAPSHOT
19.20.2 PRODUCT PORTFOLIO
19.20.3 RECENT DEVELOPMENT
19.20.3.1 PIPELINE UPDATE
19.21 ENGENEIC
19.21.1 COMPANY SNAPSHOT
19.21.2 PRODUCT PORTFOLIO
19.21.3 RECENT DEVELOPMENTS
19.21.3.1 AWARDS
19.22 GENENTA SCIENCE
19.22.1 COMPANY SNAPSHOT
19.22.2 PRODUCT PORTFOLIO
19.22.3 RECENT DEVELOPMENT
19.22.3.1 EVENT
19.23 JAZZ PHARMACEUTICALS, INC.
19.23.1 COMPANY SNAPSHOT
19.23.2 REVENUE ANALYSIS
19.23.3 PRODUCT PORTFOLIO
19.23.4 RECENT DEVELOPMENT
19.23.4.1 ACQUISITION
19.24 LOXO ONCOLOGY (A SUBSIDIARY OF ELI LILLY)
19.24.1 COMPANY SNAPSHOT
19.24.2 PRODUCT PORTFOLIO
19.24.3 RECENT DEVELOPMENT
19.25 NOVARTIS AG
19.25.1 COMPANY SNAPSHOT
19.25.2 REVENUE ANALYSIS
19.25.3 PRODUCT PORTFOLIO
19.25.4 RECENT DEVELOPMENT
19.26 VBL THERAPEUTICS
19.26.1 COMPANY SNAPSHOT
19.26.2 PRODUCT PORTFOLIO
19.26.3 RECENT DEVELOPMENT
19.26.3.1 EVENT
19.26.3.2 AWARD
19.27 VIATRIS INC
19.27.1 COMPANY SNAPSHOT
19.27.2 REVENUE ANALYSIS
19.27.3 PRODUCT PORTFOLIO
19.27.4 RECENT DEVELOPMENT
19.27.4.1 AGREEMENT
19.28 ZYDUS PHARMACEUTICALS, INC.
19.28.1 COMPANY SNAPSHOT
19.28.2 PRODUCT PORTFOLIO
19.28.3 RECENT DEVELOPMENTS
20 QUESTIONNAIRE
21 RELATED REPORTS
List of Table
TABLE 1 PIPELINE ANALYSIS FOR GLIOBLASTOMA MULTIFORME TREATMENT MARKET
TABLE 2 ASIA PACIFIC GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TYPE, 2020-2029 (USD MILLION)
TABLE 3 ASIA PACIFIC PRIMARY (DE NOVO) IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 4 ASIA PACIFIC SECONDARY IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 5 ASIA PACIFIC GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 6 ASIA PACIFIC SURGERY IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 7 ASIA PACIFIC RADIOTHERAPY IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 8 ASIA PACIFIC RADIOTHERAPY IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 9 ASIA PACIFIC MEDICATIONS IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 10 ASIA PACIFIC MEDICATIONS IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 11 ASIA PACIFIC TEMOZOLOMIDE IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 12 ASIA PACIFIC NITROSOUREAS DRUGS IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 13 ASIA PACIFIC CARMUSTINE IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 14 ASIA PACIFIC TARGETED THERAPY IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 15 ASIA PACIFIC ANTI-EPILEPTICS IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 16 ASIA PACIFIC CORTICOSTEROIDS IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 17 ASIA PACIFIC GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY PATIENT TYPE, 2020-2029 (USD MILLION)
TABLE 18 ASIA PACIFIC ADULTS IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 19 ASIA PACIFIC ADULT IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY PATIENT TYPE, 2020-2029 (USD MILLION)
TABLE 20 ASIA PACIFIC GERIATRIC IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 21 ASIA PACIFIC GERIATRIC IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY PATIENT TYPE, 2020-2029 (USD MILLION)
TABLE 22 ASIA PACIFIC CHILD IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 23 ASIA PACIFIC GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY DRUG TYPE, 2020-2029 (USD MILLION)
TABLE 24 ASIA PACIFIC GENERICS IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 25 ASIA PACIFIC BRANDED IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 26 ASIA PACIFIC GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY ROUTE OF ADMINISTRATION, 2020-2029 (USD MILLION)
TABLE 27 ASIA PACIFIC PARENTERAL IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 28 ASIA PACIFIC ORAL IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 29 ASIA PACIFIC ORAL IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY ROUTE OF ADMINISTRATION, 2020-2029 (USD MILLION)
TABLE 30 ASIA PACIFIC OTHERS IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 31 ASIA PACIFIC GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY END USER, 2020-2029 (USD MILLION)
TABLE 32 ASIA PACIFIC HOSPITAL IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 33 ASIA PACIFIC CLINICS IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 34 ASIA PACIFIC HOME HEALTHCARE IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 35 ASIA PACIFIC OTHERS IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 36 ASIA PACIFIC GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY DISTRIBUTION CHANNEL, 2020-2029 (USD MILLION)
TABLE 37 ASIA PACIFIC HOSPITAL PHARMACY IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 38 ASIA PACIFIC RETAIL PHARMACY IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 39 ASIA PACIFIC ONLINE PHARMACY IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 40 ASIA PACIFIC OTHERS IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 41 ASIA-PACIFIC GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY COUNTRY, 2020-2029 (USD MILLION)
TABLE 42 ASIA-PACIFIC GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TYPE, 2020-2029 (USD MILLION)
TABLE 43 ASIA-PACIFIC GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 44 ASIA-PACIFIC RADIOTHERAPY IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 45 ASIA-PACIFIC MEDICATIONS IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 46 ASIA-PACIFIC TEMOZOLOMIDE IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 47 ASIA-PACIFIC NITROSOUREAS DRUGS IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 48 ASIA-PACIFIC CARMUSTINE IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 49 ASIA-PACIFIC TARGETED THERAPY IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 50 ASIA-PACIFIC ANTI-EPILEPTICS IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 51 ASIA-PACIFIC CORTICOSTEROIDS IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 52 ASIA-PACIFIC GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY PATIENT TYPE, 2020-2029 (USD MILLION)
TABLE 53 ASIA-PACIFIC ADULT IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY PATIENT TYPE, 2020-2029 (USD MILLION)
TABLE 54 ASIA-PACIFIC GERIATRIC IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY PATIENT TYPE, 2020-2029 (USD MILLION)
TABLE 55 ASIA-PACIFIC GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY DRUG TYPE, 2020-2029 (USD MILLION)
TABLE 56 ASIA-PACIFIC GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY ROUTE OF ADMINISTRATION, 2020-2029 (USD MILLION)
TABLE 57 ASIA-PACIFIC ORAL IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY ROUTE OF ADMINISTRATION, 2020-2029 (USD MILLION)
TABLE 58 ASIA-PACIFIC GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY END USER, 2020-2029 (USD MILLION)
TABLE 59 ASIA-PACIFIC GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY DISTRIBUTION CHANNEL, 2020-2029 (USD MILLION)
TABLE 60 JAPAN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TYPE, 2020-2029 (USD MILLION)
TABLE 61 JAPAN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 62 JAPAN RADIOTHERAPY IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 63 JAPAN MEDICATIONS IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 64 JAPAN TEMOZOLOMIDE IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 65 JAPAN NITROSOUREAS DRUGS IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 66 JAPAN CARMUSTINE IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 67 JAPAN TARGETED THERAPY IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 68 JAPAN ANTI-EPILEPTICS IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 69 JAPAN CORTICOSTEROIDS IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 70 JAPAN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY PATIENT TYPE, 2020-2029 (USD MILLION)
TABLE 71 JAPAN ADULT IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY PATIENT TYPE, 2020-2029 (USD MILLION)
TABLE 72 JAPAN GERIATRIC IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY PATIENT TYPE, 2020-2029 (USD MILLION)
TABLE 73 JAPAN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY DRUG TYPE, 2020-2029 (USD MILLION)
TABLE 74 JAPAN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY ROUTE OF ADMINISTRATION, 2020-2029 (USD MILLION)
TABLE 75 JAPAN ORAL IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY ROUTE OF ADMINISTRATION, 2020-2029 (USD MILLION)
TABLE 76 JAPAN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY END USER, 2020-2029 (USD MILLION)
TABLE 77 JAPAN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY DISTRIBUTION CHANNEL, 2020-2029 (USD MILLION)
TABLE 78 CHINA GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TYPE, 2020-2029 (USD MILLION)
TABLE 79 CHINA GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 80 CHINA RADIOTHERAPY IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 81 CHINA MEDICATIONS IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 82 CHINA TEMOZOLOMIDE IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 83 CHINA NITROSOUREAS DRUGS IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 84 CHINA CARMUSTINE IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 85 CHINA TARGETED THERAPY IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 86 CHINA ANTI-EPILEPTICS IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 87 CHINA CORTICOSTEROIDS IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 88 CHINA GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY PATIENT TYPE, 2020-2029 (USD MILLION)
TABLE 89 CHINA ADULT IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY PATIENT TYPE, 2020-2029 (USD MILLION)
TABLE 90 CHINA GERIATRIC IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY PATIENT TYPE, 2020-2029 (USD MILLION)
TABLE 91 CHINA GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY DRUG TYPE, 2020-2029 (USD MILLION)
TABLE 92 CHINA GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY ROUTE OF ADMINISTRATION, 2020-2029 (USD MILLION)
TABLE 93 CHINA ORAL IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY ROUTE OF ADMINISTRATION, 2020-2029 (USD MILLION)
TABLE 94 CHINA GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY END USER, 2020-2029 (USD MILLION)
TABLE 95 CHINA GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY DISTRIBUTION CHANNEL, 2020-2029 (USD MILLION)
TABLE 96 INDIA GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TYPE, 2020-2029 (USD MILLION)
TABLE 97 INDIA GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 98 INDIA RADIOTHERAPY IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 99 INDIA MEDICATIONS IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 100 INDIA TEMOZOLOMIDE IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 101 INDIA NITROSOUREAS DRUGS IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 102 INDIA CARMUSTINE IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 103 INDIA TARGETED THERAPY IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 104 INDIA ANTI-EPILEPTICS IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 105 INDIA CORTICOSTEROIDS IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 106 INDIA GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY PATIENT TYPE, 2020-2029 (USD MILLION)
TABLE 107 INDIA ADULT IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY PATIENT TYPE, 2020-2029 (USD MILLION)
TABLE 108 INDIA GERIATRIC IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY PATIENT TYPE, 2020-2029 (USD MILLION)
TABLE 109 INDIA GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY DRUG TYPE, 2020-2029 (USD MILLION)
TABLE 110 INDIA GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY ROUTE OF ADMINISTRATION, 2020-2029 (USD MILLION)
TABLE 111 INDIA ORAL IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY ROUTE OF ADMINISTRATION, 2020-2029 (USD MILLION)
TABLE 112 INDIA GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY END USER, 2020-2029 (USD MILLION)
TABLE 113 INDIA GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY DISTRIBUTION CHANNEL, 2020-2029 (USD MILLION)
TABLE 114 AUSTRALIA GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TYPE, 2020-2029 (USD MILLION)
TABLE 115 AUSTRALIA GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 116 AUSTRALIA RADIOTHERAPY IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 117 AUSTRALIA MEDICATIONS IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 118 AUSTRALIA TEMOZOLOMIDE IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 119 AUSTRALIA NITROSOUREAS DRUGS IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 120 AUSTRALIA CARMUSTINE IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 121 AUSTRALIA TARGETED THERAPY IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 122 AUSTRALIA ANTI-EPILEPTICS IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 123 AUSTRALIA CORTICOSTEROIDS IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 124 AUSTRALIA GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY PATIENT TYPE, 2020-2029 (USD MILLION)
TABLE 125 AUSTRALIA ADULT IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY PATIENT TYPE, 2020-2029 (USD MILLION)
TABLE 126 AUSTRALIA GERIATRIC IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY PATIENT TYPE, 2020-2029 (USD MILLION)
TABLE 127 AUSTRALIA GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY DRUG TYPE, 2020-2029 (USD MILLION)
TABLE 128 AUSTRALIA GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY ROUTE OF ADMINISTRATION, 2020-2029 (USD MILLION)
TABLE 129 AUSTRALIA ORAL IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY ROUTE OF ADMINISTRATION, 2020-2029 (USD MILLION)
TABLE 130 AUSTRALIA GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY END USER, 2020-2029 (USD MILLION)
TABLE 131 AUSTRALIA GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY DISTRIBUTION CHANNEL, 2020-2029 (USD MILLION)
TABLE 132 SOUTH KOREA GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TYPE, 2020-2029 (USD MILLION)
TABLE 133 SOUTH KOREA GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 134 SOUTH KOREA RADIOTHERAPY IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 135 SOUTH KOREA MEDICATIONS IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 136 SOUTH KOREA TEMOZOLOMIDE IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 137 SOUTH KOREA NITROSOUREAS DRUGS IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 138 SOUTH KOREA CARMUSTINE IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 139 SOUTH KOREA TARGETED THERAPY IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 140 SOUTH KOREA ANTI-EPILEPTICS IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 141 SOUTH KOREA CORTICOSTEROIDS IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 142 SOUTH KOREA GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY PATIENT TYPE, 2020-2029 (USD MILLION)
TABLE 143 SOUTH KOREA ADULT IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY PATIENT TYPE, 2020-2029 (USD MILLION)
TABLE 144 SOUTH KOREA GERIATRIC IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY PATIENT TYPE, 2020-2029 (USD MILLION)
TABLE 145 SOUTH KOREA GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY DRUG TYPE, 2020-2029 (USD MILLION)
TABLE 146 SOUTH KOREA GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY ROUTE OF ADMINISTRATION, 2020-2029 (USD MILLION)
TABLE 147 SOUTH KOREA ORAL IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY ROUTE OF ADMINISTRATION, 2020-2029 (USD MILLION)
TABLE 148 SOUTH KOREA GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY END USER, 2020-2029 (USD MILLION)
TABLE 149 SOUTH KOREA GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY DISTRIBUTION CHANNEL, 2020-2029 (USD MILLION)
TABLE 150 SINGAPORE GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TYPE, 2020-2029 (USD MILLION)
TABLE 151 SINGAPORE GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 152 SINGAPORE RADIOTHERAPY IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 153 SINGAPORE MEDICATIONS IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 154 SINGAPORE TEMOZOLOMIDE IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 155 SINGAPORE NITROSOUREAS DRUGS IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 156 SINGAPORE CARMUSTINE IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 157 SINGAPORE TARGETED THERAPY IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 158 SINGAPORE ANTI-EPILEPTICS IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 159 SINGAPORE CORTICOSTEROIDS IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 160 SINGAPORE GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY PATIENT TYPE, 2020-2029 (USD MILLION)
TABLE 161 SINGAPORE ADULT IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY PATIENT TYPE, 2020-2029 (USD MILLION)
TABLE 162 SINGAPORE GERIATRIC IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY PATIENT TYPE, 2020-2029 (USD MILLION)
TABLE 163 SINGAPORE GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY DRUG TYPE, 2020-2029 (USD MILLION)
TABLE 164 SINGAPORE GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY ROUTE OF ADMINISTRATION, 2020-2029 (USD MILLION)
TABLE 165 SINGAPORE ORAL IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY ROUTE OF ADMINISTRATION, 2020-2029 (USD MILLION)
TABLE 166 SINGAPORE GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY END USER, 2020-2029 (USD MILLION)
TABLE 167 SINGAPORE GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY DISTRIBUTION CHANNEL, 2020-2029 (USD MILLION)
TABLE 168 THAILAND GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TYPE, 2020-2029 (USD MILLION)
TABLE 169 THAILAND GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 170 THAILAND RADIOTHERAPY IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 171 THAILAND MEDICATIONS IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 172 THAILAND TEMOZOLOMIDE IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 173 THAILAND NITROSOUREAS DRUGS IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 174 THAILAND CARMUSTINE IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 175 THAILAND TARGETED THERAPY IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 176 THAILAND ANTI-EPILEPTICS IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 177 THAILAND CORTICOSTEROIDS IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 178 THAILAND GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY PATIENT TYPE, 2020-2029 (USD MILLION)
TABLE 179 THAILAND ADULT IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY PATIENT TYPE, 2020-2029 (USD MILLION)
TABLE 180 THAILAND GERIATRIC IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY PATIENT TYPE, 2020-2029 (USD MILLION)
TABLE 181 THAILAND GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY DRUG TYPE, 2020-2029 (USD MILLION)
TABLE 182 THAILAND GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY ROUTE OF ADMINISTRATION, 2020-2029 (USD MILLION)
TABLE 183 THAILAND ORAL IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY ROUTE OF ADMINISTRATION, 2020-2029 (USD MILLION)
TABLE 184 THAILAND GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY END USER, 2020-2029 (USD MILLION)
TABLE 185 THAILAND GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY DISTRIBUTION CHANNEL, 2020-2029 (USD MILLION)
TABLE 186 INDONESIA GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TYPE, 2020-2029 (USD MILLION)
TABLE 187 INDONESIA GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 188 INDONESIA RADIOTHERAPY IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 189 INDONESIA MEDICATIONS IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 190 INDONESIA TEMOZOLOMIDE IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 191 INDONESIA NITROSOUREAS DRUGS IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 192 INDONESIA CARMUSTINE IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 193 INDONESIA TARGETED THERAPY IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 194 INDONESIA ANTI-EPILEPTICS IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 195 INDONESIA CORTICOSTEROIDS IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 196 INDONESIA GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY PATIENT TYPE, 2020-2029 (USD MILLION)
TABLE 197 INDONESIA ADULT IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY PATIENT TYPE, 2020-2029 (USD MILLION)
TABLE 198 INDONESIA GERIATRIC IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY PATIENT TYPE, 2020-2029 (USD MILLION)
TABLE 199 INDONESIA GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY DRUG TYPE, 2020-2029 (USD MILLION)
TABLE 200 INDONESIA GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY ROUTE OF ADMINISTRATION, 2020-2029 (USD MILLION)
TABLE 201 INDONESIA ORAL IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY ROUTE OF ADMINISTRATION, 2020-2029 (USD MILLION)
TABLE 202 INDONESIA GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY END USER, 2020-2029 (USD MILLION)
TABLE 203 INDONESIA GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY DISTRIBUTION CHANNEL, 2020-2029 (USD MILLION)
TABLE 204 PHILIPPINES GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TYPE, 2020-2029 (USD MILLION)
TABLE 205 PHILIPPINES GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 206 PHILIPPINES RADIOTHERAPY IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 207 PHILIPPINES MEDICATIONS IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 208 PHILIPPINES TEMOZOLOMIDE IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 209 PHILIPPINES NITROSOUREAS DRUGS IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 210 PHILIPPINES CARMUSTINE IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 211 PHILIPPINES TARGETED THERAPY IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 212 PHILIPPINES ANTI-EPILEPTICS IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 213 PHILIPPINES CORTICOSTEROIDS IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 214 PHILIPPINES GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY PATIENT TYPE, 2020-2029 (USD MILLION)
TABLE 215 PHILIPPINES ADULT IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY PATIENT TYPE, 2020-2029 (USD MILLION)
TABLE 216 PHILIPPINES GERIATRIC IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY PATIENT TYPE, 2020-2029 (USD MILLION)
TABLE 217 PHILIPPINES GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY DRUG TYPE, 2020-2029 (USD MILLION)
TABLE 218 PHILIPPINES GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY ROUTE OF ADMINISTRATION, 2020-2029 (USD MILLION)
TABLE 219 PHILIPPINES ORAL IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY ROUTE OF ADMINISTRATION, 2020-2029 (USD MILLION)
TABLE 220 PHILIPPINES GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY END USER, 2020-2029 (USD MILLION)
TABLE 221 PHILIPPINES GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY DISTRIBUTION CHANNEL, 2020-2029 (USD MILLION)
TABLE 222 MALAYSIA GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TYPE, 2020-2029 (USD MILLION)
TABLE 223 MALAYSIA GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 224 MALAYSIA RADIOTHERAPY IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 225 MALAYSIA MEDICATIONS IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 226 MALAYSIA TEMOZOLOMIDE IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 227 MALAYSIA NITROSOUREAS DRUGS IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 228 MALAYSIA CARMUSTINE IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 229 MALAYSIA TARGETED THERAPY IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 230 MALAYSIA ANTI-EPILEPTICS IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 231 MALAYSIA CORTICOSTEROIDS IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 232 MALAYSIA GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY PATIENT TYPE, 2020-2029 (USD MILLION)
TABLE 233 MALAYSIA ADULT IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY PATIENT TYPE, 2020-2029 (USD MILLION)
TABLE 234 MALAYSIA GERIATRIC IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY PATIENT TYPE, 2020-2029 (USD MILLION)
TABLE 235 MALAYSIA GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY DRUG TYPE, 2020-2029 (USD MILLION)
TABLE 236 MALAYSIA GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY ROUTE OF ADMINISTRATION, 2020-2029 (USD MILLION)
TABLE 237 MALAYSIA ORAL IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY ROUTE OF ADMINISTRATION, 2020-2029 (USD MILLION)
TABLE 238 MALAYSIA GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY END USER, 2020-2029 (USD MILLION)
TABLE 239 MALAYSIA GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY DISTRIBUTION CHANNEL, 2020-2029 (USD MILLION)
TABLE 240 VIETNAM GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TYPE, 2020-2029 (USD MILLION)
TABLE 241 VIETNAM GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 242 VIETNAM RADIOTHERAPY IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 243 VIETNAM MEDICATIONS IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 244 VIETNAM TEMOZOLOMIDE IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 245 VIETNAM NITROSOUREAS DRUGS IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 246 VIETNAM CARMUSTINE IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 247 VIETNAM TARGETED THERAPY IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 248 VIETNAM ANTI-EPILEPTICS IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 249 VIETNAM CORTICOSTEROIDS IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 250 VIETNAM GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY PATIENT TYPE, 2020-2029 (USD MILLION)
TABLE 251 VIETNAM ADULT IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY PATIENT TYPE, 2020-2029 (USD MILLION)
TABLE 252 VIETNAM GERIATRIC IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY PATIENT TYPE, 2020-2029 (USD MILLION)
TABLE 253 VIETNAM GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY DRUG TYPE, 2020-2029 (USD MILLION)
TABLE 254 VIETNAM GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY ROUTE OF ADMINISTRATION, 2020-2029 (USD MILLION)
TABLE 255 VIETNAM ORAL IN GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY ROUTE OF ADMINISTRATION, 2020-2029 (USD MILLION)
TABLE 256 VIETNAM GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY END USER, 2020-2029 (USD MILLION)
TABLE 257 VIETNAM GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY DISTRIBUTION CHANNEL, 2020-2029 (USD MILLION)
TABLE 258 REST OF ASIA-PACIFIC GLIOBLASTOMA MULTIFORME TREATMENT MARKET, BY TYPE, 2020-2029 (USD MILLION)
List of Figure
FIGURE 1 ASIA PACIFIC GLIOBLASTOMA MULTIFORME TREATMENT MARKET: SEGMENTATION
FIGURE 2 ASIA PACIFIC GLIOBLASTOMA MULTIFORME TREATMENT MARKET : DATA TRIANGULATION
FIGURE 3 ASIA PACIFIC GLIOBLASTOMA MULTIFORME TREATMENT MARKET: DROC ANALYSIS
FIGURE 4 ASIA PACIFIC GLIOBLASTOMA MULTIFORME TREATMENT MARKET: ASIA PACIFIC VS REGIONAL MARKET ANALYSIS
FIGURE 5 ASIA PACIFIC GLIOBLASTOMA MULTIFORME TREATMENT MARKET: COMPANY RESEARCH ANALYSIS
FIGURE 6 ASIA PACIFIC GLIOBLASTOMA MULTIFORME TREATMENT MARKET: MULTIVARIATE MODELLING
FIGURE 7 ASIA PACIFIC GLIOBLASTOMA MULTIFORME TREATMENT MARKET: INTERVIEW DEMOGRAPHICS
FIGURE 8 ASIA PACIFIC GLIOBLASTOMA MULTIFORME TREATMENT MARKET: DBMR MARKET POSITION GRID
FIGURE 9 ASIA PACIFIC GLIOBLASTOMA MULTIFORME TREATMENT MARKET: MARKET END USER COVERAGE GRID
FIGURE 10 ASIA PACIFIC GLIOBLASTOMA MULTIFORME TREATMENT MARKET: VENDOR SHARE ANALYSIS
FIGURE 11 ASIA PACIFIC GLIOBLASTOMA MULTIFORME TREATMENT MARKET: SEGMENTATION
FIGURE 12 NORTH AMERICA IS EXPECTED TO DOMINATE AND ASIA-PACIFIC IS GROWING AT THE FASTEST PACE IN ASIA PACIFIC GLIOBLASTOMA MULTIFORME TREATMENT MARKET IN THE FORECAST PERIOD OF 2022 TO 2029
FIGURE 13 INCREASE IN THE PREVALENCE OF GLIOBLASTOMA MULTIFORME AND INCREASE IN PIPELINE PRODUCTS ARE DRIVING THE ASIA PACIFIC GLIOBLASTOMA MULTIFORME TREATMENT MARKET IN THE FORECAST PERIOD OF 2022 TO 2029
FIGURE 14 PRIMARY (DE NOVO) SEGMENT IS EXPECTED TO ACCOUNT FOR THE LARGEST SHARE OF THE ASIA PACIFIC GLIOBLASTOMA MULTIFORME TREATMENT MARKET IN 2022 & 2029
FIGURE 15 DRIVERS, RESTRAINTS, OPPORTUNITIES, AND CHALLENGES OF THE ASIA PACIFIC GLIOBLASTOMA MULTIFORME TREATMENT MARKET
FIGURE 16 ASIA PACIFIC GLIOBLASTOMA MULTIFORME TREATMENT MARKET: BY TYPE, 2021
FIGURE 17 ASIA PACIFIC GLIOBLASTOMA MULTIFORME TREATMENT MARKET: BY TYPE, 2022-2029 (USD MILLION)
FIGURE 18 ASIA PACIFIC GLIOBLASTOMA MULTIFORME TREATMENT MARKET: BY TYPE, CAGR (2022-2029)
FIGURE 19 ASIA PACIFIC GLIOBLASTOMA MULTIFORME TREATMENT MARKET: BY TYPE, LIFELINE CURVE
FIGURE 20 ASIA PACIFIC GLIOBLASTOMA MULTIFORME TREATMENT MARKET: BY TREATMENT, 2021
FIGURE 21 ASIA PACIFIC GLIOBLASTOMA MULTIFORME TREATMENT MARKET: BY TREATMENT, 2022-2029 (USD MILLION)
FIGURE 22 ASIA PACIFIC GLIOBLASTOMA MULTIFORME TREATMENT: BY TREATMENT, CAGR (2022-2029)
FIGURE 23 ASIA PACIFIC GLIOBLASTOMA MULTIFORME TREATMENT: BY TREATMENT, LIFELINE CURVE
FIGURE 24 ASIA PACIFIC GLIOBLASTOMA MULTIFORME TREATMENT MARKET: BY PATIENT TYPE, 2021
FIGURE 25 ASIA PACIFIC GLIOBLASTOMA MULTIFORME TREATMENT MARKET: BY PATIENT TYPE, 2022-2029 (USD MILLION)
FIGURE 26 ASIA PACIFIC GLIOBLASTOMA MULTIFORME TREATMENT MARKET: BY PATIENT TYPE, CAGR (2022-2029)
FIGURE 27 ASIA PACIFIC GLIOBLASTOMA MULTIFORME TREATMENT MARKET: BY PATIENT TYPE, LIFELINE CURVE
FIGURE 28 ASIA PACIFIC GLIOBLASTOMA MULTIFORME TREATMENT MARKET: BY DRUG TYPE, 2021
FIGURE 29 ASIA PACIFIC GLIOBLASTOMA MULTIFORME TREATMENT MARKET: BY DRUG TYPE, 2022-2029 (USD MILLION)
FIGURE 30 ASIA PACIFIC GLIOBLASTOMA MULTIFORME TREATMENT MARKET: BY DRUG TYPE, CAGR (2022-2029)
FIGURE 31 ASIA PACIFIC GLIOBLASTOMA MULTIFORME TREATMENT MARKET: BY DRUG TYPE, LIFELINE CURVE
FIGURE 32 ASIA PACIFIC GLIOBLASTOMA MULTIFORME TREATMENT MARKET: BY ROUTE OF ADMINISTRATION, 2021
FIGURE 33 ASIA PACIFIC GLIOBLASTOMA MULTIFORME TREATMENT MARKET: BY ROUTE OF ADMINISTRATION, 2022-2029 (USD MILLION)
FIGURE 34 ASIA PACIFIC GLIOBLASTOMA MULTIFORME TREATMENT MARKET: BY ROUTE OF ADMINISTRATION, CAGR (2022-2029)
FIGURE 35 ASIA PACIFIC GLIOBLASTOMA MULTIFORME TREATMENT MARKET: BY ROUTE OF ADMINISTRATION, LIFELINE CURVE
FIGURE 36 ASIA PACIFIC GLIOBLASTOMA MULTIFORME TREATMENT MARKET: BY END USER, 2021
FIGURE 37 ASIA PACIFIC GLIOBLASTOMA MULTIFORME TREATMENT MARKET: BY END USER, 2022-2029 (USD MILLION)
FIGURE 38 ASIA PACIFIC GLIOBLASTOMA MULTIFORME TREATMENT MARKET: BY END USER, CAGR (2022-2029)
FIGURE 39 ASIA PACIFIC GLIOBLASTOMA MULTIFORME TREATMENT MARKET: BY END USER, LIFELINE CURVE
FIGURE 40 ASIA PACIFIC GLIOBLASTOMA MULTIFORME TREATMENT MARKET: BY DISTRIBUTION CHANNEL, 2021
FIGURE 41 ASIA PACIFIC GLIOBLASTOMA MULTIFORME TREATMENT MARKET: BY DISTRIBUTION CHANNEL, 2022-2029 (USD MILLION)
FIGURE 42 ASIA PACIFIC GLIOBLASTOMA MULTIFORME TREATMENT MARKET: BY DISTRIBUTION CHANNEL, CAGR (2022-2029)
FIGURE 43 ASIA PACIFIC GLIOBLASTOMA MULTIFORME TREATMENT MARKET: BY DISTRIBUTION CHANNEL, LIFELINE CURVE
FIGURE 44 ASIA-PACIFIC GLIOBLASTOMA MULTIFORME TREATMENT MARKET: SNAPSHOT (2022)
FIGURE 45 ASIA-PACIFIC GLIOBLASTOMA MULTIFORME TREATMENT MARKET: BY COUNTRY (2022)
FIGURE 46 ASIA-PACIFIC GLIOBLASTOMA MULTIFORME TREATMENT MARKET: BY COUNTRY (2022 & 2029)
FIGURE 47 ASIA-PACIFIC GLIOBLASTOMA MULTIFORME TREATMENT MARKET: BY COUNTRY (2021 & 2029)
FIGURE 48 ASIA-PACIFIC GLIOBLASTOMA MULTIFORME TREATMENT MARKET: BY TYPE (2022 & 2029)
FIGURE 49 ASIA PACIFIC GLIOBLASTOMA MULTIFORME TREATMENT MARKET: COMPANY SHARE 2021 (%)

Research Methodology
Data collection and base year analysis are done using data collection modules with large sample sizes. The stage includes obtaining market information or related data through various sources and strategies. It includes examining and planning all the data acquired from the past in advance. It likewise envelops the examination of information inconsistencies seen across different information sources. The market data is analysed and estimated using market statistical and coherent models. Also, market share analysis and key trend analysis are the major success factors in the market report. To know more, please request an analyst call or drop down your inquiry.
The key research methodology used by DBMR research team is data triangulation which involves data mining, analysis of the impact of data variables on the market and primary (industry expert) validation. Data models include Vendor Positioning Grid, Market Time Line Analysis, Market Overview and Guide, Company Positioning Grid, Patent Analysis, Pricing Analysis, Company Market Share Analysis, Standards of Measurement, Global versus Regional and Vendor Share Analysis. To know more about the research methodology, drop in an inquiry to speak to our industry experts.
Customization Available
Data Bridge Market Research is a leader in advanced formative research. We take pride in servicing our existing and new customers with data and analysis that match and suits their goal. The report can be customized to include price trend analysis of target brands understanding the market for additional countries (ask for the list of countries), clinical trial results data, literature review, refurbished market and product base analysis. Market analysis of target competitors can be analyzed from technology-based analysis to market portfolio strategies. We can add as many competitors that you require data about in the format and data style you are looking for. Our team of analysts can also provide you data in crude raw excel files pivot tables (Fact book) or can assist you in creating presentations from the data sets available in the report.