Europe Breast Reconstruction Market
Market Size in USD Million
CAGR :
% 
 USD
679.14 Million
USD
1,098.89 Million
2024
2032
USD
679.14 Million
USD
1,098.89 Million
2024
2032
| 2025 –2032 | |
| USD 679.14 Million | |
| USD 1,098.89 Million | |
|
|
|
Breast Reconstruction Market Analysis
The re-development of the breast in a woman's body is known as breast reconstruction. It entails constructing a natural-looking breast using autologous tissue or prosthetic material. Implants or prostheses, which include silicone or saline implants, and autologous or skin flap surgery, which uses tissue from another region of the body, are the two types of breast reconstruction surgery.
Europe Breast Reconstruction Market Size
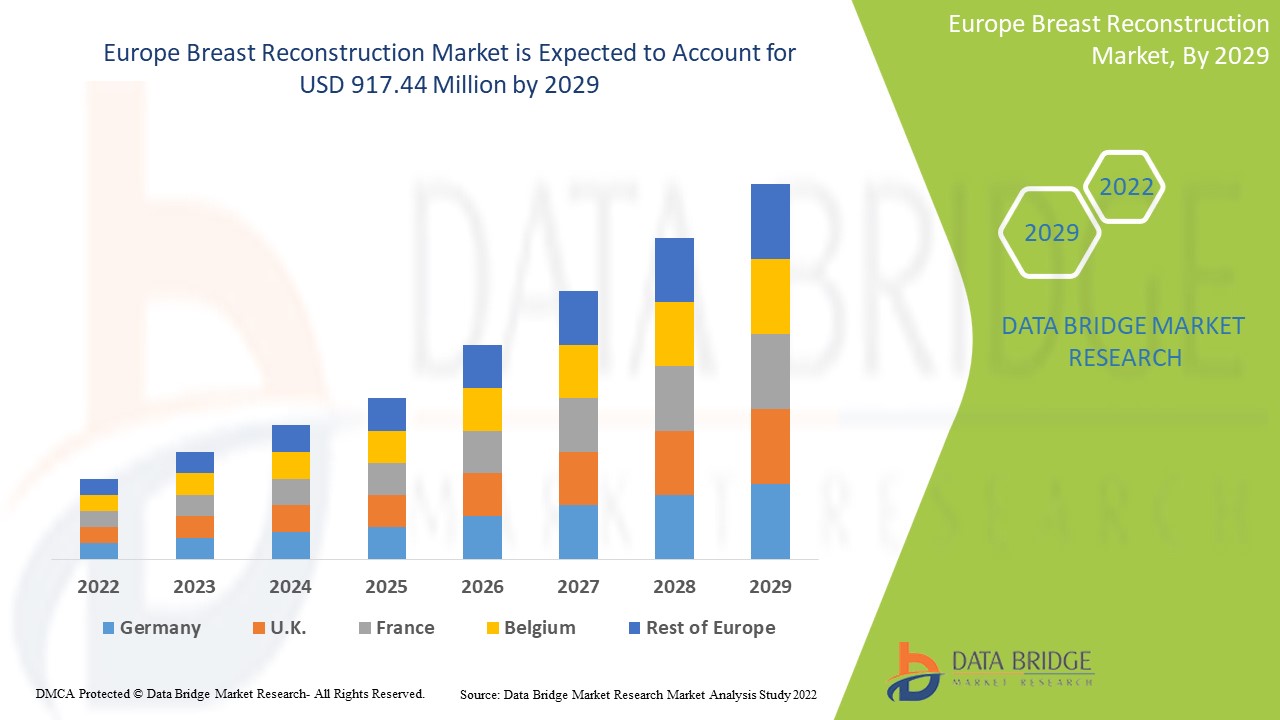
Europe breast reconstruction market size was valued at USD 679.14 million in 2024 and is projected to reach USD 1098.89 million by 2032, with a CAGR of 6.2% during the forecast period of 2025 to 2032.
Report Scope and Market Segmentation
|
Attributes |
Breast Reconstruction Key Market Insights |
|
Segmentation |
|
|
Countries Covered |
Germany, France, U.K., Netherlands, Switzerland, Belgium, Russia, Italy, Spain, Turkey, Rest of Europe in Europe |
|
Key Market Players |
POLYTECH Health & Aesthetics GmbH (Germany), CEREPLAS (France), Guangzhou Wanhe Plastic Material Co. Ltd (China), ALLERGAN (Ireland), Johnson & Johnson Private Limited (U.S), GC Aesthetics (U.K), Laboratories Arion (France), Sientra Inc. (California), HANSBIOMED CO. LTD (South Korea), Sientra, Inc. (U.S), Deal Implant Incorporated (U.S), Integra LifeSciences (U.S), PMT Corporation (U.S), RTI Surgical (U.S), Establishment Labs S.A. (U.S), Silimed (Brazil) |
|
Market Opportunities |
|
Breast Reconstruction Market Definition
Breast reconstruction is a surgical treatment that restores the contour of the breasts. To contour the breasts, silicone devices filled with silicone gel or salt water (saline) are used in conjunction with breast implants. Breast implant surgery is a difficult process. Various treatments for breast restoration are currently available, including nipple repair and a regenerative medicine approach employing 3D Printed Tissue Scaffolds. Nipple rebuilding treatments have progressed from a local skin flagship surgical method to current tissue engineering approaches.
Breast Reconstruction Market Dynamics
Drivers
- Growing breast cancer prevalence and incidences
Higher mastectomy rates, increased cancer risk due to elevated oestrogen and progesterone levels, and growing awareness of breast reconstruction choices lead to an increase in the number of women choosing for reconstruction treatments.
- Technological enhancements in the design
The use of novel technology that improves the comfort and safety of breast reconstruction surgeries and the availability of qualified plastic surgeons could boost industry demand.
- Growing number of breast cancer patients
Breast cancer is the fifth biggest cause of mortality among women from cancer. Breast cancer affects as least one out of every four women diagnosed with cancer, and this worrying increase has prompted the need for efficient therapy. Breast reconstruction surgeries are in high demand due to the ever-increasing prevalence of breast cancer around the world. Breast reconstruction is growing because of an increase in the frequency of hereditary illnesses in women and increased healthcare spending and government policy.
- Inventions and availability of numerous techniques proving beneficial
The desire for improved benefits and safety in these procedures has grown. Technological advances are expected to result in game-changing improvements in breast reconstruction surgery outcomes. The employment of contemporary imaging technologies in conjunction with ADMs simplifies the technique.
Opportunities
One of the technologies likely to influence the breast reconstruction markets is 3D printing, which is being utilized to build soft absorbable shells with silicon filling to integrate with the specific medical requirements of patients. The increased use of silicone and saline breast implants, ADMs, and tissue expanders is expected to be a major trend in breast reconstruction in the coming years.
Restraints/Challenges
High surgical costs and difficulties, such as adverse effects of surgical operations, changes in nipple and breast sensation, scar development, and, in some cases, rupture with deflation of saline-filled implants associated with breast reconstructions, have stifled the breast reconstruction market's growth. These issues are linked to Acellular Dermal Matrices (ADMs), as hematoma, skin necrosis, and seroma are the most commonly reported complications following ADM breast reconstruction procedures.
This breast reconstruction market report provides details of new recent developments, trade regulations, import-export analysis, production analysis, value chain optimization, market share, impact of domestic and localized market players, analyses opportunities in terms of emerging revenue pockets, changes in market regulations, strategic market growth analysis, market size, category market growths, application niches and dominance, product approvals, product launches, geographic expansions, technological innovations in the market. To gain more info on the breast reconstruction market contact Data Bridge Market Research for an Analyst Brief, our team will help you take an informed market decision to achieve market growth.
Breast Reconstruction Market Scope
The breast reconstruction market is segmented on the basis of product, type, shape, technology, placement, procedure and end user. The growth amongst these segments will help you analyze meagre growth segments in the industries and provide the users with a valuable market overview and market insights to help them make strategic decisions for identifying core market applications.
Product
- Breast Implants
- Silicone Implants
- Saline Implants
- Tissue Expanders
- Acellular Dermal Matrix
Type
- Unilateral
- Bilateral
Shape
- Round Implants
- Anatomical Implants
Technology
- Inframammary
- Peri-Areolar
- Trans-Axillary
- Transumbilical
Placement
- Dual-Plane Insertion
- Subglandular Insertion
- Submuscular Insertion
Procedure
- Immediate Procedures
- Delayed Procedures
- Revision Procedures
End User
- Hospitals
- Cosmetology Clinics
- Ambulatory Surgical Centers
- Others
Europe Breast Reconstruction Market Share
The breast reconstruction market is analyzed and market size insights and trends are provided by country, product, type, shape, technology, placement, procedure and end user as referenced above.
The countries covered in the breast reconstruction market report are Germany, France, U.K., Netherlands, Switzerland, Belgium, Russia, Italy, Spain, Turkey, and Rest of Europe in Europe.
The country section of the report also provides individual market impacting factors and changes in regulation in the market domestically that impacts the current and future trends of the market. Data points like down-stream and upstream value chain analysis, technical trends and porter's five forces analysis, case studies are some of the pointers used to forecast the market scenario for individual countries. Also, the presence and availability of global brands and their challenges faced due to large or scarce competition from local and domestic brands, impact of domestic tariffs and trade routes are considered while providing forecast analysis of the country data.
Breast Reconstruction Market Share
The breast reconstruction market competitive landscape provides details by competitor. Details included are company overview, company financials, revenue generated, market potential, investment in research and development, new market initiatives, global presence, production sites and facilities, production capacities, company strengths and weaknesses, product launch, product width and breadth, application dominance. The above data points provided are only related to the companies' focus related to breast reconstruction market.
Europe Breast Reconstruction Market Leaders Operating in the Market Are:
- POLYTECH Health & Aesthetics GmbH (Germany)
- CEREPLAS (France)
- Guangzhou Wanhe Plastic Material Co., Ltd (China)
- ALLERGAN (Ireland)
- Johnson & Johnson Private Limited (U.S)
- GC Aesthetics (U.K)
- Laboratories Arion (France)
- Sientra Inc. (California)
- HANSBIOMED CO. LTD (South Korea)
- Sientra, Inc. (U.S)
- Deal Implant Incorporated (U.S)
- Integra LifeSciences (U.S)
- PMT Corporation (U.S)
- RTI Surgical (U.S)
- Establishment Labs S.A. (U.S)
- Silimed (Brazil)
Latest Developments in Europe Breast Reconstruction Market
- In 2022, Mentor Worldwide LLC (Johnson & Johnson) will have a new MENTOR Memory Gel enhance breast implant authorised by the US Food and Drug Administration
- POLYTECH Health & Aesthetics gained FDA approval for its textured breast implants in 2021. (macrotextured and polyurethane-coated implants)
- In 2020, Sientra got pre-market approval from the US Food and Drug Administration for its OPUS-branded breast implant products
SKU-
Get online access to the report on the World's First Market Intelligence Cloud
- Interactive Data Analysis Dashboard
- Company Analysis Dashboard for high growth potential opportunities
- Research Analyst Access for customization & queries
- Competitor Analysis with Interactive dashboard
- Latest News, Updates & Trend analysis
- Harness the Power of Benchmark Analysis for Comprehensive Competitor Tracking
Research Methodology
Data collection and base year analysis are done using data collection modules with large sample sizes. The stage includes obtaining market information or related data through various sources and strategies. It includes examining and planning all the data acquired from the past in advance. It likewise envelops the examination of information inconsistencies seen across different information sources. The market data is analysed and estimated using market statistical and coherent models. Also, market share analysis and key trend analysis are the major success factors in the market report. To know more, please request an analyst call or drop down your inquiry.
The key research methodology used by DBMR research team is data triangulation which involves data mining, analysis of the impact of data variables on the market and primary (industry expert) validation. Data models include Vendor Positioning Grid, Market Time Line Analysis, Market Overview and Guide, Company Positioning Grid, Patent Analysis, Pricing Analysis, Company Market Share Analysis, Standards of Measurement, Global versus Regional and Vendor Share Analysis. To know more about the research methodology, drop in an inquiry to speak to our industry experts.
Customization Available
Data Bridge Market Research is a leader in advanced formative research. We take pride in servicing our existing and new customers with data and analysis that match and suits their goal. The report can be customized to include price trend analysis of target brands understanding the market for additional countries (ask for the list of countries), clinical trial results data, literature review, refurbished market and product base analysis. Market analysis of target competitors can be analyzed from technology-based analysis to market portfolio strategies. We can add as many competitors that you require data about in the format and data style you are looking for. Our team of analysts can also provide you data in crude raw excel files pivot tables (Fact book) or can assist you in creating presentations from the data sets available in the report.