Global Btk Inhibitor Drug Market
Market Size in USD Billion
CAGR :
% 
 USD
8.73 Billion
USD
14.88 Billion
2025
2033
USD
8.73 Billion
USD
14.88 Billion
2025
2033
| 2026 - 2033 | |
| USD 8.73 Billion | |
| USD 14.88 Billion | |
|
|
|
|
BTK Inhibitor Drug Market Size
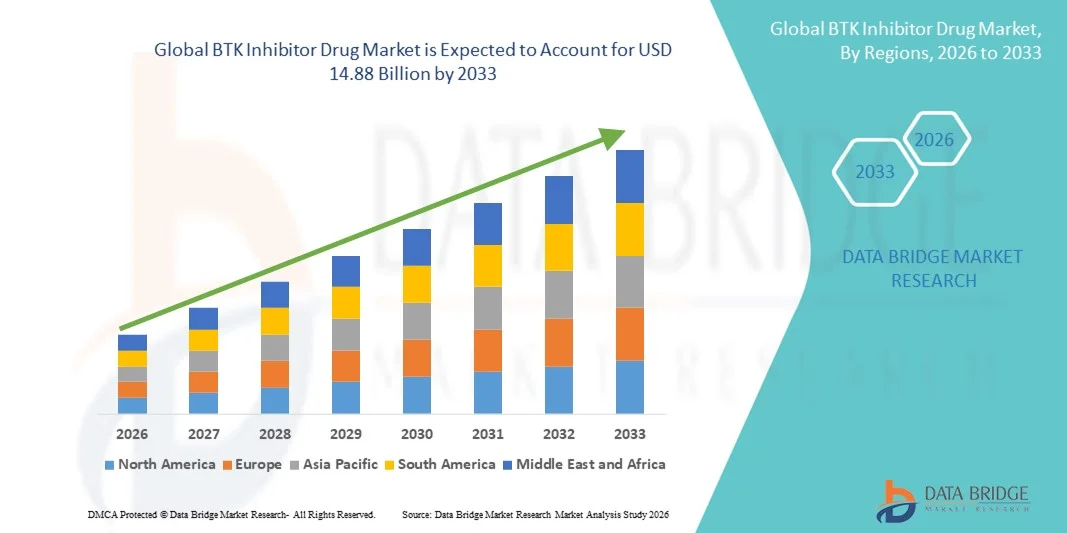
- The global BTK inhibitor drug market size was valued at USD 8.73 billion in 2025 and is expected to reach USD 14.88 billion by 2033, at a CAGR of 6.90% during the forecast period
- The market growth is largely fueled by the increasing prevalence of hematologic malignancies, rising investment in oncology R&D, and the development of next-generation BTK inhibitors with improved efficacy and safety profiles
- Furthermore, expanding indications into autoimmune and inflammatory disorders, along with growing adoption in clinical settings, is positioning BTK inhibitors as a key targeted therapy option. These converging factors are accelerating the uptake of BTK inhibitor drugs, thereby significantly boosting the industry's growth
BTK Inhibitor Drug Market Analysis
- BTK inhibitors, providing targeted inhibition of Bruton's tyrosine kinase in B-cell malignancies, are increasingly vital components of modern oncology treatment regimens due to their high specificity, oral administration convenience, and efficacy across multiple hematologic cancers
- The escalating demand for BTK inhibitors is primarily fueled by the rising prevalence of chronic lymphocytic leukemia (CLL), mantle cell lymphoma (MCL), and Waldenström’s macroglobulinemia (WM), growing investments in oncology R&D, and increasing adoption of next-generation BTK inhibitors with improved safety profiles
- North America dominated the BTK inhibitor drug market with the largest revenue share of 42.7% in 2025, characterized by early approval of BTK drugs, strong healthcare infrastructure, high awareness among oncologists, and a robust presence of key industry players, with the U.S. experiencing substantial growth driven by both established pharmaceutical companies and emerging biotech firms focusing on selective and dual BTK inhibitors
- Asia-Pacific is expected to be the fastest growing region in the BTK inhibitor drug market during the forecast period due to rising cancer incidence, improving healthcare access, and increasing investments in local manufacturing and clinical trials
- Chronic lymphocytic leukemia segment dominated the BTK inhibitor market with a market share of 38.9% in 2025, driven by its high prevalence, well-established treatment protocols incorporating BTK inhibitors, and favorable clinical outcomes in patients treated with these targeted therapies
Report Scope and BTK Inhibitor Drug Market Segmentation
|
Attributes |
BTK Inhibitor Drug Key Market Insights |
|
Segments Covered |
|
|
Countries Covered |
North America
Europe
Asia-Pacific
Middle East and Africa
South America
|
|
Key Market Players |
|
|
Market Opportunities |
|
|
Value Added Data Infosets |
In addition to the insights on market scenarios such as market value, growth rate, segmentation, geographical coverage, and major players, the market reports curated by the Data Bridge Market Research also include in-depth expert analysis, patient epidemiology, pipeline analysis, pricing analysis, and regulatory framework |
BTK Inhibitor Drug Market Trends
Expansion into Autoimmune and Inflammatory Disorders
- A significant and accelerating trend in the global BTK inhibitor drug market is the exploration of applications beyond oncology, including autoimmune diseases such as rheumatoid arthritis and lupus, reflecting broader therapeutic potential
- For instance, clinical trials of BTK inhibitors such as Evobrutinib in multiple sclerosis demonstrate promising efficacy and safety, signaling potential market expansion
- BTK inhibitors in non-oncology indications enable targeted modulation of B-cell signaling, offering therapeutic benefits for patients with chronic autoimmune conditions
- Integration with precision medicine approaches allows treatment plans tailored to patient immune profiles, enhancing efficacy while minimizing adverse effects
- This trend towards diversified indications is reshaping the competitive landscape, encouraging pharmaceutical companies to invest in pipeline expansion and combination therapies
- The demand for BTK inhibitors across both oncology and autoimmune therapy segments is growing rapidly, as healthcare providers increasingly prioritize targeted, disease-modifying treatments
- Increasing collaborations between biotech firms and academic research institutions are driving innovation and accelerating clinical development of novel BTK inhibitor formulations
- Growing patient awareness and advocacy for targeted therapies are influencing prescription patterns and accelerating adoption in both emerging and developed markets
BTK Inhibitor Drug Market Dynamics
Driver
Rising Prevalence of Hematologic Malignancies and Oncology R&D
- The increasing incidence of chronic lymphocytic leukemia (CLL), mantle cell lymphoma (MCL), and Waldenström’s macroglobulinemia (WM), combined with expanding oncology research, is a significant driver for BTK inhibitor adoption
- For instance, in June 2025, AbbVie announced expansion of its BTK inhibitor trials in first-line CLL therapy, reflecting rising R&D investments and clinical focus
- As oncologists seek targeted therapies with better efficacy and tolerability, BTK inhibitors offer compelling advantages over conventional chemotherapies, including oral dosing and lower systemic toxicity
- Furthermore, the growing number of next-generation BTK inhibitors with improved selectivity and fewer off-target effects is driving physician confidence and patient adoption
- The strong pipeline of BTK inhibitors for both existing and emerging indications is fueling market growth, encouraging healthcare providers to integrate these drugs into standard treatment protocols
- Strategic partnerships and collaborations between pharmaceutical companies and biotech firms further accelerate innovation and accessibility, contributing to rising market demand
- Increasing regulatory approvals across multiple countries are expanding global availability and facilitating earlier patient access to BTK inhibitors
- Enhanced clinical data and real-world evidence demonstrating improved patient outcomes are further reinforcing physician adoption and market growth
Restraint/Challenge
Adverse Effects and High Treatment Costs
- Safety concerns related to side effects such as bleeding, atrial fibrillation, and hypertension pose a challenge to broader BTK inhibitor adoption in clinical practice
- For instance, post-marketing studies of first-generation BTK inhibitors such as Ibrutinib reported cardiovascular complications in certain patient populations, necessitating careful monitoring
- Managing these adverse effects requires additional healthcare resources and physician oversight, which can limit uptake among patients with comorbidities
- In addition, the high cost of BTK inhibitor therapy, particularly in regions without reimbursement or insurance coverage, can restrict patient access and adoption
- While generic versions and next-generation drugs may reduce costs over time, the perceived premium remains a barrier, especially in emerging markets or for patients requiring long-term treatment
- Overcoming these challenges through improved safety profiles, patient education, and broader insurance coverage will be crucial for sustaining BTK inhibitor market growth
- Limited awareness among patients and some healthcare providers about newer BTK inhibitors can slow adoption in certain regions
- Stringent regulatory requirements and lengthy clinical trial processes for new indications can delay product launches and expansion into emerging markets
BTK Inhibitor Drug Market Scope
The market is segmented on the basis of product type, drug type, application, and end user.
- By Product Type
On the basis of product type, the BTK inhibitor drug market is segmented into First‑Generation BTK Inhibitors and Second‑Generation BTK Inhibitors. First‑Generation BTK Inhibitors dominated the market with the largest revenue share in 2025, driven by their early approval, widespread clinical adoption, and well-established efficacy in treating chronic lymphocytic leukemia (CLL) and mantle cell lymphoma (MCL). Physicians often prefer first-generation BTK inhibitors for their proven track record, extensive real-world data, and established dosing regimens. The market also sees strong demand for these inhibitors due to broad insurance coverage in developed regions and high familiarity among oncologists. Furthermore, first-generation BTK inhibitors such as Ibrutinib continue to maintain dominance due to their ability to manage multiple B-cell malignancies effectively.
Second‑Generation BTK Inhibitors are expected to witness the fastest growth during 2026–2033, fueled by improvements in selectivity and safety profiles that reduce off-target effects and adverse events. These next-generation inhibitors, such as Acalabrutinib and Zanubrutinib, are gaining adoption in both oncology and emerging autoimmune indications. Increased physician confidence, ongoing clinical trials for broader indications, and patient preference for better-tolerated therapies are contributing to their rapid uptake. Moreover, strategic launches in emerging markets and combination therapy applications further drive market expansion.
- By Drug Type
On the basis of drug type, the BTK inhibitor market is segmented into Selective BTK Inhibitors, Non‑Selective BTK Inhibitors, and Dual BTK Inhibitors. Selective BTK Inhibitors dominated the market in 2025 due to their targeted mechanism of action, which minimizes off-target toxicity and improves patient adherence. These inhibitors are highly preferred in oncology settings where safety and long-term administration are critical. The demand is further supported by their effectiveness across multiple B-cell malignancies and growing clinical evidence demonstrating superior tolerability compared to first-generation non-selective drugs. Leading products such as Acalabrutinib are widely prescribed in both the U.S. and Europe, reinforcing the dominance of selective BTK inhibitors.
Dual BTK Inhibitors are expected to witness the fastest CAGR from 2026 to 2033, driven by their ability to simultaneously target multiple signaling pathways, enhancing therapeutic efficacy. These inhibitors are gaining attention in research pipelines and clinical trials for challenging B-cell malignancies and autoimmune conditions. The potential for broader indications, reduced resistance development, and novel combination therapy applications positions dual BTK inhibitors as the next growth frontier in the market. Increased investment from biotech companies and regulatory approvals for novel dual inhibitors also accelerate adoption.
- By Application
On the basis of application, the market is segmented into Chronic Lymphocytic Leukemia (CLL), Mantle Cell Lymphoma (MCL), Waldenström’s Macroglobulinemia (WM), Follicular Lymphoma, Small Lymphocytic Lymphoma (SLL), Marginal Zone Lymphoma (MZL), Chronic Graft‑versus‑Host Disease (cGVHD), and Autoimmune & Inflammatory Disorders. Chronic Lymphocytic Leukemia (CLL) dominated the BTK inhibitor market in 2025 with a market share of 38.9%, owing to its high prevalence and the well-established efficacy of BTK inhibitors in managing disease progression. Physicians often prescribe BTK inhibitors as first-line therapy for CLL due to their superior clinical outcomes and oral administration convenience. The market dominance is reinforced by strong reimbursement coverage, real-world efficacy data, and long-term patient survival benefits. The demand is further supported by ongoing clinical trials exploring combination therapies and next-generation BTK inhibitors for CLL management.
Autoimmune & Inflammatory Disorders are expected to witness the fastest growth during 2026–2033, driven by expanding clinical trials and approvals for indications such as rheumatoid arthritis, lupus, and multiple sclerosis. The growing understanding of B-cell signaling in autoimmune pathologies, coupled with unmet therapeutic needs, positions BTK inhibitors as promising disease-modifying treatments. In addition, increasing awareness among healthcare providers and patients, coupled with strategic launches by pharmaceutical companies, accelerates adoption in this emerging segment.
- By End User
On the basis of end user, the BTK inhibitor market is segmented into hospitals, specialty clinics, and home healthcare. Hospitals dominated the market in 2025 due to their extensive oncology infrastructure, availability of specialized oncologists, and access to advanced treatment protocols. Hospitals are the preferred setting for BTK inhibitor administration and monitoring, particularly for patients with complex B-cell malignancies. The market dominance is supported by centralized healthcare systems, clinical trial participation, and the integration of BTK inhibitors into standard treatment regimens. Hospitals also offer long-term patient monitoring and support, ensuring better outcomes and reinforcing adoption.
Specialty Clinics are expected to witness the fastest growth during 2026–2033, fueled by the expansion of outpatient oncology and autoimmune care facilities. These clinics provide targeted therapy management, personalized monitoring, and easier access to BTK inhibitors, reducing hospital dependency. The growth is further supported by increasing partnerships with pharmaceutical companies, rising patient preference for outpatient care, and expansion of specialized autoimmune and oncology treatment centers in emerging regions.
BTK Inhibitor Drug Market Regional Analysis
- North America dominated the BTK inhibitor drug market with the largest revenue share of 42.7% in 2025, characterized by early approval of BTK drugs, strong healthcare infrastructure, high awareness among oncologists, and a robust presence of key industry players
- Physicians and healthcare providers in the region highly value the efficacy, safety, and well-established clinical data of BTK inhibitors, making them the preferred therapy for chronic lymphocytic leukemia (CLL), mantle cell lymphoma (MCL), and other B-cell malignancies
- This widespread adoption is further supported by strong insurance coverage, well-developed healthcare facilities, active clinical trial participation, and growing awareness among oncologists, establishing BTK inhibitors as a key targeted therapy option across both hospitals and specialty clinics
U.S. BTK Inhibitor Drug Market Insight
The U.S. BTK inhibitor drug market captured the largest revenue share of 82% in 2025 within North America, fueled by the early approval of multiple BTK inhibitors and the high prevalence of hematologic malignancies such as CLL and MCL. Physicians and healthcare providers are increasingly prioritizing targeted therapies due to their efficacy, oral administration convenience, and improved safety profiles compared to traditional chemotherapies. The growing number of clinical trials, strong reimbursement coverage, and awareness among oncologists further propels market adoption. In addition, collaborations between biotech firms and pharmaceutical companies are accelerating the availability of next-generation BTK inhibitors. The integration of BTK inhibitors into standard treatment protocols for both oncology and emerging autoimmune indications is significantly contributing to market growth.
Europe BTK Inhibitor Drug Market Insight
The Europe BTK inhibitor market is projected to expand at a substantial CAGR throughout the forecast period, primarily driven by the rising prevalence of B-cell malignancies and favorable regulatory approvals. The increase in specialized oncology centers and rising awareness among healthcare providers are fostering the adoption of BTK inhibitors. European physicians also value the efficacy and tolerability of these targeted therapies, particularly for CLL and MCL patients. The region is experiencing significant growth across hospitals, specialty clinics, and research institutions, with BTK inhibitors being increasingly incorporated into both first-line and combination therapy regimens.
U.K. BTK Inhibitor Drug Market Insight
The U.K. BTK inhibitor market is anticipated to grow at a noteworthy CAGR during the forecast period, driven by expanding homegrown oncology research, rising incidence of hematologic cancers, and strong adoption of targeted therapies. In addition, physician awareness of next-generation selective BTK inhibitors is encouraging their use in both oncology and autoimmune indications. The U.K.’s well-established healthcare infrastructure and active participation in clinical trials are expected to continue stimulating market growth, ensuring wider access and accelerated adoption of BTK inhibitor therapies.
Germany BTK Inhibitor Drug Market Insight
The Germany BTK inhibitor market is expected to expand at a considerable CAGR during the forecast period, fueled by increasing prevalence of CLL, MCL, and WM, and strong R&D in the oncology and autoimmune sectors. Germany’s well-developed healthcare infrastructure, combined with a focus on precision medicine and innovative therapies, promotes the adoption of BTK inhibitors. The integration of BTK inhibitors into specialized hospital programs and outpatient clinics is also becoming increasingly common, with a preference for therapies offering improved tolerability and patient outcomes.
Asia-Pacific BTK Inhibitor Drug Market Insight
The Asia-Pacific BTK inhibitor market is poised to grow at the fastest CAGR of 23% during the forecast period of 2026 to 2033, driven by rising cancer incidence, improving healthcare access, and increasing regulatory approvals in countries such as China, Japan, and India. The region’s growing focus on oncology and autoimmune research, supported by government initiatives promoting healthcare innovation, is driving adoption. Furthermore, as APAC emerges as a hub for clinical trials and local manufacturing of BTK inhibitors, the accessibility and affordability of these therapies are expanding to a wider patient base.
Japan BTK Inhibitor Drug Market Insight
The Japan BTK inhibitor market is gaining momentum due to the country’s advanced healthcare system, high awareness of targeted therapies, and rising prevalence of B-cell malignancies. Japanese healthcare providers prioritize the use of BTK inhibitors for their demonstrated efficacy and manageable safety profiles. The adoption is also fueled by integration into specialized oncology centers and research institutions. Moreover, Japan’s aging population is likely to increase demand for safer, patient-friendly oral therapies across both residential care and hospital settings.
India BTK Inhibitor Drug Market Insight
The India BTK inhibitor market accounted for the largest market revenue share in Asia-Pacific in 2025, attributed to increasing hematologic cancer prevalence, expanding oncology infrastructure, and rising healthcare awareness. India stands as one of the fastest-growing markets for targeted therapies, and BTK inhibitors are becoming increasingly prescribed in hospitals, specialty clinics, and clinical trial programs. Government initiatives supporting cancer treatment access, coupled with growing local pharmaceutical manufacturing and availability of affordable options, are key factors propelling market growth in India.
BTK Inhibitor Drug Market Share
The BTK Inhibitor Drug industry is primarily led by well-established companies, including:
- AbbVie Inc. (U.S.)
- AstraZeneca (U.K.)
- BeiGene, Ltd. (China)
- Pfizer Inc. (U.S.)
- Novartis AG (Switzerland)
- F. Hoffmann-La Roche Ltd (Switzerland)
- Takeda Pharmaceutical Company Limited (Japan)
- Eisai Co., Ltd. (Japan)
- Merck & Co., Inc. (U.S.)
- Sanofi (France)
- InnoCare Pharma (China)
- Bristol-Myers Squibb Company (U.S.)
- Gilead Sciences, Inc. (U.S.)
- Eli Lilly and Company (U.S.)
- Biogen Inc. (U.S.)
- Ono Pharmaceutical Co., Ltd. (Japan)
- TG Therapeutics, Inc. (U.S.)
- Corvus Pharmaceuticals, Inc. (U.S.)
- Sun Pharmaceutical Industries Ltd. (India)
What are the Recent Developments in Global BTK Inhibitor Drug Market?
- In September 2025, Novartis received U.S. FDA approval for Rhapsido® (remibrutinib) the first oral, targeted BTK inhibitor approved for the treatment of adult patients with chronic spontaneous urticaria (CSU) who remain symptomatic despite antihistamine therapy, marking a major expansion of BTK inhibitor use into autoimmune skin disorders beyond oncology
- In April 2025, China’s National Medical Products Administration (NMPA) approved InnoCare Pharma’s BTK inhibitor orelabrutinib as a first‑line treatment for CLL/SLL, MZL, and relapsed/refractory MCL, significantly broadening BTK inhibitor access in Asia
- In February 2025, the European Medicines Agency’s CHMP issued a positive opinion recommending approval of Lilly’s Jaypirca (pirtobrutinib) for adults with relapsed or refractory CLL previously treated with a BTK inhibitor, signaling regulatory momentum for the drug in the EU
- In December 2023, the U.S. FDA granted accelerated approval to pirtobrutinib (Jaypirca) for adults with chronic lymphocytic leukemia (CLL) or small lymphocytic lymphoma (SLL) previously treated with a BTK inhibitor and a BCL‑2 inhibitor, expanding its clinical use in hematologic malignancies
- In January 2023, the U.S. FDA approved Jaypirca™ (pirtobrutinib) as the first non‑covalent (reversible) BTK inhibitor for adult patients with relapsed or refractory mantle cell lymphoma (MCL) who had at least two prior lines of therapy, including a BTK inhibitor — marking a key advancement in targeted BTK cancer
SKU-
Get online access to the report on the World's First Market Intelligence Cloud
- Interactive Data Analysis Dashboard
- Company Analysis Dashboard for high growth potential opportunities
- Research Analyst Access for customization & queries
- Competitor Analysis with Interactive dashboard
- Latest News, Updates & Trend analysis
- Harness the Power of Benchmark Analysis for Comprehensive Competitor Tracking
Research Methodology
Data collection and base year analysis are done using data collection modules with large sample sizes. The stage includes obtaining market information or related data through various sources and strategies. It includes examining and planning all the data acquired from the past in advance. It likewise envelops the examination of information inconsistencies seen across different information sources. The market data is analysed and estimated using market statistical and coherent models. Also, market share analysis and key trend analysis are the major success factors in the market report. To know more, please request an analyst call or drop down your inquiry.
The key research methodology used by DBMR research team is data triangulation which involves data mining, analysis of the impact of data variables on the market and primary (industry expert) validation. Data models include Vendor Positioning Grid, Market Time Line Analysis, Market Overview and Guide, Company Positioning Grid, Patent Analysis, Pricing Analysis, Company Market Share Analysis, Standards of Measurement, Global versus Regional and Vendor Share Analysis. To know more about the research methodology, drop in an inquiry to speak to our industry experts.
Customization Available
Data Bridge Market Research is a leader in advanced formative research. We take pride in servicing our existing and new customers with data and analysis that match and suits their goal. The report can be customized to include price trend analysis of target brands understanding the market for additional countries (ask for the list of countries), clinical trial results data, literature review, refurbished market and product base analysis. Market analysis of target competitors can be analyzed from technology-based analysis to market portfolio strategies. We can add as many competitors that you require data about in the format and data style you are looking for. Our team of analysts can also provide you data in crude raw excel files pivot tables (Fact book) or can assist you in creating presentations from the data sets available in the report.