Растущая распространенность рака создает более высокий спрос на эффективные варианты лечения. Методы абляции опухолей предлагают минимально инвазивную альтернативу традиционной хирургии для лечения солидных опухолей. Поскольку заболеваемость раком продолжает расти, растет потребность в методах лечения, которые могут эффективно воздействовать на опухоли и уничтожать их, что приводит к росту рынка абляции опухолей.
Доступ к полному отчету по адресу https://www.databridgemarketresearch.com/reports/global-tumour-ablation-market
Процедуры абляции опухолей особенно ценны для пациентов с неоперабельными опухолями. К ним могут относиться опухоли в критических или труднодоступных областях или случаи, когда хирургическое вмешательство сопряжено с более высоким риском из-за общего состояния здоровья пациента. Абляция опухолей является жизнеспособным вариантом лечения для таких пациентов, позволяя уничтожить опухоль без серьезной операции.
Например,
- В 2020 году ВОЗ сообщила, что рак является основной причиной заболеваемости и смертности во всем мире: около 19,3 млн новых случаев рака и 10 млн смертей, связанных с раком.
- По данным МАИР (Международное агентство по исследованию рака), колоректальный рак является третьим по частоте диагностируемым заболеванием в мире. В 2020 году в мире было зарегистрировано около 1,9 миллиона новых случаев колоректального рака
Более того, растущая распространенность рака стимулирует спрос на процедуры абляции опухолей как эффективный и минимально инвазивный вариант лечения. Необходимость решения проблемы растущей заболеваемости раком в сочетании с достижениями в технологиях визуализации и предпочтением менее инвазивных процедур подпитывает рост рынка абляции опухолей.
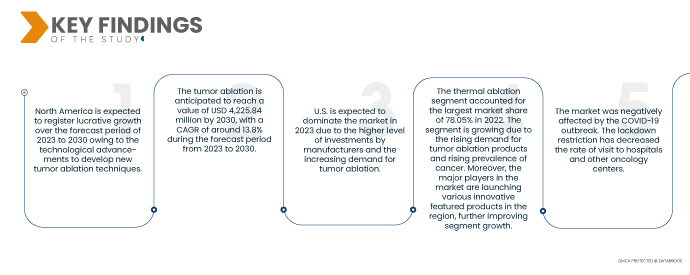
По данным исследования рынка Data Bridge, ожидается, что среднегодовой темп роста мирового рынка абляции опухолей составит 13,8% в прогнозируемый период с 2023 по 2030 год, а к 2030 году он, как ожидается, достигнет 4 225,84 млн долларов США. Прогнозируется, что растущий технологический прогресс будет способствовать росту рынка.
Основные выводы исследования
Растущая склонность хирургов и пациентов к минимально инвазивным процедурам
Минимально инвазивные процедуры предполагают меньшие разрезы или введение игл, чем традиционные открытые операции. Это приводит к меньшему повреждению тканей, уменьшению кровопотери и снижению риска осложнений. Такие процедуры обычно требуют более короткого пребывания в больнице и более быстрого восстановления. Пациенты часто могут возобновить свою обычную деятельность раньше, что улучшает качество их жизни.
Минимально инвазивные методы часто используют передовые технологии визуализации, позволяющие хирургам визуализировать и точно нацеливаться на опухоли. Это повышает точность и помогает сохранить здоровые окружающие ткани. Инструменты разработаны для обеспечения большей маневренности и гибкости, что позволяет хирургам выполнять сложные процедуры с улучшенным контролем и точностью. Процедуры предполагают меньшие разрезы, что снижает риск послеоперационных инфекций по сравнению с открытыми операциями.
Например,
- В исследовании, опубликованном в журнале European Urology Focus в 2020 году, сообщается о растущем использовании HIFU в качестве альтернативы хирургическому вмешательству или лучевой терапии при лечении рака предстательной железы.
- Исследование, опубликованное в журнале Journal of Thoracic Disease в 2019 году, изучало использование микроволновой абляции (MWA) для лечения опухолей легких. Авторы сообщили о растущей тенденции использования MWA в качестве минимально инвазивной альтернативы хирургии опухолей легких. Исследование подчеркнуло преимущества MWA с точки зрения сокращения сроков пребывания в больнице, более короткого времени восстановления и лучшего сохранения функции легких.
Растущая склонность хирургов и пациентов к минимально инвазивным процедурам, таким как абляция опухолей, обусловлена желанием улучшить результаты лечения пациентов, ускорить выздоровление и сократить расходы на здравоохранение. Эта тенденция подтолкнула рост мирового рынка абляции опухолей, что привело к более широкому принятию и развитию минимально инвазивных методов.
Область отчета и сегментация рынка
Отчет Метрика
|
Подробности
|
Прогнозируемый период
|
2023-2030
|
Базовый год
|
2022
|
Исторические годы
|
2021 (Возможность настройки на 2015–2020 гг.)
|
Количественные единицы
|
Доход в млн. долл. США
|
Охваченные сегменты
|
По типу (термическая абляция и химическая абляция), типу лечения (чрескожное, лапароскопическое и хирургическое), типу рака (рак молочной железы, рак легких, рак простаты, рак печени, рак почки, рак костей и другие), конечным пользователям (больницы, онкологические центры и другие), каналу сбыта (прямые торги и розничные продажи)
|
Страны, охваченные
|
США, Канада, Мексика, Германия, Великобритания, Франция, Италия, Россия, Испания, Нидерланды, Швейцария, Бельгия, Турция, Остальная Европа, Китай, Япония, Индия, Австралия, Южная Корея, Индонезия, Филиппины, Таиланд, Малайзия, Сингапур, Остальная часть Азиатско-Тихоокеанского региона, Бразилия, Аргентина, Остальная часть Южной Америки, Саудовская Аравия, Южная Африка, ОАЭ, Египет, Израиль, Остальной Ближний Восток и Африка
|
Охваченные участники рынка
|
Boston Scientific Corporation (США), Medtronic (США), Olympus (Германия), Stryker (США), Biotronik. (Германия), Merit Medical Systems. (Юта), HealthTronics, Inc. (США), Insightec (Израиль), AngioDynamics (Нью-Йорк), Integra LifeSciences (США), Bioventus LLC. (США), Ethicon Inc. (США), EDAP TMS (Овернь-Рона-Альпы), Sonablate Corp. (США), BVM Medical (Англия), Terumo Europe NV (Бельгия) и IceCure Medical (Израиль)
|
Данные, отраженные в отчете
|
Помимо аналитических данных о рыночных сценариях, таких как рыночная стоимость, темпы роста, сегментация, географический охват и основные игроки, рыночные отчеты, подготовленные Data Bridge Market Research, также включают в себя углубленный экспертный анализ, эпидемиологию пациентов, анализ воронки продаж, анализ ценообразования и нормативную базу.
|
Анализ сегмента:
Мировой рынок услуг по удалению опухолей разделен на пять основных сегментов, таких как тип, виды лечения, тип рака, конечные пользователи и канал сбыта.
- В зависимости от типа мировой рынок абляции опухолей сегментирован на термическую абляцию и химическую абляцию.
Ожидается, что в 2023 году сегмент термической абляции будет доминировать на мировом рынке абляции опухолей.
Ожидается, что в 2023 году сегмент термической абляции будет доминировать на рынке с долей рынка 78,12% и среднегодовым темпом роста 13,9% в прогнозируемый период с 2023 по 2030 год. Ожидается, что он будет доминировать на рынке из-за растущего спроса со стороны населения.
- По типу лечения рынок сегментирован на перкутанное, лапароскопическое и хирургическое. Ожидается, что в 2023 году перкутанное направление будет доминировать на рынке с долей рынка 56,32%, увеличившись с CAGR 14,1% в прогнозируемый период с 2023 по 2030 год.
- По типу рака рынок сегментирован на рак груди, рак легких, рак простаты, рак печени, рак почек, рак костей и др. Ожидается, что в 2023 году сегмент рака груди будет доминировать на рынке с долей рынка 33,50%, увеличившись с CAGR 15,0% в прогнозируемый период с 2023 по 2030 год.
- На основе конечных пользователей рынок сегментирован на больницы, онкологические центры и т. д. Ожидается, что в 2023 году сегмент больниц будет доминировать на рынке с долей рынка 53,94%, увеличиваясь со среднегодовым темпом роста 13,9% в прогнозируемый период с 2023 по 2030 год.
- По каналу сбыта рынок сегментируется на прямые торги и розничные продажи.
Ожидается, что в 2023 году сегмент прямых тендеров будет доминировать на мировом рынке абляции опухолей.
Ожидается, что в 2023 году сегмент прямых тендеров будет доминировать на рынке с долей рынка 57,20%, а среднегодовой темп роста составит 13,9% в прогнозируемый период с 2023 по 2030 год. Ожидается, что он будет доминировать из-за растущего спроса на процедуры абляции опухолей.
Основные игроки
Компания Data Bridge Market Research выделяет следующие компании в качестве игроков на мировом рынке абляции опухолей: Boston Scientific Corporation (США), Medtronic (США), Olympus (Германия), Stryker (США), Biotronik.(Германия), Merit Medical Systems.(Юта), HealthTronics, Inc.(США), Insightec (Израиль), AngioDynamics (Нью-Йорк), Integra LifeSciences (США), Bioventus LLC.(США), Ethicon Inc.(США), EDAP TMS (Овернь-Рона-Альпы), Sonablate Corp.(США), BVM Medical (Англия), Terumo Europe NV (Бельгия) и IceCure Medical (Израиль) и другие.
Развитие рынка
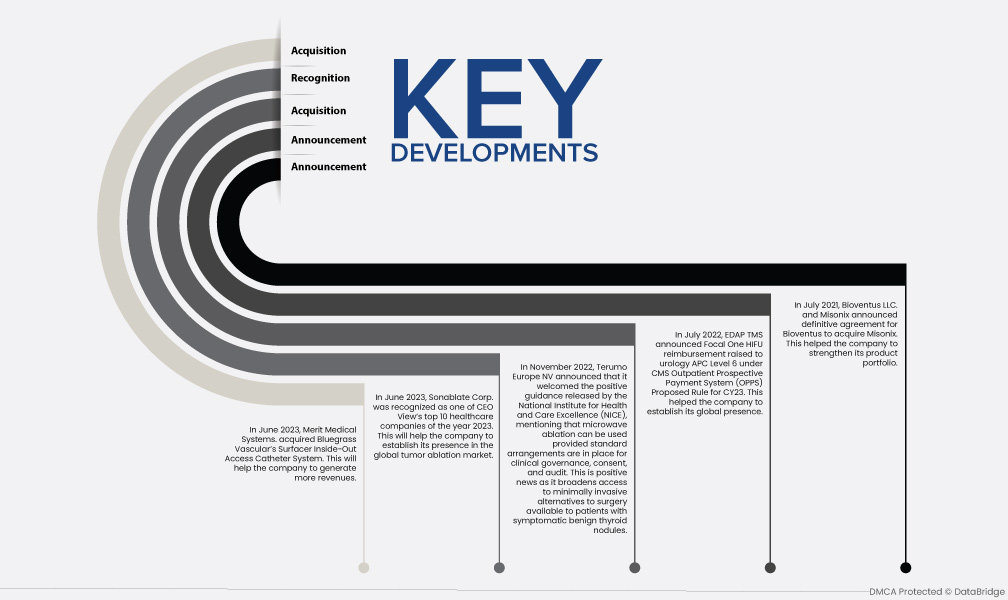
- В июне 2023 года Merit Medical Systems приобрела систему катетера Surfacer Inside-Out Access Catheter System компании Bluegrass Vascular. Это поможет компании увеличить доходы
- В июне 2023 года компания Sonablate Corp. была признана одной из 10 лучших компаний в сфере здравоохранения 2023 года по версии CEO View. Это поможет компании укрепить свое присутствие на мировом рынке абляции опухолей.
- В ноябре 2022 года компания Terumo Europe NV объявила, что приветствует позитивное руководство, выпущенное Национальным институтом здравоохранения и совершенствования медицинской помощи (NICE), в котором упоминается, что микроволновая абляция может использоваться при условии наличия стандартных механизмов клинического управления, согласия и аудита. Это позитивная новость, поскольку она расширяет доступ к минимально инвазивным альтернативам хирургии, доступным для пациентов с симптоматическими доброкачественными узлами щитовидной железы.
- В июле 2022 года EDAP TMS объявила о повышении возмещения Focal One HIFU до уровня APC 6 в урологии в соответствии с предлагаемым правилом CMS Outpatient Prospective Payment System (OPPS) для CY23. Это помогло компании установить свое глобальное присутствие
- В июле 2021 года Bioventus LLC и Misonix объявили о достижении окончательного соглашения о приобретении Misonix компанией Bioventus. Это помогло компании укрепить свой продуктовый портфель
Региональный анализ
Географически мировым рынком абляции опухолей охвачены следующие страны: США, Канада, Мексика, Германия, Великобритания, Франция, Италия, Россия, Испания, Нидерланды, Швейцария, Бельгия, Турция, остальные страны Европы, Китай, Япония, Индия, Австралия, Южная Корея, Индонезия, Филиппины, Таиланд, Малайзия, Сингапур, остальные страны Азиатско-Тихоокеанского региона, Бразилия, Аргентина, остальные страны Южной Америки, Саудовская Аравия, Южная Африка, ОАЭ, Египет, Израиль, остальные страны Ближнего Востока и Африки.
Согласно анализу Data Bridge Market Research:
Северная Америка является доминирующим и наиболее быстрорастущим регионом на мировом рынке абляции опухолей в прогнозируемый период с 2023 по 2030 год.
Ожидается, что Северная Америка будет доминировать на рынке из-за роста случаев рака в США и других странах региона. Северная Америка продолжит доминировать на рынке с точки зрения доли рынка и доходов и раскроет свое доминирование в течение прогнозируемого периода. Это связано с растущим внедрением передовых технологий и запуском новых продуктов в этом регионе. Кроме того, ожидается, что растущий спрос на абляцию опухолей еще больше увеличит темпы роста рынка этого региона.
Для получения более подробной информации об отчете о мировом рынке абляции опухолей нажмите здесь – https://www.databridgemarketresearch.com/reports/global-tumour-ablation-market