Europe Iron Deficiency Anemia Therapy Market
Market Size in USD Million
CAGR :
% 
 USD
1,085.60 Million
USD
3,683.60 Million
2022
2030
USD
1,085.60 Million
USD
3,683.60 Million
2022
2030
| 2023 –2030 | |
| USD 1,085.60 Million | |
| USD 3,683.60 Million | |
|
|
|
Europe Iron-Deficiency Anemia Therapy Market Analysis and Size
Anemia is one of the most prevalent blood disorders in the current times. Intravenous iron drugs are facing much use due to its improved quality of treatment outside the dialysis, rising the number of dialysis patients, increasing number of diabetic patients globally and increasing aging population. R&D in the treatment of iron deficiency anemia will create a competitive advantage for several manufacturers with new and innovative therapies in effective ways to develop and assess iron deficiency anemia therapy.
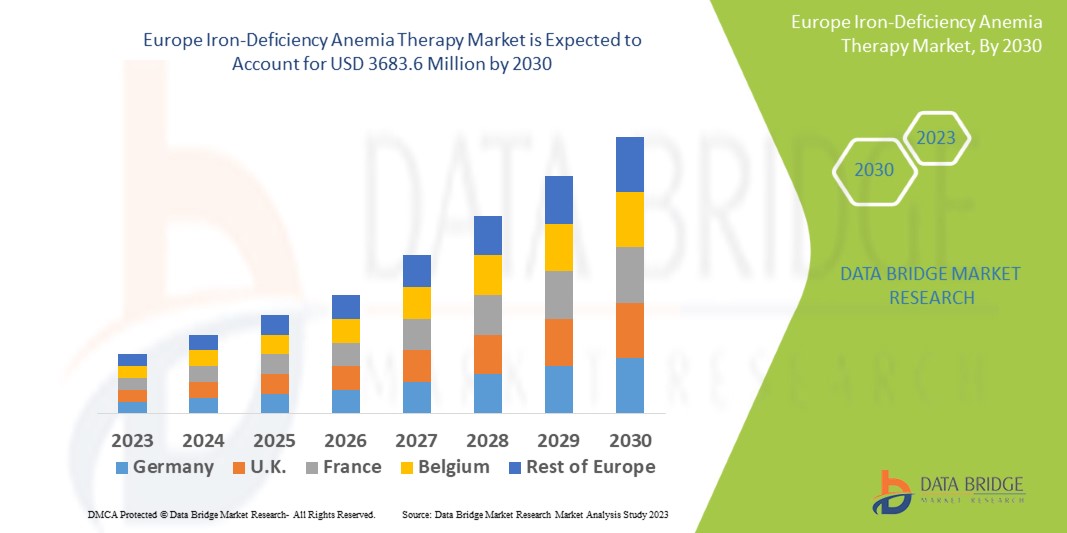
Data Bridge Market Research analyses a growth rate in the Europe iron-deficiency anemia therapy market in the forecast period 2023-2030. The expected CAGR of Europe iron-deficiency anemia therapy market is tend to be around 16.5% in the mentioned forecast period. The market was valued at USD 1085.6 million in 2022, and it would grow upto USD 3683.6 million by 2030. In addition to the insights on market scenarios such as market value, growth rate, segmentation, geographical coverage, and major players, the market reports curated by the Data Bridge Market Research also include depth expert analysis, patient epidemiology, pipeline analysis, pricing analysis, and regulatory framework.
Europe Iron-Deficiency Anemia Therapy Market Scope and Segmentation
|
Report Metric |
Details |
|
Forecast Period |
2023 to 2030 |
|
Base Year |
2022 |
|
Historic Years |
2021 (Customizable to 2015 - 2020) |
|
Quantitative Units |
Revenue in USD Million, Volumes in Units, Pricing in USD |
|
Segments Covered |
Therapy (Parenteral Iron Therapy and Oral Iron Therapy), Therapy Areas (Inflammatory Bowel Disease, Renal, Obstetrics and Gynecology, Congestive Heart Failure (CHF), Oncology and Others) Population (Adults, Pediatric and Geriatric), End-Users (Hospitals, Homecare, Speciality Centres, Others), Distribution Channel (Hospital Pharmacy, Online Pharmacy, Retail Pharmacy) |
|
Countries Covered |
Germany, France, U.K., Netherlands, Switzerland, Belgium, Russia, Italy, Spain, Turkey, Rest of Europe in Europe |
|
Market Players Covered |
Teva Pharmaceutical Industries Ltd. (Israel), Sanofi (France), Pfizer Inc. (U.S.), Novartis AG (Switzerland), Bayer AG (Germany), Merck & Co., Inc. (U.S.), Abbott (U.S.), AbbVie Inc. (U.S.), Sun Pharmaceutical Industries Ltd. (India), Hikma Pharmaceuticals PLC (U.K.), Covis Pharma GmbH (U.S.), PHARMACOSMOS A/S (Denmark), Vifor Pharma Management Ltd. (Switzerland), Orion Corporation (Finland), Shield Therapeutics (U.K.), Advanz Pharmaceutical (U.K.), Alkem Labs (India) |
|
Market Opportunities |
|
Market Definition
Iron deficiency anemia is a type of anemia that occurs when the body's iron stores are reduced. Anemia is specified by an inadequacy of healthy red blood cells. RBCs supply oxygen to human tissues. Iron is important in the production of haemoglobin. Haemoglobin deficiency shows lack of oxygen in the body. It's essential to keep your iron levels in check. Consumption of iron-rich foods such as meat, vegetables, fruits, and cereals can help to treat or prevent iron deficiency. Vitamin C can help to prevent iron deficiency.
Europe Iron-Deficiency Anemia Therapy Market Dynamics
Drivers
- Rising Incidences of Chronic Kidney Disease
According to the records of National Kidney Foundation, Inc., the two primary causes of chronic kidney disease are diabetes and hypertension. Data shows that about 10% of the European population is affected by chronic kidney disease, with more than 2 million individuals getting dialysis on a regular basis. Large number of individuals who are suffering from this disease are not aware until serious symptoms appear. It is anticipated that chronic kidney cases will rise across highly economic countries, such as India and China that infers an increase in the aged population over the upcoming years.
- Growing Adoption of Oral Iron Therapy
The oral iron therapy is anticipated to witness huge growth during the forecast period due to the factors such as rise in the patient population suffering from anemia and anemia-causing diseases. In addition to this, increasing number of research that supports the use of oral iron therapy as a substitute to intravenous treatments in hospital settings is also anticipated to increase the adoption of oral iron therapy among people, thus boosting the segment growth
Opportunities
- Increase in Anaemia
According to the records of the National Heart, Lung, and Blood Institute, an expected 3 million individuals suffer from anaemia in Europe. Rising incidence of target diseases, such as rheumatoid arthritis, autoimmune diseases, kidney conditions, liver disorders, thyroid diseases, cancer, and inflammatory bowel diseases, is a primary factor helping in the growth of the market for intravenous iron. According to the records of WHO, about 42% of children below the age of 5 years and 40% of pregnant women suffer from some form of anaemia. Therefore, it creates opportunity for the market growth.
- Increasing Incidence of Cancer
A rising number of cancer patients tend to develop iron deficiency anemia. Common types of cancer that are widely associated with anemia includes bone cancer, cervical cancer, blood cancer, colon cancer, and prostate cancer. Lung and breast tumors have been specified with the highest prevalence of anemia among solid tumors. Hematologic malignancies such as lymphoma, leukemia, and multiple myeloma produce abnormal blood cells that as a result harm the immune system and cause anemia. Cancer treatments such as chemotherapy and radiation therapy also increase anemia in cancer patients. This creates more opportunities in the market.
Restraints/Challenges
- Stringent Regulations by Different Associations
Numerous strict regulations are related with the intravenous iron drugs. Various government associations, such as FDA and European Medicines Agency have imposed stringent regulations for intravenous iron drugs approval and usage which takes longer to get approved and launched. Therefore, it hampers the market growth.
- Serious Effects of Iron Supplements
There are few adverse effects that are related with the intake of iron drugs. Common effects of include digestive problems such as loss of appetite, constipation, nausea, stomach aches, and vomiting. The overdose of these drugs may also lead to tiredness or weakness, shallow or rapid breathing, convulsions, fatigue, bluish skin, pale skin or fingernails. Thus, this hampers the market growth.
This Europe iron-deficiency anemia therapy market report provides details of new recent developments, trade regulations, import-export analysis, production analysis, value chain optimization, market share, impact of domestic and localized market players, analyses opportunities in terms of emerging revenue pockets, changes in market regulations, strategic market growth analysis, market size, category market growths, application niches and dominance, product approvals, product launches, geographic expansions, technological innovations in the market. To gain more info on the Europe iron-deficiency anemia therapy market contact Data Bridge Market Research for an Analyst Brief, our team will help you take an informed market decision to achieve market growth.
COVID-19 Impact on Europe Iron-Deficiency Anemia Therapy Market
The COVID-19 outbreak has disturbed the manufacturing and supply of intravenous iron drugs, however, increased initiatives by manufacturers and government to meet the demand has aided the growth of the market for intravenous iron drugs. In addition to this, numerous studies have recommended that Covid-19 infection has a huge risk of Acute Kidney Injury (AKI) and CKD, which, in turn, has fuelled the demand for intravenous iron drugs to treat CKD-related anemia. Health authorities advised that COVID-19 patients with an acute kidney injury should be tested often as their risk of developing CKD is higher than others. Thus, COVID-19 had a major impact on the market growth.
Europe Iron-Deficiency Anemia Therapy Market Scope
The Europe iron-deficiency anemia therapy market is segmented on the basis of therapy, therapy areas, population, distribution channel and end-user. The growth amongst these segments will help you analyze meagre growth segments in the industries and provide the users with a valuable market overview and market insights to help them make strategic decisions for identifying core market applications.
Therapy
- Parenteral Iron Therapy
- Oral Iron Therapy
Therapy Areas
- Inflammatory Bowel Disease
- Renal
- Obstetrics and Gynecology
- Congestive Heart Failure (CHF)
- Oncology
- Others
Population
- Adults
- Pediatric
- Geriatric
End-Users
- Hospitals
- Specialty Clinics
- Homecare
- Others
Distribution Channel
- Hospital Pharmacy
- Retail Pharmacy
- Online Pharmacy
- Others
Iron-Deficiency Anemia Therapy Market Regional Analysis/Insights
The Europe iron-deficiency anemia therapy market is analyzed and market size insights and trends are provided by therapy, therapy areas, population, distribution channel and end-user as referenced above.
The major countries covered in the europe iron-deficiency anemia therapy market report are the Germany, France, U.K., Netherlands, Switzerland, Belgium, Russia, Italy, Spain, Turkey, Rest of Europe in Europe.
Germany is leading the market because of the quality and management of health care system, growing research and development activities of anemia drugs among scientists. Also, increased awareness, growing investment by government firm and presence of generic players is leading the market growth.
The country section of the report also provides individual market impacting factors and changes in regulation in the market domestically that impacts the current and future trends of the market. Also, the presence and availability of global brands and their challenges faced due to large or scarce competition from local and domestic brands, impact of domestic tariffs and trade routes are considered while providing forecast analysis of the country data.
Competitive Landscape and Europe Iron-Deficiency Anemia Therapy Market Share Analysis
The Europe iron-deficiency anemia therapy market competitive landscape provides details by competitor. Details included are company overview, company financials, revenue generated, market potential, investment in research and development, new market initiatives, global presence, production sites and facilities, production capacities, company strengths and weaknesses, product launch, product width and breadth, application dominance. The above data points provided are only related to the companies' focus related to Europe iron-deficiency anemia therapy market.
Key players operating in the Europe iron-deficiency anemia therapy market include:
- Teva Pharmaceutical Industries Ltd. (Israel)
- Sanofi (France)
- Pfizer Inc. (U.S.)
- GSK plc (U.K.)
- Novartis AG (Switzerland)
- Bayer AG (Germany)
- Cipla Inc. (U.S.)
- Abbott (U.S.)
- AbbVie Inc. (U.S.)
- Sun Pharmaceutical Industries Ltd. (India)
- Aurobindo Pharma (India)
- Lupin (India)
- Hikma Pharmaceuticals PLC (U.K.)
- Covis Pharma GmbH (U.S.)
- PHARMACOSMOS A/S (Denmark)
- Vifor Pharma Management Ltd. (Switzerland)
- Orion Corporation (Finland)
- Shield Therapeutics (U.K.)
- Advanz Pharmaceutical (U.K.)
- Alkem Labs (India)
SKU-
Get online access to the report on the World's First Market Intelligence Cloud
- Interactive Data Analysis Dashboard
- Company Analysis Dashboard for high growth potential opportunities
- Research Analyst Access for customization & queries
- Competitor Analysis with Interactive dashboard
- Latest News, Updates & Trend analysis
- Harness the Power of Benchmark Analysis for Comprehensive Competitor Tracking
Table of Content
1 INTRODUCTION
1.1 OBJECTIVES OF THE STUDY
1.2 MARKET DEFINITION
1.3 OVERVIEW OF EUROPE IRON-DEFICIENCY ANEMIA THERAPY MARKET
1.4 CURRENCY AND PRICING
1.5 LIMITATION
1.6 MARKETS COVERED
2 MARKET SEGMENTATION
2.1 KEY TAKEAWAYS
2.2 ARRIVING AT THE EUROPE IRON-DEFICIENCY ANEMIA THERAPY MARKET SIZE
2.2.1 VENDOR POSITIONING GRID
2.2.2 TECHNOLOGY LIFE LINE CURVE
2.2.3 TRIPOD DATA VALIDATION MODEL
2.2.4 MARKET GUIDE
2.2.5 MULTIVARIATE MODELLING
2.2.6 TOP TO BOTTOM ANALYSIS
2.2.7 CHALLENGE MATRIX
2.2.8 APPLICATION COVERAGE GRID
2.2.9 STANDARDS OF MEASUREMENT
2.2.10 VENDOR SHARE ANALYSIS
2.2.11 EPIDEMIOLOGY MODELING
2.2.12 DATA POINTS FROM KEY PRIMARY INTERVIEWS
2.2.13 DATA POINTS FROM KEY SECONDARY DATABASES
2.3 EUROPE IRON-DEFICIENCY ANEMIA THERAPY MARKET: RESEARCH SNAPSHOT
2.4 ASSUMPTIONS
3 EXECUTIVE SUMMARY
4 PREMIUM INSIGHTS
4.1 PORTER'S 5 FORCES
4.2 PESTEL ANALYSIS
5 INDUSTRY INSIGHTS
5.1 PATENT ANALYSIS
5.1.1 PATENT LANDSCAPE
5.1.2 USPTO NUMBER
5.1.3 PATENT EXPIRY
5.1.4 EPIO NUMBER
5.1.5 PATENT STRENGTH AND QUALITY
5.1.6 PATENT CLAIMS
5.1.7 PATENT CITATIONS
5.1.8 PATENT LITIGATION AND LICENSING
5.1.9 FILE OF PATENT
5.1.10 PATENT RECEIVED CONTRIES
5.1.11 TECHNOLOGY BACKGROUND
5.2 DRUG TREATMENT RATE BY MATURED MARKETS
5.3 DEMOGRAPHIC TRENDS: IMPACTS ON ALL INCIDENCE RATES
5.4 PATIENT FLOW DIAGRAM
5.5 KEY PRICING STRATEGIES
5.6 KEY PATIENT ENROLLMENT STRATEGIES
5.7 INTERVIEWS WITH SPECIALIST
5.8 OTHER KOL SNAPSHOTS
6 EPIDEMIOLOGY
6.1 INCIDENCE OF ALL BY GENDER
6.2 TREATMENT RATE
6.3 MORTALITY RATE
6.4 DRUG ADHERENCE AND THERAPY SWITCH MODEL
6.5 PATIENT TREATMENT SUCCESS RATES
7 MERGERS AND ACQUISITION
7.1 LICENSING
7.2 COMMERCIALIZATION AGREEMENTS
8 REGULATORY FRAMEWORK
8.1 REGULATORY APPROVAL PROCESS
8.2 GEOGRAPHIES’ EASE OF REGULATORY APPROVAL
8.3 REGULATORY APPROVAL PATHWAYS
8.4 LICENSING AND REGISTRATION
8.5 POST-MARKETING SURVEILLANCE
8.6 GOOD MANUFACTURING PRACTICES (GMPS) GUIDELINES
9 PIPELINE ANALYSIS
9.1 CLINICAL TRIALS AND PHASE ANALYSIS
9.2 DRUG THERAPY PIPELINE
9.3 PHASE III CANDIDATES
9.4 PHASE II CANDIDATES
9.5 PHASE I CANDIDATES
9.6 OTHERS (PRE-CLINICAL AND RESEARCH)
TABLE 1 CLINICAL TRIAL MARKET FOR EUROPE IRON-DEFICIENCY ANEMIA THERAPY
Company Name Therapeutic Area
XX XX
XX XX
XX XX
XX XX
XX XX
XX XX
Sources: Press Releases, Annual Reports, SEC Filings, Investor Presentations, Other Government Sources, Analysis Based on Inputs from Secondary, Expert Interviews
TABLE 2 DISTRIBUTION OF PRODUCTS AND PROJECTS BY PHASE
Phase Number of Projects
Preclinical/Research Projects XX
Clinical Development XX
Phase I XX
Phase II XX
Phase III XX
U.S. Filed/Approved But Not Yest Marketed XX
Total XX
Sources: Press Releases, Annual Reports, SEC Filings, Investor Presentations, Other Government Sources, Analysis Based on Inputs from Secondary, Expert Interviews
TABLE 3 DISTRIBUTION OF PROJECTS BY THERAPEUTIC AREA AND PHASE
Therapeutic Area Preclinical/ Research Project
XX XX
XX XX
XX XX
XX XX
XX XX
Total Projects XX
Sources: Press Releases, Annual Reports, SEC Filings, Investor Presentations, Other Government Sources, Analysis Based on Inputs from Secondary, Expert Interviews
TABLE 4 DISTRIBUTION OF PROJECTS BY SCIENTIFIC APPROACH AND PHASE
Technology Preclinical/ Research Project
XX XX
XX XX
XX XX
XX XX
XX XX
Total Projects XX
Sources: Press Releases, Annual Reports, SEC Filings, Investor Presentations, Other Government Sources, Analysis Based on Inputs from Secondary, Expert Interviews
FIGURE 1 TOP ENTITIES BASED ON R&D GLANCE FOR EUROPE IRON-DEFICIENCY ANEMIA THERAPY
Sources: Press Releases, Annual Reports, SEC Filings, Investor Presentations, Other Government Sources, Analysis Based on Inputs from Secondary, Expert Interviews
10 MARKETED DRUG ANALYSIS
10.1 DRUG
10.1.1 BRAND NAME
10.1.2 GENERICS NAME
10.2 THERAPEUTIC INDIACTION
10.3 PHARACOLOGICAL CLASS OD THE DRUG
10.4 DRUG PRIMARY INDICATION
10.5 MARKET STATUS
10.6 MEDICATION TYPE
10.7 DRUG DOSAGES FORM
10.8 DOSAGES AVAILABILITY
10.9 PACKAGING TYPE
10.1 DRUG ROUTE OF ADMINISTRATION
10.11 DOSING FREQUENCY
10.12 DRUG INSIGHT
10.13 AN OVERVIEW OF THE DRUG DEVELOPMENT ACTIVITIES SUCH AS REGULATORY MILSTONE, SAFETY DATA AND EFFICACY DATA, MARKET EXCLUSIVITY DATA.
10.13.1 FORECAST MARKET OUTLOOK
10.13.2 CROSS COMPETITION
10.13.3 THERAPEUTIC PORTFOLIO
10.13.4 CURRENT DEVELOPMENT SCENARIO
11 MARKET ACCESS
11.1 10-YEAR MARKET FORECAST
11.2 CLINICAL TRIAL RECENT UPDATES
11.3 ANNUAL NEW FDA APPROVED DRUGS
11.4 DRUGS MANUFACTURER AND DEALS
11.5 MAJOR DRUG UPTAKE
11.6 CURRENT TREATMENT PRACTICES
11.7 IMPACT OF UPCOMING THERAPY
12 R & D ANALYSIS
12.1 COMPARATIVE ANALYSIS
12.2 DRUG DEVELOPMENTAL LANDSCAPE
12.3 IN-DEPTH INSIGHTS ON REGULATORY MILESTONES
12.4 THERAPEUTIC ASSESSMENT
13 ASSET-BASED COLLABORATIONS AND PARTNERSHIPSMARKET OVERVIEW
13.1 DRIVERS
13.2 RESTRAINS
13.3 OPPURTUNITY
13.4 CHALLENGES
14 EUROPE IRON-DEFICIENCY ANEMIA THERAPY MARKET, BY THERAPY TYPE
14.1 OVERVIEW
14.2 ORAL IRON THERPAY
14.2.1 FERROUS SULFATE
14.2.1.1. BY DOSAGE FORM
14.2.1.1.1. CAPSULE
14.2.1.1.2. TABLET
14.2.2 FERROUS GLUCONATE
14.2.2.1. BY DOSAGE FORM
14.2.2.1.1. CAPSULE
14.2.2.1.2. TABLET
14.2.3 FERROUS FUMARATE
14.2.3.1. BY DOSAGE FORM
14.2.3.1.1. CAPSULE
14.2.3.1.2. TABLET
14.2.4 POLYSACCHARIDE IRON COMPLEX (PIC)
14.2.4.1. BY DOSAGE FORM
14.2.4.1.1. CAPSULE
14.2.4.1.2. TABLET
14.2.5 OTHERS
14.2.5.1. OVER THE COUNTER DRUGS
14.2.5.2. OFF-LABEL GENERIC DRUGS
14.3 PARENTRAL IRON THERAPY
14.3.1 IRON DEXTRAN
14.3.2 IRON SUCROSE
14.3.3 FERRIC GLUCONATE
14.3.4 SODIUM FERRIC GLUCONATE COMPLEX
14.3.5 IRON ISOMALTOSIDE
14.4 ERYTHROPOIESIS-STIMULATING AGENTS (ESAS) THERAPY
14.4.1 BY ROUTE OF ADMINISTRTAION
14.4.1.1. INTRAVENOUS
14.4.1.2. SUBCUTANEOUS
14.5 OTHERS
15 EUROPE IRON-DEFICIENCY ANEMIA THERAPY MARKET, BY TYPE
15.1 OVERVIEW
15.2 BRANDED
15.3 GENERIC
16 EUROPE IRON-DEFICIENCY ANEMIA THERAPY MARKET, BY APPLICATION
16.1 OVERVIEW
16.2 INFLAMMATORY BOWEL DISEASE
16.2.1 ORAL IRON SUPPLEMENTS THERAPY
16.2.2 PARENTERAL IRON THERAPY
16.2.3 OTHERS
16.3 RENAL
16.3.1 ORAL IRON SUPPLEMENTS THERAPY
16.3.2 PARENTERAL IRON THERAPY
16.3.3 OTHERS
16.4 OBSTETRICS
16.4.1 ORAL IRON SUPPLEMENTS THERAPY
16.4.2 PARENTERAL IRON THERAPY
16.4.3 OTHERS
16.5 GYNECOLOGY
16.5.1 ORAL IRON SUPPLEMENTS THERAPY
16.5.2 PARENTERAL IRON THERAPY
16.5.3 OTHERS
16.6 CONGESTIVE HEART FAILURE (CHF)
16.6.1 ORAL IRON SUPPLEMENTS THERAPY
16.6.2 PARENTERAL IRON THERAPY
16.6.3 OTHERS
16.7 ONCOLOGY
16.7.1 ORAL IRON SUPPLEMENTS THERAPY
16.7.2 PARENTERAL IRON THERAPY
16.7.3 OTHERS
16.8 OTHERS
17 EUROPE IRON-DEFICIENCY ANEMIA THERAPY MARKET, BY CONDITION
17.1 OVERVIEW
17.2 MILD
17.3 MODERATE
17.4 SEVERE
18 EUROPE IRON-DEFICIENCY ANEMIA THERAPY MARKET, BY AGE GROUP
18.1 OVERVIEW
18.2 PEDIATRIC
18.3 ADULTS
18.4 GERIATRICS
19 EUROPE IRON-DEFICIENCY ANEMIA THERAPY MARKET, BY GENDER
19.1 OVERVIEW
19.2 MALE
19.3 FEMALE
20 EUROPE IRON-DEFICIENCY ANEMIA THERAPY MARKET, BY END USER
20.1 OVERVIEW
20.2 HOSPITALS
20.3 SPECIALTY CLINICS
20.4 HOMECARE SETTING
20.5 OTHERS
21 EUROPE IRON-DEFICIENCY ANEMIA THERAPY MARKET, BY DISTRIBUTION CHANNEL
21.1 OVERVIEW
21.2 HOSPITALS PHARMACY
21.3 ONLINE PHARMACY
21.4 RETAIL PHARMACY
21.5 OTHERS
22 EUROPE IRON-DEFICIENCY ANEMIA THERAPY MARKET, BY GEOGRAPHY
EUROPE IRON-DEFICIENCY ANEMIA THERAPY MARKET, (ALL SEGMENTATION PROVIDED ABOVE IS REPRESENTED IN THIS CHAPTER BY COUNTRY)
22.1 EUROPE
22.1.1 GERMANY.
22.1.1.1. GERMANY . IRON-DEFICIENCY ANEMIA THERAPY MARKET, BY THERAPY TYPE
22.1.1.2. GERMANY . IRON-DEFICIENCY ANEMIA THERAPY MARKET, BY CONDITION
22.1.1.3. GERMANY IRON-DEFICIENCY ANEMIA THERAPY MARKET, BY AGR GROUP
22.1.1.4. GERMANY IRON-DEFICIENCY ANEMIA THERAPY MARKET, BY GENDER
22.1.1.5. GERMANY IRON-DEFICIENCY ANEMIA THERAPY MARKET, BY END USER
22.1.1.6. GERMANY IRON-DEFICIENCY ANEMIA THERAPY MARKET, BY DISTRIBUTION CHANNEL
22.1.2 FRANCE
22.1.3 U.K.
22.1.4 ITALY
22.1.5 SPAIN
22.1.6 RUSSIA
22.1.7 TURKEY
22.1.8 BELGIUM
22.1.9 NETHERLANDS
22.1.10 SWITZERLAND
22.1.11 REST OF EUROPE
22.2 KEY PRIMARY INSIGHTS: BY MAJOR COUNTRIES
23 EUROPE IRON-DEFICIENCY ANEMIA THERAPY MARKET, SWOT AND DBMR ANALYSIS
24 EUROPE IRON-DEFICIENCY ANEMIA THERAPY MARKET, COMPANY LANDSCAPE
24.1 COMPANY SHARE ANALYSIS: EUROPE
24.2 MERGERS & ACQUISITIONS
24.3 NEW PRODUCT DEVELOPMENT & APPROVALS
24.4 EXPANSIONS
24.5 REGULATORY CHANGES
24.6 PARTNERSHIP AND OTHER STRATEGIC DEVELOPMENTS
25 EUROPE IRON-DEFICIENCY ANEMIA THERAPY MARKET, COMPANY PROFILE
25.1 ABBOTT
25.1.1 COMPANY OVERVIEW
25.1.2 REVENUE ANALYSIS
25.1.3 GEOGRAPHIC PRESENCE
25.1.4 PRODUCT PORTFOLIO
25.1.5 RECENT DEVELOPMENTS
25.2 SANOFI
25.2.1 COMPANY OVERVIEW
25.2.2 REVENUE ANALYSIS
25.2.3 GEOGRAPHIC PRESENCE
25.2.4 PRODUCT PORTFOLIO
25.2.5 RECENT DEVELOPMENTS
25.3 COVIS PHARMA GMBH
25.3.1 COMPANY OVERVIEW
25.3.2 REVENUE ANALYSIS
25.3.3 GEOGRAPHIC PRESENCE
25.3.4 PRODUCT PORTFOLIO
25.3.5 RECENT DEVELOPMENTS
25.4 DAIICHI SANKYO, INC
25.4.1 COMPANY OVERVIEW
25.4.2 REVENUE ANALYSIS
25.4.3 GEOGRAPHIC PRESENCE
25.4.4 PRODUCT PORTFOLIO
25.4.5 RECENT DEVELOPMENTS
25.5 PHARMACOSMOS THERAPEUTICS INC
25.5.1 COMPANY OVERVIEW
25.5.2 REVENUE ANALYSIS
25.5.3 GEOGRAPHIC PRESENCE
25.5.4 PRODUCT PORTFOLIO
25.5.5 RECENT DEVELOPMENTS
25.6 ABBVIE.
25.6.1 COMPANY OVERVIEW
25.6.2 REVENUE ANALYSIS
25.6.3 GEOGRAPHIC PRESENCE
25.6.4 PRODUCT PORTFOLIO
25.6.5 RECENT DEVELOPMENTS
25.7 NOVARTIS AG
25.7.1 COMPANY OVERVIEW
25.7.2 REVENUE ANALYSIS
25.7.3 GEOGRAPHIC PRESENCE
25.7.4 PRODUCT PORTFOLIO
25.7.5 RECENT DEVELOPMENTS
25.8 LUPIN
25.8.1 COMPANY OVERVIEW
25.8.2 REVENUE ANALYSIS
25.8.3 GEOGRAPHIC PRESENCE
25.8.4 PRODUCT PORTFOLIO
25.8.5 RECENT DEVELOPMENTS
25.9 CSL VIFOR
25.9.1 COMPANY OVERVIEW
25.9.2 REVENUE ANALYSIS
25.9.3 GEOGRAPHIC PRESENCE
25.9.4 PRODUCT PORTFOLIO
25.9.5 RECENT DEVELOPMENTS
25.1 SHIELD THERAPEUTICS PLC
25.10.1 COMPANY OVERVIEW
25.10.2 REVENUE ANALYSIS
25.10.3 GEOGRAPHIC PRESENCE
25.10.4 PRODUCT PORTFOLIO
25.10.5 RECENT DEVELOPMENTS
25.11 ALKEM LABORATORIES LTD.
25.11.1 COMPANY OVERVIEW
25.11.2 REVENUE ANALYSIS
25.11.3 GEOGRAPHIC PRESENCE
25.11.4 PRODUCT PORTFOLIO
25.11.5 RECENT DEVELOPMENTS
25.12 KERYX BIOPHARMACEUTICALS, INC. (AKEBIA THERAPEUTICS INC)
25.12.1 COMPANY OVERVIEW
25.12.2 REVENUE ANALYSIS
25.12.3 GEOGRAPHIC PRESENCE
25.12.4 PRODUCT PORTFOLIO
25.12.5 RECENT DEVELOPMENTS
25.12.6
NOTE: THE COMPANIES PROFILED IS NOT EXHAUSTIVE LIST AND IS AS PER OUR PREVIOUS CLIENT REQUIREMENT. WE PROFILE MORE THAN 100 COMPANIES IN OUR STUDY AND HENCE THE LIST OF COMPANIES CAN BE MODIFIED OR REPLACED ON REQUEST
26 RELATED REPORTS
27 CONCLUSION
28 QUESTIONNAIRE
29 ABOUT DATA BRIDGE MARKET RESEARCH

Research Methodology
Data collection and base year analysis are done using data collection modules with large sample sizes. The stage includes obtaining market information or related data through various sources and strategies. It includes examining and planning all the data acquired from the past in advance. It likewise envelops the examination of information inconsistencies seen across different information sources. The market data is analysed and estimated using market statistical and coherent models. Also, market share analysis and key trend analysis are the major success factors in the market report. To know more, please request an analyst call or drop down your inquiry.
The key research methodology used by DBMR research team is data triangulation which involves data mining, analysis of the impact of data variables on the market and primary (industry expert) validation. Data models include Vendor Positioning Grid, Market Time Line Analysis, Market Overview and Guide, Company Positioning Grid, Patent Analysis, Pricing Analysis, Company Market Share Analysis, Standards of Measurement, Global versus Regional and Vendor Share Analysis. To know more about the research methodology, drop in an inquiry to speak to our industry experts.
Customization Available
Data Bridge Market Research is a leader in advanced formative research. We take pride in servicing our existing and new customers with data and analysis that match and suits their goal. The report can be customized to include price trend analysis of target brands understanding the market for additional countries (ask for the list of countries), clinical trial results data, literature review, refurbished market and product base analysis. Market analysis of target competitors can be analyzed from technology-based analysis to market portfolio strategies. We can add as many competitors that you require data about in the format and data style you are looking for. Our team of analysts can also provide you data in crude raw excel files pivot tables (Fact book) or can assist you in creating presentations from the data sets available in the report.