Global Biologics Market
Market Size in USD Billion
CAGR :
% 
 USD
264.00 Billion
USD
596.65 Billion
2021
2029
USD
264.00 Billion
USD
596.65 Billion
2021
2029
| 2022 –2029 | |
| USD 264.00 Billion | |
| USD 596.65 Billion | |
|
|
|
|
Market Analysis and Size
Biologics have acquired a lot of momentum in recent years since they are used to treat a variety of diseases and ailments, such as Crohn's disease, ulcerative colitis, rheumatoid arthritis, and other autoimmune diseases. The product's widespread use is expected to drive growth in the global biologics market in the coming years. Biologics are at the cutting edge of research, supporting the most recent advances. Such breakthroughs are expected to result in revolutionary treatments that give patients new treatment options. New developments and unique therapies are being developed for the production of new biologics, indicating that the global biologics market is anticipated to have a positive future outlook.
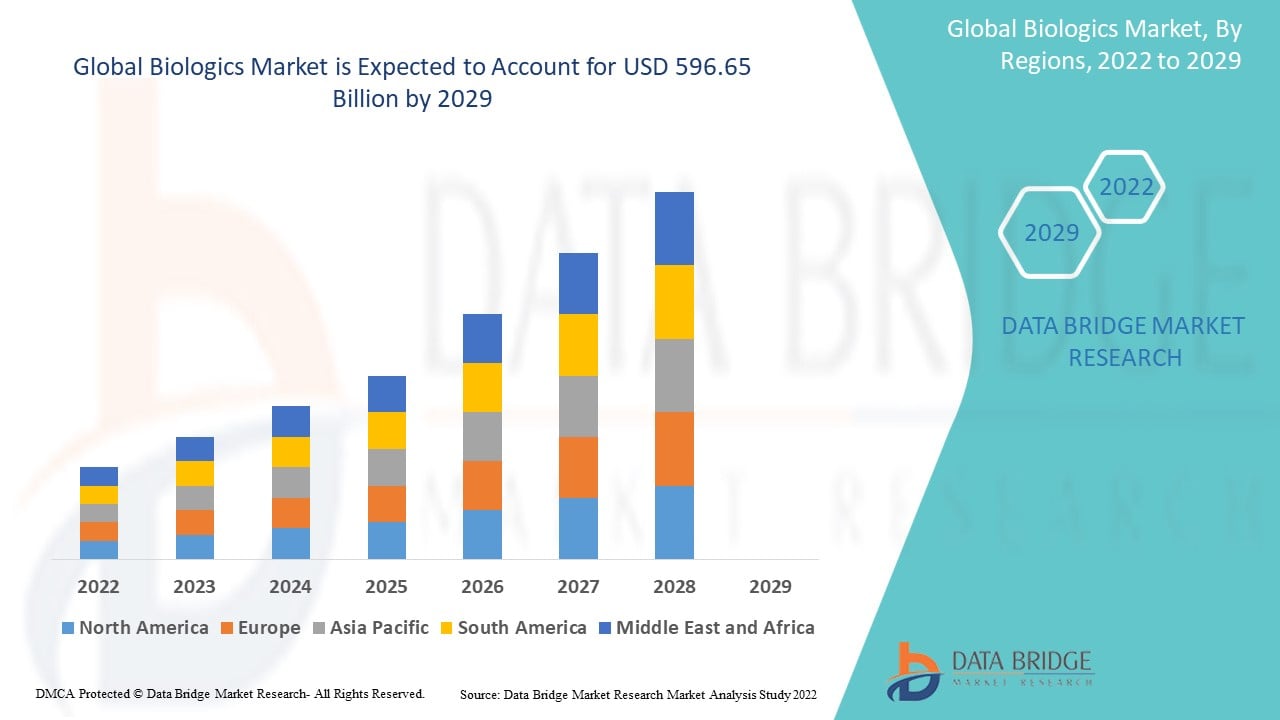
Data Bridge Market Research analyses that the biologics market was valued at USD 264 billion in 2021 and is expected to reach USD 596.65 billion by 2029, registering a CAGR of 9.25% during the forecast period of 2022 to 2029. In addition to the market insights such as market value, growth rate, market segments, geographical coverage, market players, and market scenario, the market report curated by the Data Bridge Market Research team includes in-depth expert analysis, patient epidemiology, pipeline analysis, pricing analysis, and regulatory framework.
Report Scope and Market Segmentation
|
Report Metric |
Details |
|
Forecast Period |
2022 to 2029 |
|
Base Year |
2021 |
|
Historic Years |
2020 (Customizable to 2019 - 2014) |
|
Quantitative Units |
Revenue in USD Billion, Volumes in Units, Pricing in USD |
|
Segments Covered |
Type (Tumor Necrosis Factor (TNF) Inhibitor, T-cell Inhibitor, Selective Co-stimulation Modulators, Interleukin-6 (IL-6) or Interleukin-17 Blocker, Interleukin-1 (IL-1) Blocker, B-cell Inhibitor), Drug Class (Monoclonal Antibody, Recombinant Insulin, Vaccine, Blood Factor, Human Growth Hormone, Erythropoietin, Fusion Protein, Recombinant Enzyme, Interferon, Colony-stimulating Factor, Gene Therapy, Cell Therapy, Oligonucleotides, Others), Therapeutic Application (Oncology, Autoimmune/Immunologic Diseases, Metabolic Disorders, Ophthalmic Diseases, Respiratory Disorders, Inflammatory Bowel Diseases (IBDs), Neurological Disorders, Cardiovascular Diseases (CVDs), Musculoskeletal Disorders (MSDs), Infectious Diseases, Others), Source (Microbial, Mammalian, Others), Manufacturing (Outsourced, In-House), Drug Type (Branded Drugs, Generic Drugs), Mode of Purchase (Prescription Drugs, Over-The-Counter (OTC) Drugs), Dosage Form (Injection, Tablets, Others), Route of Administration (Oral, Parenteral, Others), End-Users (Hospitals, Specialty Clinics, Homecare, Others), Distribution Channel (Hospital Pharmacy, Retail Pharmacy, Online Pharmacy, Others) |
|
Countries Covered |
U.S., Canada and Mexico in North America, Germany, France, U.K., Netherlands, Switzerland, Belgium, Russia, Italy, Spain, Turkey, Rest of Europe in Europe, China, Japan, India, South Korea, Singapore, Malaysia, Australia, Thailand, Indonesia, Philippines, Rest of Asia-Pacific (APAC) in the Asia-Pacific (APAC), Saudi Arabia, U.A.E, South Africa, Egypt, Israel, Rest of Middle East and Africa (MEA) as a part of Middle East and Africa (MEA), Brazil, Argentina and Rest of South America as part of South America |
|
Market Players Covered |
Pfizer Inc. (US), GlaxoSmithKline plc (UK), Novartis AG (Switzerland), Mylan N.V. (US), Teva Pharmaceutical Industries Ltd.(Israel), Sanofi (France), Boehringer Ingelheim International GmbH. (Germany), AstraZeneca (UK), Johnson & Johnson Private Limited (US), Merck & Co., Inc. (US), F. Hoffmann-La Roche Ltd. (Switzerland), Bristol-Myers Squibb Company (US), Eli Lilly and Company (US), Allergan (Ireland), Abbott (US), LEO Pharma A/S (Denmark), Sun Pharmaceutical Industries Ltd. (India), Aurobindo Pharma (India), Lupin (India), Hikma Pharmaceuticals PLC (UK), Zydus Cadila (India) |
|
Market Opportunities |
|
Market Definition
Biologics, commonly referred to as biological medications, are disease-modifying antirheumatic drugs (DMARDs) that are manufactured from live creatures or contain living organism components. Vaccines, blood, blood components, tissues, cells, allergies, genes, and recombinant proteins are among the items derived from humans, insects, microbes, animals, plants, and birds. These products regulate the development of essential proteins, alter human hormones and cells, and produce compounds that either activate or suppress the immune system. They also alter the way natural biologic intracellular and cellular activities function.
Biologics Market Dynamics
Drivers
- Rise in the prevalence of chronic diseases
The surging prevalence of chronic diseases is a major factor driving the biologics market's growth rate during the forecast period of 2022-2029. As per World Health Organization (WHO), chronic diseases kill more than 41 million people worldwide each year. The most common chronic disease, cardiovascular disease, kills 17.9 million people per year, followed by cancer, diabetes and respiratory disorders. These four groups are responsible for 80% of all chronic disease mortality. Chronic diseases have prompted the development of sophisticated diagnostics and therapeutics. Biologics are medicines that have been genetically modified to target a portion of the immune system that causes inflammation.
- Increasing investment for healthcare infrastructure
Another significant factor influencing the growth rate of biologics market is the rising healthcare expenditure which helps in improving its infrastructure. Also, various government organizations aims to improve the healthcare infrastructure by increasing funding and this will further influence the market dynamics.
Furthermore, rising initiatives by public and private organizations to spread awareness will expand the biologics market. Additionally, sedentary lifestyle of people and surging geriatric population will result in the expansion of biologics market. Along with this, favourable reimbursement policies will enhance the market's growth rate.
Opportunities
- Increase in the number of research and development activities
Moreover, the market's growth is fueled by an increase in the number of research and development activities. This will provide beneficial opportunities for the biologics market growth. In addition, scientists and researchers are studying species and expression systems to improve biological products' productivity. In addition, a number of pharmaceutical companies are conducting research and development (R and D) to increase the efficacy of oral medications for rheumatoid arthritis and Crohn's disease. Along with this, rising drug approvals and launches will further propel the market's growth rate.
Moreover, rising investment for the development of advanced technologies and an increase in the number of emerging markets will further provide beneficial opportunities for the growth of the biologics market during the forecast period.
Restraints/Challenges
On the other hand, the high cost associated with drug development and distribution will obstruct the market's growth rate. The lack of healthcare infrastructure in developing economies and dearth of skilled professionals will challenge the biologics market. Additionally, lack of awareness about the utilization of biologics in research centers will act as restrain and further impede the growth rate of market during the forecast period of 2022-2029. Biologics are sensitive to heat and light, due to which they require a superior refrigeration procedure that is not consistent over the world. This is estimated to obstruct the market's growth rate. .
This biologics market report provides details of new recent developments, trade regulations, import-export analysis, production analysis, value chain optimization, market share, impact of domestic and localized market players, analyses opportunities in terms of emerging revenue pockets, changes in market regulations, strategic market growth analysis, market size, category market growths, application niches and dominance, product approvals, product launches, geographic expansions, technological innovations in the market. To gain more info on the biologics market contact Data Bridge Market Research for an Analyst Brief, our team will help you take an informed market decision to achieve market growth.
Patient Epidemiology Analysis
Biologics market also provides you with detailed market analysis for patient analysis, prognosis and cures. Prevalence, incidence, mortality, adherence rates are some of the data variables that are available in the report. Direct or indirect impact analyses of epidemiology to market growth are analysed to create a more robust and cohort multivariate statistical model for forecasting the market in the growth period.
COVID-19 Impact on Biologics Market
The COVID-19 outbreak and subsequent lockdown in numerous countries around the world had a huge impact on the financial status of enterprises in all sectors. The private healthcare sector is one of the areas where the pandemic had a significant impact. The coronavirus pandemic had a significant influence on medicine development, production, supply, and the businesses of different healthcare corporations around the world. The outbreak has resulted in the shutdown of industrial establishments, with the exception of those that manufacture critical commodities, and disruptions in product supply chains. As a result, the COVID-19 outbreak had impacted the economy in three ways: directly influencing production and demand, causing distribution channel disruptions, and having a financial impact on firms and financial markets.
On the brighter side, during the pandemic, there was a high need for biologics due to the increased COVID-19 burden, which further fuelled the market's growth rate. The National Research Council of Canada and CanSino Biologics, located in China, partnered in May 2020 to develop a COVID-19 vaccine for clinical use in Canada. Ad5-nCoV is an adenovirus Type 5 vector-based vaccination that is currently being developed in Wuhan, China. As a result, the COVID-19 outbreak had a positive impacted on the biologics market. Furthermore, the number of research and development activities on biologics for the treatment of COVID-19 is also increased. Additionally, the governments of various countries are also taking new initiatives for promoting the healthcare sector and ensuring the availability of biologics
Recent Development
- In November 2021, Bristol-Myers Squibb Company had received the U.S. Food and Drug Administration approval for Opdivo (nivolumab) plus Yervoy (ipilimumab) combined with limited chemotherapy as first-line treatment of metastatic or recurrent non-small cell lung cancer. Patients with squamous or non-squamous illness, independent of PD-L1 expression, are eligible for the treatment. 1 The FDA's Real-Time Oncology Examine (RTOR) pilot programme was used to review this application, which aims to ensure that safe and effective medicines are offered to patients as soon as feasible.
Global Biologics Market Scope
The biologics market is segmented on the basis of type, drug class, therapeutic application, source, manufacturing, drug type, mode of purchase, dosage form, route of administration, end-users and distribution channel. The growth amongst these segments will help you analyze meagre growth segments in the industries and provide the users with a valuable market overview and market insights to help them make strategic decisions for identifying core market applications.
Type
- Tumor Necrosis Factor (TNF) Inhibitor
- T-cell Inhibitor
- Selective Co-stimulation Modulators
- Interleukin-6 (IL-6) or Interleukin-17 Blocker
- Interleukin-1 (IL-1) Blocker
- B-cell Inhibitor
Drug Class
- Monoclonal Antibody
- Recombinant Insulin
- Vaccine
- Blood Factor
- Human Growth Hormone
- Erythropoietin
- Fusion Protein
- Recombinant Enzyme
- Interferon
- Colony-stimulating Factor
- Gene Therapy
- Cell Therapy
- Oligonucleotides
- Others
- Peptide
- Botulinum Toxin
- Others
Therapeutic Application
- Oncology
- Autoimmune/Immunologic Diseases
- Metabolic Disorders
- Ophthalmic Diseases
- Respiratory Disorders
- Inflammatory Bowel Diseases (IBDs)
- Neurological Disorders
- Cardiovascular Diseases (CVDs)
- Musculoskeletal Disorders (MSDs)
- Infectious Diseases
- Others
Source
- Microbial
- Mammalian
- Others
Manufacturing
- Outsourced
- In-House
Drug Type
- Branded Drugs
- Generic Drugs
Mode of Purchase
- Prescription Drugs
- Over-The-Counter (OTC) Drugs
Dosage Form
- Injection
- Tablets
- Others
Route of Administration
- Parenteral
- Oral
- Others
End-Users
- Hospitals
- Specialty Clinics
- Homecare
- Others
Distribution Channel
- Hospital Pharmacy
- Retail Pharmacy
- Online Pharmacy
- Others
Biologics Market Regional Analysis/Insights
The biologics market is analysed and market size insights and trends are provided by country, drug type, drug class, therapeutic application, source, manufacturing, drug type, mode of purchase, dosage form, route of administration, end-users and distribution channel as referenced above.
The countries covered in the biologics market report are U.S., Canada and Mexico in North America, Germany, France, U.K., Netherlands, Switzerland, Belgium, Russia, Italy, Spain, Turkey, Rest of Europe in Europe, China, Japan, India, South Korea, Singapore, Malaysia, Australia, Thailand, Indonesia, Philippines, Rest of Asia-Pacific (APAC) in the Asia-Pacific (APAC), Saudi Arabia, U.A.E, South Africa, Egypt, Israel, Rest of Middle East and Africa (MEA) as a part of Middle East and Africa (MEA), Brazil, Argentina and Rest of South America as part of South America
North America dominates the biologics market in terms of market share and market revenue and will continue to flourish its dominance during the forecast period. This is due to the presence of major key players and rising healthcare expenditure will further propel the market's growth rate in this region. Additionally, increase in strategic collaborations for the development of biologics treatment for acute as well as chronic diseases will further propel the market's growth rate in this region.
Asia-Pacific are expected to grow during the forecast period of 2022-2029 due to surging prevalence of rheumatoid arthritis, kidney and liver disorders in this region. Also, development of healthcare infrastructure and rising geriatric population will further propel the market's growth rate in this region.
The country section of the report also provides individual market impacting factors and changes in regulation in the market domestically that impacts the current and future trends of the market. Data points like down-stream and upstream value chain analysis, technical trends and porter's five forces analysis, case studies are some of the pointers used to forecast the market scenario for individual countries. Also, the presence and availability of global brands and their challenges faced due to large or scarce competition from local and domestic brands, impact of domestic tariffs and trade routes are considered while providing forecast analysis of the country data.
Competitive Landscape and Biologics Market Share Analysis
The biologics market competitive landscape provides details by competitor. Details included are company overview, company financials, revenue generated, market potential, investment in research and development, new market initiatives, global presence, production sites and facilities, production capacities, company strengths and weaknesses, product launch, product width and breadth, application dominance. The above data points provided are only related to the companies' focus related to biologics market.
Some of the major players operating in the biologics market are:
- Pfizer Inc. (US)
- GlaxoSmithKline plc (UK)
- Novartis AG (Switzerland)
- Mylan N.V. (US)
- Teva Pharmaceutical Industries Ltd. (Israel)
- Sanofi (France)
- Boehringer Ingelheim International GmbH. (Germany)
- AstraZeneca (UK)
- Johnson & Johnson Private Limited (US)
- Merck & Co., Inc. (US)
- F. Hoffmann-La Roche Ltd. (Switzerland)
- Bristol-Myers Squibb Company (US)
- Eli Lilly and Company (US)
- Allergan (Ireland)
- Abbott (US)
- LEO Pharma A/S (Denmark)
- Sun Pharmaceutical Industries Ltd. (India)
- Aurobindo Pharma (India)
- Lupin (India)
- Hikma Pharmaceuticals PLC (UK)
- Zydus Cadila (India)
SKU-
Get online access to the report on the World's First Market Intelligence Cloud
- Interactive Data Analysis Dashboard
- Company Analysis Dashboard for high growth potential opportunities
- Research Analyst Access for customization & queries
- Competitor Analysis with Interactive dashboard
- Latest News, Updates & Trend analysis
- Harness the Power of Benchmark Analysis for Comprehensive Competitor Tracking
Table of Content
1 INTRODUCTION
1.1 OBJECTIVES OF THE STUDY
1.2 MARKET DEFINITION
1.3 OVERVIEW OF GLOBAL BIOLOGICS MARKET
1.4 CURRENCY AND PRICING
1.5 LIMITATION
1.6 MARKETS COVERED
2 MARKET SEGMENTATION
2.1 KEY TAKEAWAYS
2.2 ARRIVING AT THE GLOBAL XX SIZE
2.2.1.1. VENDOR POSITIONING GRID
2.2.1.2. TECHNOLOGY LIFE LINE CURVE
2.2.1.3. TRIPOD DATA VALIDATION MODEL
2.2.1.4. MARKET GUIDE
2.2.1.5. MULTIVARIATE MODELLING
2.2.1.6. TOP TO BOTTOM ANALYSIS
2.2.1.7. CHALLENGE MATRIX
2.2.1.8. APPLICATION COVERAGE GRID
2.2.1.9. STANDARDS OF MEASUREMENT
2.2.1.10. VENDOR SHARE ANALYSIS
2.2.1.11. DATA POINTS FROM KEY PRIMARY INTERVIEWS
2.2.1.12. DATA POINTS FROM KEY SECONDARY DATABASES
2.3 GLOBAL BIOLOGICS MARKET: RESEARCH SNAPSHOT
2.4 ASSUMPTIONS
3 EXECUTIVE SUMMARY
4 PREMIUM INSIGHTS
4.1 PESTEL ANALYSIS
4.2 PORTER’S FIVE FORCES MODEL
5 INDUSTRY INSIGHTS
5.1 PATENT ANALYSIS
5.1.1.1. PATENT LANDSCAPE
5.1.1.2. USPTO NUMBER
5.1.1.3. PATENT EXPIRY
5.1.1.4. EPIO NUMBER
5.1.1.5. PATENT STRENGTH AND QUALITY
5.1.1.6. PATENT CLAIMS
5.1.1.7. PATENT CITATIONS
5.1.1.8. PATENT LITIGATION AND LICENSING
5.1.1.9. FILE OF PATENT
5.1.1.10. PATENT RECEIVED CONTRIES
5.1.1.11. TECHNOLOGY BACKGROUND
5.2 DRUG TREATMENT RATE BY MATURED MARKETS
5.3 DEMOGRAPHIC TRENDS: IMPACTS ON ALL INCIDENCE RATES
5.4 PATIENT FLOW DIAGRAM
5.5 KEY PRICING STRATEGIES
5.6 KEY PATIENT ENROLLMENT STRATEGIES
5.7 INTERVIEWS WITH SPECIALIST
5.8 OTHER KOL SNAPSHOTS
6 EPIDEMIOLOGY
6.1 INCIDENCE OF ALL BY GENDER
6.2 TREATMENT RATE
6.3 MORTALITY RATE
6.4 DRUG ADHERENCE AND THERAPY SWITCH MODEL
6.5 PATIENT TREATMENT SUCCESS RATES
7 MERGERS AND ACQUISITION
7.1 LICENSING
7.2 COMMERCIALIZATION AGREEMENTS
8 REGULATORY FRAMEWORK
8.1 REGULATORY APPROVAL PROCESS
8.2 GEOGRAPHIES’ EASE OF REGULATORY APPROVAL
8.3 REGULATORY APPROVAL PATHWAYS
8.4 LICENSING AND REGISTRATION
8.5 POST-MARKETING SURVEILLANCE
8.6 GOOD MANUFACTURING PRACTICES (GMPS) GUIDELINES
9 PIPELINE ANALYSIS
9.1 CLINICAL TRIALS AND PHASE ANALYSIS
9.2 DRUG THERAPY PIPELINE
9.3 PHASE III CANDIDATES
9.4 PHASE II CANDIDATES
9.5 PHASE I CANDIDATES
9.6 OTHERS (PRE-CLINICAL AND RESEARCH)
TABLE 1 GLOBAL CLINICAL TRIAL MARKET FOR BIOLOGICS
Company Name Therapeutic Area
XX XX
XX XX
XX XX
XX XX
XX XX
XX XX
Sources: Press Releases, Annual Reports, SEC Filings, Investor Presentations, Other Government Sources, Analysis Based on Inputs from Secondary, Expert Interviews
TABLE 2 DISTRIBUTION OF PRODUCTS AND PROJECTS BY PHASE
Phase Number of Projects
Preclinical/Research Projects XX
Clinical Development XX
Phase I XX
Phase II XX
Phase III XX
U.S. Filed/Approved But Not Yest Marketed XX
Total XX
Sources: Press Releases, Annual Reports, SEC Filings, Investor Presentations, Other Government Sources, Analysis Based on Inputs from Secondary, Expert Interviews
TABLE 3 DISTRIBUTION OF PROJECTS BY THERAPEUTIC AREA AND PHASE
Therapeutic Area Preclinical/ Research Project
XX XX
XX XX
XX XX
XX XX
XX XX
Total Projects XX
Sources: Press Releases, Annual Reports, SEC Filings, Investor Presentations, Other Government Sources, Analysis Based on Inputs from Secondary, Expert Interviews
TABLE 4 DISTRIBUTION OF PROJECTS BY SCIENTIFIC APPROACH AND PHASE
Technology Preclinical/ Research Project
XX XX
XX XX
XX XX
XX XX
XX XX
Total Projects XX
Sources: Press Releases, Annual Reports, SEC Filings, Investor Presentations, Other Government Sources, Analysis Based on Inputs from Secondary, Expert Interviews
FIGURE 1 TOP ENTITIES BASED ON R&D GLANCE FOR GLOBAL BIOLOGICS MARKET
Sources: Press Releases, Annual Reports, SEC Filings, Investor Presentations, Other Government Sources, Analysis Based on Inputs from Secondary, Expert Interviews
10 MARKETED DRUG ANALYSIS
10.1 DRUG
10.1.1.1. BRAND NAME
10.1.1.2. GENERICS NAME
10.2 THERAPEUTIC INDIACTION
10.3 PHARACOLOGICAL CLASS OD THE DRUG
10.4 DRUG PRIMARY INDICATION
10.5 MARKET STATUS
10.6 MEDICATION TYPE
10.7 DRUG DOSAGES FORM
10.8 DOSAGES AVAILABILITY
10.9 PACKAGING TYPE
10.1 DRUG ROUTE OF ADMINISTRATION
10.11 DOSING FREQUENCY
10.12 DRUG INSIGHT
10.13 AN OVERVIEW OF THE DRUG DEVELOPMENT ACTIVITIES SUCH AS REGULATORY MILSTONE, SAFETY DATA AND EFFICACY DATA, MARKET EXCLUSIVITY DATA.
10.13.1.1. FORECAST MARKET OUTLOOK
10.13.1.2. CROSS COMPETITION
10.13.1.3. THERAPEUTIC PORTFOLIO
10.13.1.4. CURRENT DEVELOPMENT SCENARIO
11 MARKET ACCESS
11.1 10-YEAR MARKET FORECAST
11.2 CLINICAL TRIAL RECENT UPDATES
11.3 ANNUAL NEW FDA APPROVED DRUGS
11.4 DRUGS MANUFACTURER AND DEALS
11.5 MAJOR DRUG UPTAKE
11.6 CURRENT TREATMENT PRACTICES
11.7 IMPACT OF UPCOMING THERAPY
12 MARKET OVERVIEW
12.1 DRIVERS
12.2 RESTRAINTS
12.3 OPPORTUNITIES
12.4 CHALLENGES
13 GLOBAL BIOLOGICS MARKET, BY TYPE
13.1 OVERVIEW
13.2 TUMOR NECROSIS FACTOR-A (TNF) INHIBITORS
13.2.1.1. BY TYPE
13.2.1.2. ADALIMUMAB
13.2.1.3. CERTOLIZUMAB PEGOL
13.2.1.4. ETANERCEPT
13.2.1.5. INFLIXIMAB
13.2.1.6. GOLIMUMAB
13.2.1.7. OTHERS
13.2.1.8. BY ROUTE OF ADMINISTRATION
13.2.1.9. INJECTION
13.2.1.10. INFUSION
13.2.1.11. OTHERS
13.2.1.12. BY SOURCE MATERIAL
13.2.1.13. MICROBIAL
13.2.1.14. MAMMALIAN
13.2.1.15. OTHERS
13.3 B-CELL INHIBITORS
13.3.1.1. BY TYPE
13.3.1.2. RITUXIMAB
13.3.1.3. OTHERS
13.3.1.4. BY ROUTE OF ADMINISTRATION
13.3.1.5. INJECTION
13.3.1.6. INFUSION
13.3.1.7. OTHERS
13.3.1.8. BY SOURCE MATERIAL
13.3.1.9. MICROBIAL
13.3.1.10. MAMMALIAN
13.3.1.11. OTHERS
13.4 INTERLEUKIN INHIBITORS
13.4.1.1. BY TYPE
13.4.1.2. ANAKINRA
13.4.1.3. CANAKINUMAB
13.4.1.4. RILONACEPT
13.4.1.5. SECUKINUMAB
13.4.1.6. IXEKIZUMAB
13.4.1.7. TOCILIZUMAB
13.4.1.8. SARILUMAB
13.4.1.9. OTHERS
13.4.1.10. BY ROUTE OF ADMINISTRATION
13.4.1.11. INJECTION
13.4.1.12. INFUSION
13.4.1.13. OTHERS
13.4.1.14. BY SOURCE MATERIAL
13.4.1.15. MICROBIAL
13.4.1.16. MAMMALIAN
13.4.1.17. OTHERS
13.5 T-CELL INHIBITORS (ABATACEPT)
13.6 OTHERS
14 GLOBAL BIOLOGICS MARKET, BY DRUG CLASSIFICATION
14.1 OVERVIEW
14.2 BRANDED DRUGS
14.2.1.1. HUMIRA
14.2.1.2. RITUXAN
14.2.1.3. ENBREL
14.2.1.4. HERCEPTIN
14.2.1.5. AVASTIN
14.2.1.6. REMICADE
14.2.1.7. NEULASTA
14.2.1.8. AVONEX
14.2.1.9. LUCENTIS
14.2.1.10. ORENCIA
14.2.1.11. EYLEA
14.2.1.12. SIMULECT
14.2.1.13. OTHERS
14.3 GENERIC DRUGS
15 GLOBAL BIOLOGICS MARKET, BY DRUG CLASS
15.1 OVERVIEW
15.2 MONOCLONAL ANTIBODIES (MABS)
15.2.1.1. ANTI-CANCER MABS
15.2.1.2. IMMUNOLOGICAL MABS
15.2.1.3. ANTI-INFECTIVE MONOCLONAL ANTIBODIES (MABS)
15.2.1.4. CARDIOVASCULAR AND CEREBROVASCULAR MABS
15.2.1.5. NEUROPHARMACOLOGICAL MABS
15.2.1.6. OTHERS MABS
15.3 THERAPEUTIC PROTEINS
15.3.1.1. METABOLIC DISORDERS THERAPEUTIC PROTEINS
15.3.1.2. CANCER THERAPEUTIC PROTEINS
15.3.1.3. CARDIOVASCULAR THERAPEUTIC PROTEINS
15.3.1.4. IMMUNOLOGICAL THERAPEUTIC PROTEINS
15.3.1.5. OTHERS THERAPEUTIC PROTEINS
15.4 VACCINES
15.4.1.1. ANTI-INFECTIVE VACCINES
15.4.1.2. AUTOIMMUNITY VACCINES
15.4.1.3. OTHERS
15.5 CELLULAR BASED BIOLOGICS
15.6 RECOMBINANT INSULIN
15.7 GENE BASED BIOLOGICS
15.8 OTHER PRODUCTS
16 GLOBAL BIOLOGICS MARKET, BY ROUTE OF ADMINISTRATION
16.1 OVERVIEW
16.2 INJECTION
16.3 INFUSION
16.4 OTHERS
17 GLOBAL BIOLOGICS MARKET, BY APPLICATION
17.1 OVERVIEW
17.2 ONCOLOGY
17.2.1.1. BREAST CANCER
17.2.1.2. LEUKEMIA
17.2.1.3. OVARIAN CANCER
17.2.1.4. NON-HODGKIN LYMPHOMA
17.2.1.5. PROSTATE CANCER
17.2.1.6. COLORECTAL CANCER
17.2.1.7. OTHERS
17.2.1.8. BLADDER CANCER
17.2.1.9. LUNG CANCER
17.2.1.10. OTHERS
17.3 AUTOIMMUNE DISEASES
17.3.1.1. SYSTEMIC SCLEROSIS
17.3.1.2. CROHN’S DISEASE
17.3.1.3. RHEUMATOID ARTHRITIS
17.3.1.4. SYSTEMIC LUPUS ERYTHEMATOUS
17.3.1.5. OTHERS
17.3.1.6. SJOGREN'S SYNDROME
17.3.1.7. MULTIPLE SCLEROSIS
17.3.1.8. PERNICIOUS ANEMIA
17.3.1.9. OTHERS
17.4 DIABETES
17.5 INFECTIOUS DISEASES
17.6 CARDIOVASCULAR DISEASES
17.7 OPHTHALMIC CONDITIONS
17.8 DERMATOLOGICAL DISEASES
17.9 OTHERS
18 GLOBAL BIOLOGICS MARKET, BY SOURCE MATERIAL
18.1 OVERVIEW
18.2 MICROBIAL
18.3 MAMMALIAN
18.4 OTHERS
19 GLOBAL BIOLOGICS MARKET, BY END USER
19.1 OVERVIEW
19.2 HOSPITALS
19.2.1.1. ACUTE CARE HOSPITALS
19.2.1.2. LONG-TERM CARE HOSPITALS
19.2.1.3. NURSING FACILITIES
19.2.1.4. REHABILITATION CENTERS
19.3 SPECIALTY CLINICS
19.4 AMBULATORY SURGICAL CENTERS
19.5 OTHERS
20 GLOBAL BIOLOGICS MARKET, BY DISTRIBUTION CHANNEL
20.1 OVERVIEW
20.2 DIRECT TENDER
20.3 RETAIL SALES
20.3.1.1. HOSPITAL PHARMACIES
20.3.1.2. RETAIL PHARMACIES
20.3.1.3. ONLINE PHARMACIES
20.4 OTHERS (IF ANY)
21 GLOBAL BIOLOGICS MARKET, SWOT AND DBMR ANALYSIS
22 GLOBAL BIOLOGICS MARKET, COMPANY LANDSCAPE
22.1 COMPANY SHARE ANALYSIS: GLOBAL
22.2 COMPANY SHARE ANALYSIS: NORTH AMERICA
22.3 COMPANY SHARE ANALYSIS: EUROPE
22.4 COMPANY SHARE ANALYSIS: ASIA-PACIFIC
22.5 MERGERS & ACQUISITIONS
22.6 NEW PRODUCT DEVELOPMENT & APPROVALS
22.7 EXPANSIONS
22.8 REGULATORY CHANGES
22.9 PARTNERSHIP AND OTHER STRATEGIC DEVELOPMENTS
23 GLOBAL BIOLOGICS MARKET, BY REGION
23.1 GLOBAL BIOLOGICS MARKET, (ALL SEGMENTATION PROVIDED ABOVE IS REPRESENTED IN THIS CHAPTER BY COUNTRY)
23.2 NORTH AMERICA
23.2.1.1. U.S.
23.2.1.2. CANADA
23.2.1.3. MEXICO
23.3 EUROPE
23.3.1.1. GERMANY
23.3.1.2. U.K.
23.3.1.3. ITALY
23.3.1.4. FRANCE
23.3.1.5. SPAIN
23.3.1.6. RUSSIA
23.3.1.7. SWITZERLAND
23.3.1.8. TURKEY
23.3.1.9. BELGIUM
23.3.1.10. NETHERLANDS
23.3.1.11. DENMARK
23.3.1.12. SWEDEN
23.3.1.13. POLAND
23.3.1.14. NORWAY
23.3.1.15. FINLAND
23.3.1.16. REST OF EUROPE
23.4 ASIA-PACIFIC
23.4.1.1. JAPAN
23.4.1.2. CHINA
23.4.1.3. SOUTH KOREA
23.4.1.4. INDIA
23.4.1.5. SINGAPORE
23.4.1.6. THAILAND
23.4.1.7. INDONESIA
23.4.1.8. MALAYSIA
23.4.1.9. PHILIPPINES
23.4.1.10. AUSTRALIA
23.4.1.11. NEW ZEALAND
23.4.1.12. VIETNAM
23.4.1.13. TAIWAN
23.4.1.14. REST OF ASIA-PACIFIC
23.5 SOUTH AMERICA
23.5.1.1. BRAZIL
23.5.1.2. ARGENTINA
23.5.1.3. REST OF SOUTH AMERICA
23.6 MIDDLE EAST AND AFRICA
23.6.1.1. SOUTH AFRICA
23.6.1.2. EGYPT
23.6.1.3. BAHRAIN
23.6.1.4. UNITED ARAB EMIRATES
23.6.1.5. KUWAIT
23.6.1.6. OMAN
23.6.1.7. QATAR
23.6.1.8. SAUDI ARABIA
23.6.1.9. REST OF MEA
23.7 KEY PRIMARY INSIGHTS: BY MAJOR COUNTRIES
24 GLOBAL BIOLOGICS MARKET, COMPANY PROFILE
24.1 BRISTOL-MYERS SQUIBB COMPANY
24.1.1.1. COMPANY OVERVIEW
24.1.1.2. REVENUE ANALYSIS
24.1.1.3. GEOGRAPHIC PRESENCE
24.1.1.4. PRODUCT PORTFOLIO
24.1.1.5. RECENT DEVELOPMENTS
24.2 ABBVIE
24.2.1.1. COMPANY OVERVIEW
24.2.1.2. REVENUE ANALYSIS
24.2.1.3. GEOGRAPHIC PRESENCE
24.2.1.4. PRODUCT PORTFOLIO
24.2.1.5. RECENT DEVELOPMENTS
24.3 AMGEN INC.
24.3.1.1. COMPANY OVERVIEW
24.3.1.2. REVENUE ANALYSIS
24.3.1.3. GEOGRAPHIC PRESENCE
24.3.1.4. PRODUCT PORTFOLIO
24.3.1.5. RECENT DEVELOPMENTS
24.4 GENENTECH USA, INC. (PARENT COMPANY F. HOFFMANN-LA ROCHE AG)
24.4.1.1. COMPANY OVERVIEW
24.4.1.2. REVENUE ANALYSIS
24.4.1.3. GEOGRAPHIC PRESENCE
24.4.1.4. PRODUCT PORTFOLIO
24.4.1.5. RECENT DEVELOPMENTS
24.5 BAYER AG
24.5.1.1. COMPANY OVERVIEW
24.5.1.2. REVENUE ANALYSIS
24.5.1.3. GEOGRAPHIC PRESENCE
24.5.1.4. PRODUCT PORTFOLIO
24.5.1.5. RECENT DEVELOPMENTS
24.6 SANOFI
24.6.1.1. COMPANY OVERVIEW
24.6.1.2. REVENUE ANALYSIS
24.6.1.3. GEOGRAPHIC PRESENCE
24.6.1.4. PRODUCT PORTFOLIO
24.6.1.5. RECENT DEVELOPMENTS
24.7 BIOGEN
24.7.1.1. COMPANY OVERVIEW
24.7.1.2. REVENUE ANALYSIS
24.7.1.3. GEOGRAPHIC PRESENCE
24.7.1.4. PRODUCT PORTFOLIO
24.7.1.5. RECENT DEVELOPMENTS
24.8 CLINIGEN, INC.
24.8.1.1. COMPANY OVERVIEW
24.8.1.2. REVENUE ANALYSIS
24.8.1.3. GEOGRAPHIC PRESENCE
24.8.1.4. PRODUCT PORTFOLIO
24.8.1.5. RECENT DEVELOPMENTS
24.9 NOVARTIS AG
24.9.1.1. COMPANY OVERVIEW
24.9.1.2. REVENUE ANALYSIS
24.9.1.3. GEOGRAPHIC PRESENCE
24.9.1.4. PRODUCT PORTFOLIO
24.9.1.5. RECENT DEVELOPMENTS
24.1 GSK
24.10.1.1. COMPANY OVERVIEW
24.10.1.2. REVENUE ANALYSIS
24.10.1.3. GEOGRAPHIC PRESENCE
24.10.1.4. PRODUCT PORTFOLIO
24.10.1.5. RECENT DEVELOPMENTS
24.11 SEAGEN INC.
24.11.1.1. COMPANY OVERVIEW
24.11.1.2. REVENUE ANALYSIS
24.11.1.3. GEOGRAPHIC PRESENCE
24.11.1.4. PRODUCT PORTFOLIO
24.11.1.5. RECENT DEVELOPMENTS
24.12 LILLY
24.12.1.1. COMPANY OVERVIEW
24.12.1.2. REVENUE ANALYSIS
24.12.1.3. GEOGRAPHIC PRESENCE
24.12.1.4. PRODUCT PORTFOLIO
24.12.1.5. RECENT DEVELOPMENTS
24.13 JANSSEN BIOTECH, INC. (SUBSIDIARY OF JOHNSON & JOHNSON)
24.13.1.1. COMPANY OVERVIEW
24.13.1.2. REVENUE ANALYSIS
24.13.1.3. GEOGRAPHIC PRESENCE
24.13.1.4. PRODUCT PORTFOLIO
24.13.1.5. RECENT DEVELOPMENTS
24.14 TEVA PHARMACEUTICAL INDUSTRIES LTD.
24.14.1.1. COMPANY OVERVIEW
24.14.1.2. REVENUE ANALYSIS
24.14.1.3. GEOGRAPHIC PRESENCE
24.14.1.4. PRODUCT PORTFOLIO
24.14.1.5. RECENT DEVELOPMENTS
24.15 TAKEDA PHARMACEUTICAL COMPANY LIMITED.
24.15.1.1. COMPANY OVERVIEW
24.15.1.2. REVENUE ANALYSIS
24.15.1.3. GEOGRAPHIC PRESENCE
24.15.1.4. PRODUCT PORTFOLIO
24.15.1.5. RECENT DEVELOPMENTS
24.16 UNITED THERAPEUTICS CORPORATION.
24.16.1.1. COMPANY OVERVIEW
24.16.1.2. REVENUE ANALYSIS
24.16.1.3. GEOGRAPHIC PRESENCE
24.16.1.4. PRODUCT PORTFOLIO
24.16.1.5. RECENT DEVELOPMENTS
24.17 MERCK & CO., INC.
24.17.1.1. COMPANY OVERVIEW
24.17.1.2. REVENUE ANALYSIS
24.17.1.3. GEOGRAPHIC PRESENCE
24.17.1.4. PRODUCT PORTFOLIO
24.17.1.5. RECENT DEVELOPMENTS
24.18 ASTRAZENECA
24.18.1.1. COMPANY OVERVIEW
24.18.1.2. REVENUE ANALYSIS
24.18.1.3. GEOGRAPHIC PRESENCE
24.18.1.4. PRODUCT PORTFOLIO
24.18.1.5. RECENT DEVELOPMENTS
24.19 CELLTRION HEALTHCARE CO.,LTD.
24.19.1.1. COMPANY OVERVIEW
24.19.1.2. REVENUE ANALYSIS
24.19.1.3. GEOGRAPHIC PRESENCE
24.19.1.4. PRODUCT PORTFOLIO
24.19.1.5. RECENT DEVELOPMENTS
24.2 SUN PHARMACEUTICAL INDUSTRIES, INC.
24.20.1.1. COMPANY OVERVIEW
24.20.1.2. REVENUE ANALYSIS
24.20.1.3. GEOGRAPHIC PRESENCE
24.20.1.4. PRODUCT PORTFOLIO
24.20.1.5. RECENT DEVELOPMENTS
24.21 HIKMA PHARMACEUTICALS PLC
24.21.1.1. COMPANY OVERVIEW
24.21.1.2. REVENUE ANALYSIS
24.21.1.3. GEOGRAPHIC PRESENCE
24.21.1.4. PRODUCT PORTFOLIO
24.21.1.5. RECENT DEVELOPMENTS
24.22 LEO PHARMA A/S
24.22.1.1. COMPANY OVERVIEW
24.22.1.2. REVENUE ANALYSIS
24.22.1.3. GEOGRAPHIC PRESENCE
24.22.1.4. PRODUCT PORTFOLIO
24.22.1.5. RECENT DEVELOPMENTS
24.23 BOEHRINGER INGELHEIM INTERNATIONAL GMBH.
24.23.1.1. COMPANY OVERVIEW
24.23.1.2. REVENUE ANALYSIS
24.23.1.3. GEOGRAPHIC PRESENCE
24.23.1.4. PRODUCT PORTFOLIO
24.23.1.5. RECENT DEVELOPMENTS
24.24 BIOCON BIOLOGICS LIMITED (A SUBSIDIARY OF BIOCON LIMITED)
24.24.1.1. COMPANY OVERVIEW
24.24.1.2. REVENUE ANALYSIS
24.24.1.3. GEOGRAPHIC PRESENCE
24.24.1.4. PRODUCT PORTFOLIO
24.24.1.5. RECENT DEVELOPMENTS
25 RELATED REPORTS
26 CONCLUSION
27 QUESTIONNAIRE
28 ABOUT DATA BRIDGE MARKET RESEARCH

Research Methodology
Data collection and base year analysis are done using data collection modules with large sample sizes. The stage includes obtaining market information or related data through various sources and strategies. It includes examining and planning all the data acquired from the past in advance. It likewise envelops the examination of information inconsistencies seen across different information sources. The market data is analysed and estimated using market statistical and coherent models. Also, market share analysis and key trend analysis are the major success factors in the market report. To know more, please request an analyst call or drop down your inquiry.
The key research methodology used by DBMR research team is data triangulation which involves data mining, analysis of the impact of data variables on the market and primary (industry expert) validation. Data models include Vendor Positioning Grid, Market Time Line Analysis, Market Overview and Guide, Company Positioning Grid, Patent Analysis, Pricing Analysis, Company Market Share Analysis, Standards of Measurement, Global versus Regional and Vendor Share Analysis. To know more about the research methodology, drop in an inquiry to speak to our industry experts.
Customization Available
Data Bridge Market Research is a leader in advanced formative research. We take pride in servicing our existing and new customers with data and analysis that match and suits their goal. The report can be customized to include price trend analysis of target brands understanding the market for additional countries (ask for the list of countries), clinical trial results data, literature review, refurbished market and product base analysis. Market analysis of target competitors can be analyzed from technology-based analysis to market portfolio strategies. We can add as many competitors that you require data about in the format and data style you are looking for. Our team of analysts can also provide you data in crude raw excel files pivot tables (Fact book) or can assist you in creating presentations from the data sets available in the report.