
The outbreak of Coronavirus highlights the need of effective treatment option in order to get rid of such pandemic. Various types of drugs have been validated for COVID-19 treatment among which biosimilar drugs are getting more attention.
The biosimilar drug market is growing even during the COVID-19 pandemic and this is due to the increasing demand of emergency used drugs worldwide to treat COVID-19 patients. The sales of biosimilar drugs are increasing due to increasing demand of biosimilar drugs for treatment of rheumatoid arthritis, Crohn’s disease, among others.
TABLE 1. SALES DATA OF BIOSIMILAR DRUG PROJECTED FOR 2019/2024
|
COMPANY NAME |
BIOSIMILAR |
U.S. SALES IN 2019/24 |
|
Celltrion/Pfizer |
Inflectra |
300/430 |
|
Mylan/Biocon |
Fulphila |
224/255 |
|
Cohreus Biosciences |
Udencya |
356/498 |
|
Pfizer |
Retacrit |
141/101 |
|
Amgen/Allergan |
Mvasi |
121/305 |
Biosimilar drug market is also growing due to increasing number of clinical trials in order to demonstrate the safety and efficacy of these drugs.
The demand of effective treatment for various kinds of diseases such as rheumatoid arthritis, diabetes among others is increasing during COVID-19 pandemic due to fear among people regarding COVID-19 infection.
The price of biosimilar drugs has increased during the COVID-19 pandemic as no any generic competitive drug is still available, along with increasing demand of safe and effective drug.
For instance,
This thus signifies that due to COVID-19 pandemic the price of biosimilar used for treatment of inflammatory symptoms among others caused by Coronavirus increased.
The Coronavirus led to the widespread closure of local manufacturing plants and companies and has badly affected the various region of world. Due to extensive lockdown and isolation the economic activity has affected adversely which has impacted the global economic activity.
COVID-19 is expected to be an opportunity for biosimilar market as biosimilar drugs have been proved to effective against various kinds of diseases such as autoimmune disorders, blood disorders, and infectious diseases, among others.
For instance,
FIGURE 1. APPROXIMATE NUMBER OF BREAST CANCER CASES IN 2018, INDIA, FEMALES, ALL AGES
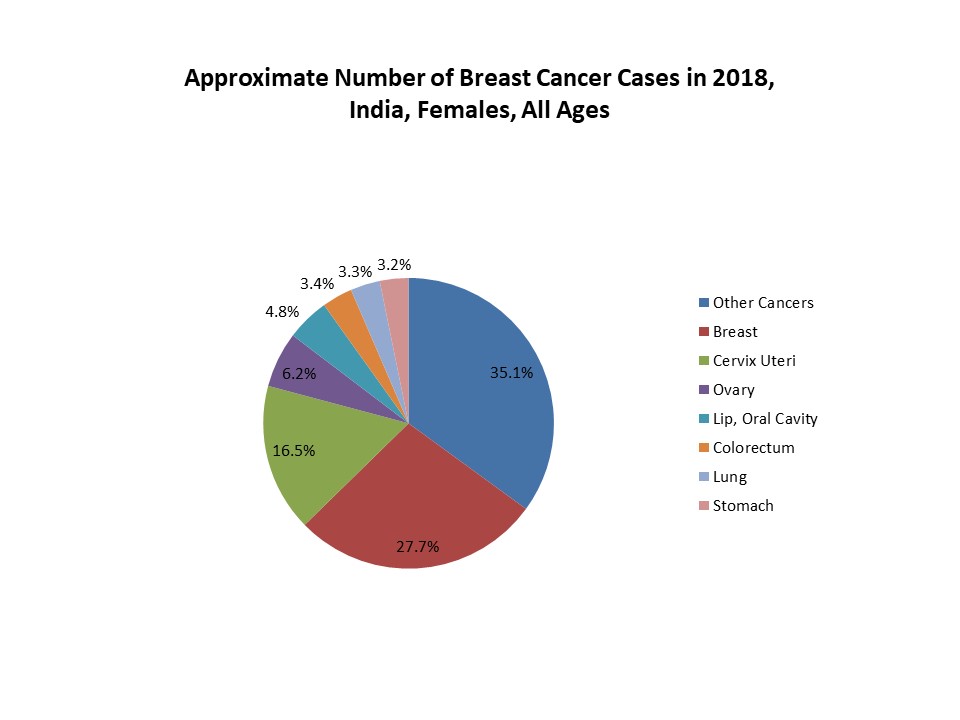 FIGURE 2. PERCENTAGE OF NEW CANCER CASES AMONG WOMEN IN 2020, CANADA
FIGURE 2. PERCENTAGE OF NEW CANCER CASES AMONG WOMEN IN 2020, CANADA
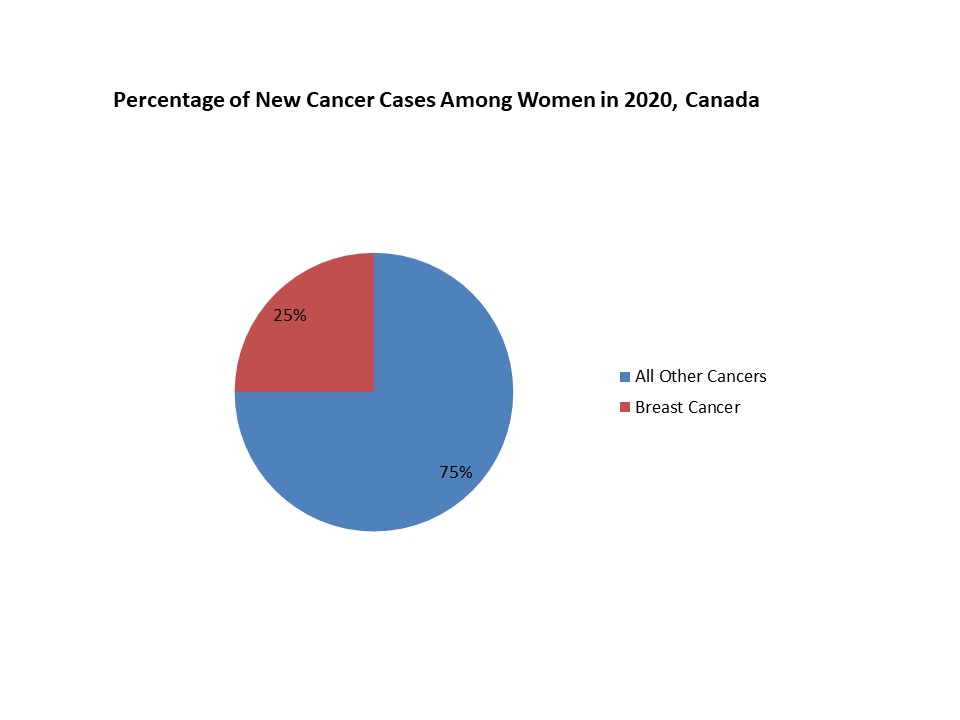 Above figure suggests that the number of breast cancer cases is rising among women all over the world which enhanced the demand of effective biosimilar treatment option, thus, increasing number of breast cancer cases led to increased demand of biosimilar drugs.
Above figure suggests that the number of breast cancer cases is rising among women all over the world which enhanced the demand of effective biosimilar treatment option, thus, increasing number of breast cancer cases led to increased demand of biosimilar drugs.
Moreover the increasing cases of COVID-19 have also paved the way for enhanced demand of biosimilars.
For instance,
This thus signifies that increasing prevalence of COVID-19 poses a life threatening effect to various types of patients already suffering from several kinds of disease among the most recent known COVID-19 pandemic.
Thus the demand of effective treatment option that is biosimilar drug has enhanced as have been proved to block IL-6 receptor which in turn reduces inflammatory affects. This thus demonstrates that COVID-19 is accelerating the demand of biosimilar market.
As COVID-19 spreading began in China the surge of medicines has been enhanced due to increasing patient’s volume which put a pressure over the supply of medicaments. Moreover decreased capacity of air freight carriers due to continuous travel ban across the globe has also led to shortage of drugs supply in the U.S., among other countries across the world.
However the market players are adopting several initiatives in order to maintain a continuous supply of biosimilar drugs.
For instance,
This suggests that increasing prevalence of COVID-19 poses threat for maintaining a continuous supply chain of biosimilar drug while initiative adopted by market players allows them to manage a continuous supply chain.
Collaboration, agreements, strategic initiatives by market players such as Pfizer, Inc., Sandoz, Boehringer Ingelheim, Amega Biotech, mAbxience among others in the biosimilar market will help them to expand their product portfolio. This in turn will lead to increasing product sales and hence will enhance the overall company’s revenue.
Biosimilar manufacturing companies are taking so many strategic decisions in order to cope up with the current scenario of COVID-19 pandemic. The companies engaged in manufacturing of biosimilar are collaborating so as to accelerate the market growth.
Companies have already taken several kinds of strategic initiatives in order to cope up with the corona virus situation.
For instance,
The increasing demand and increasing sales of biosimilar drugs are fueling the growth of biosimilar market. Thus, companies operating in the biosimilar market are adopting several strategies, including collaboration, agreements, market expansion to enhance their business. These strategic decisions by the market players helped them to attain a lucrative growth even during the COVID-19 pandemic.
As the pandemic of COVID-19 has resulted in several restrictions throughout the borders but still manufacturers of biosimilar are able to manage their stocks. Various manufacturers are continuously engage in conducting clinical trials for demonstrating the safety and efficacy of biosimilar along with this they are constantly monitoring the supply chain so as to attain a lucrative growth. Moreover, by raising price of biosimilar the companies are gaining extra profit which is helping them to combat the negative effect on overall revenue.