
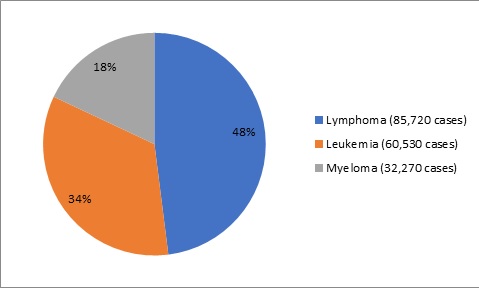
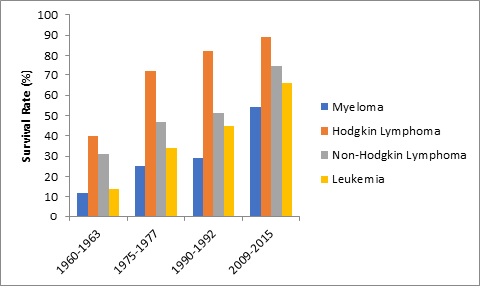
The COVID-19 pandemic has impacted different sectors of the pharmaceutical industry at distinct levels. The lockdowns imposed by the governments across the world as well the need to maintain social distancing has deeply impacted the manufacturing and distribution intensive industries including the pharmaceutical industry.
Cancer researchers and oncologists should be curious or terrified because patients are suffering from any type of cancer are at a higher risk for bad outcomes. This is because of their immunity is very low due to taking cancer treatment and chemotherapy.
The impact of the COVID-19 outbreak on the blood cancer drug testing is varying depending on the number of COVID-19 cases & cancer cases in the area. Areas with fewer cases of COVID-19 are striving to continue cancer clinical trials and hence, the blood cancer drug market may not be greatly affected in these areas. In these places, new patients are still joining trials and patients are still getting treatment.
The overall demand will be modestly impacted across the blood cancer drug market in the short-term. Hospitals and oncology centers across the country have suffered economic downsides from the elimination of non-emergency or non-urgent care. As the rate of recovery of COVID-19 cases is increasing, clinical trial institutes are planning to restart regular enrolment.
The role of blood cancer drug is pivotal in cancer treatment areas that experience significantly high demand and receive the necessary funding. The position of blood cancer drug is essential which is leading to almost unchanged demand during the pandemic. Guidelines are available stating how clinicians & researchers should proceed for processes in cancer clinical trials during the COVID-19 pandemic. Moreover, the patients will only be treated by the physicians and nurses on the specific study teams to reduce the risk of infection. However, this might lead to patients travelling long distances several times a month for routine consultations. This reduces the potential number of patients to those who can afford frequent trips to the site or paid stay near the site. The guidelines also include other considerations such as implementing COVID-19 screening procedures implementing COVID-19 screening procedures, delaying recruitment, expediting changes in trial protocol amongst others.
However, the aforementioned nonbinding guidelines have led to every organization developing its own priorities as most of the staff is focused on handling the COVID-19 crisis. On the other hand, several organizations believed that being on a clinical trial is the most important recommendation for treatment, and they want to continue conducting clinical trials for a number of reasons. This is because clinical trials are no longer the last-ditch effort but are considered to be the best chances for patients to get cured as they have specific enrolment criteria to match the specific type of cancer and its treatment that has been designed for that defect or malignancy. Hence, they consider clinical trial to be the best option especially in oncology cancer, and they want to continue as long as it’s safe and feasible.
As the patients are afraid for getting infected with the COVID-19 infection ongoing care treatment also affected. For instance; primary care providers who offer or use face to face guidance or appointments has been reduced, with switching daily services on telecommunication/tele- consultations unless an in-person consultation is deemed essential. Moreover, the psychological stress in patients receiving a diagnosis and treatment for cancer will be increased in current situation because patients may struggle to access their usual support networks, and the provision of psychological support through healthcare systems may be reduced.
Apart from the treatment, diagnosis and ongoing care implications coronavirus has effect on various clinical trials. Due to the coronavirus pandemic many pharmaceutical industries have suspended the clinical trials. In a study it is estimated that approximately over 12,000 clinical trails are going on in the area of cancer. If these trails are stopped or suspended for any reason, it will have profound impact on the industry. Beyond this, loss of the potential health benefits for patients already enrolled in the trials, the cost of an oncology phase 3 clinical trial is on average at least USD 20 million, which will impose huge financial hurdle on companies for gaining approval for trail and identification for the patients for the second time.
 IMPACT OF COVID-19 ON SUPPLY CHAIN OF BLOOD CANCER DRUG
IMPACT OF COVID-19 ON SUPPLY CHAIN OF BLOOD CANCER DRUGAmong the problems for pharmaceutical supply chains during this pandemic are the export-import restrictions and impact of COVID-19 on two of the largest global producers of active pharmaceutical ingredients (APsssIs) and generics: China and India. In February 2020, Food and Drug Administration (FDA) announced the supply chain disruption of essential medical products services import from China. The sudden outbreak of COVID-19 and its subsequent impact on restrictions on trade and movement of goods has resulted in a the closing of vast portions of the global economy, which has eventually resulted in disrupted supply chains due to limited materials and workforce, and slowdown or stopping of manufacturing. Moreover, several manufacturing facilities are focused on priority manufacturing to produce other essential medical supplies and equipment such as ventilators in the beginning of the emergency.
The trade restrictions have impacted the market inversely. During the pandemic, as the demand increases, counterfeiting and price gouging of imported devices increase as well.
As the pandemic increases cancer patients are facing major problems in treatment, diagnosis and medication. Surveys show that at least 50% of the 49 new oncology drugs launched between 2010 and 2014 are only accessible to patients in only a few countries including the United States, France, Germany, Canada, Italy and the United Kingdom. Most countries find the cost of these drugs significantly higher and this has led to limited adoption. Hence, increased drug costs are attributed to the demand for alternative oncology treatments and continued usage of older drugs.
The availability of targeted treatments, including biologics, small molecules, and immunotherapies are more likely to be offered in North America, Japan and Western Europe as compared to Eastern European countries. In addition to the issues of availability due to high cost, lack of favourable reimbursed scenario across the world is also impacting the market. To improve the availability and control the costs of cancer drugs, governments have made strict rules for the distributors, suppliers as well as pharmaceutical industries. The adverse impact of COVID-19 on movement and distribution of drugs is expected to impact the price due to lack of availability.
As the patients are afraid of getting infected and transportation facilities are drastically disrupted the demand of cancer drugs are affected by the coronavirus pandemic. Due to COVID-19, social distancing increases and a smaller number of patients visit the cancer centres which delays in elective surgeries. As a result, the demand for online delivery of drugs is increasing.
During the pandemic, pharmaceutical companies are utilizing the technology including digital media such as telecommunication, mobile applications, webinars and others to meet the patient requirement. One such initiative is EDetailing as it improves the co-ordination among healthcare providers and patients and hence improves relationships. E-Detailing is being used by the pharmaceutical companies to communicate their product information and other crucial information to necessary stakeholders. Another similar initiative is Product Information Management (PIM) solutions. A Product Information Management (PIM) enables the automation by providing a portal to offer transparency across the supply chain to all stakeholders including the patients and professionals. As the data is held on one digital platform, the risk of human errors is reduced in reporting and updates. Utilization of existing technology to make the processes efficient is expected to help pharmaceutical companies during the pandemic as well as post pandemic.
The global impact of COVID-19 is unprecedented and has been a game changer for clinicians and patients alike. Healthcare delivery is largely impacted while combating the infection and healthcare IT infrastructure has played a pivotal role in the fight. Several tools such as video visits and wearable devices are gaining tremendous acceptance and adoption where it is not possible for many patients to seek necessary medical consultation & prescription. As a result, cancer treatment market including the blood cancer drugs market is impacted negatively. This includes the availability, supply and demand of the cancer treatment products. The consumption for blood cancer drug is expected to see mild decline for a very short term. However, the long term demand will stay unaltered due to the necessity of these drugs and the push from healthcare organizations to continue conducting cancer treatment during the COVID-19 pandemic. The existing market leaders holding major share in the market have strong strategic teams and will be least impacted. For a brief duration, the supply chain of the drugs will be impacted due to restrictions on movement of goods and equipment. However, as the situation eases, the sales will bounce back. Taking strategic steps will help the companies to exhibit planning and demonstrate stability to the end users.