A novel coronavirus SARS-CoV-2 appeared in December 2019 in Wuhan, China spreading much faster than its predecessors and has already diseased millions of patients worldwide as of April 19th, 2020. As the scope of the current COVID-19 eruption has reached section of pandemics, major international efforts in public health are ongoing to control the eruption. Although, the favorable vaccine discovered to save the patient life but not everyone get the dose so there is considerable interest in repeating existing antiviral drugs for use against COVID-19.
Tocilizumab (TCZ) is a monoclonal antibody against interleukin-6 (IL-6). It may ameliorate the inflammation associated with the severe coronavirus disease and hence improve the symptoms. The monoclonal antibody targets the host immune system to infection which potentially diminishes the inflammation.
Impact on Price
A large number of challenges are being confronted by various market players due to the impending of the unusual coronavirus and different strains, one such issue is the uncertainty surrounding the impact of COVID-19 on tocilizumab demands. Due to the increased demand of life saving drugs such as tocilizumab, remdesivir and favipiravir for coronavirus treatment and stiff rivalry between the market players.
According to the reports in the month of April, as the India face second wave of coronavirus due to a sudden surge in its demand and fresh limited stocks have been imported in India. It looks as the product is present in black market and patients’ families are imposed to buy the products on higher rates to save their loved ones life.
For instance,
The impact of COVID-19 has created huge price impact on the government as well as on patient families that they have to buy the drugs on higher rate as compared to the normal rate.
Impact On Demand
Patient’s families are in panic situation due to the limited stock of the injection and higher price of the drug. The injection being sold on higher price in the black market. In that particular situation, government take over the allocation and supply of injection in the different states of India.
For instance,
The impact of COVID-19 has created an prospect for the number of drugs being clinically tested to cure the diseases or related symptoms.
Impact on Supply
As the endemic intensifies, supply chains can be at substantial risk due to over-located locations that can possibly be disrupted. The supply chain of drugs has been interrupted. The spread of COVID-19 makes it tough for governments to use the drugs, the obtainability of these systems faces constant trials due to their components of use as well as limited initial needs.
Due to the unexpected surge in cases and rising demand of the injection, the supply is disrupted. The Swiss drug maker F. Hoffmann-La Roche AG has responded to the short-supply of its drug Tocilizumab used to treat COVID-19 patients. Moreover F. Hoffmann-La Roche AG pointed out that given the rising number of infection in India, minimizing hospitalizations and easing pressure on healthcare systems was particularly important.
The trade limitations have chosen nothing more than to produce the necessary medications domestically. During the pandemic, when the disease increases, forging and price upsurges for the imported goods. This signifies that even during the COVID-19 pandemic, the market players are able to preserve supply chain.
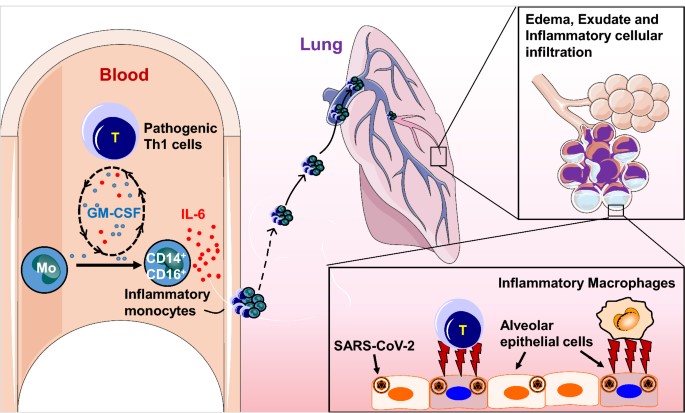
FIGURE 1. EFFECT OF TOCILIZUMAB FOR THE TREATMENT OF SEVERE COVID-19 INFECTION
As the coronavirus remains to spread to various nations, anxieties are growing about disturbances in drug production and distribution. Agreements, collaborations and initiatives of market participants such as F. Hoffmann-La Roche AG and Cipla Inc. expand their business to overcome the growing demand of Tocilizumab especially in Indian and highly affected countries. This will in turn will help to increase the growth for the products among the consumers and thus upsurge the future sales of the company.
With the increasing demand and cumulative sale of Tocilizumab drugs are fueling the growth of tocilizumab medications market in the near future.
As such, market participants are tangled in the manufacture of tocilizumab drugs increasing their business through a variety of programs including contracts, collaboration and pipeline development and market expansion. It is expected that the strategic decisions of these companies will provide significant opportunities for market participants operating in the tocilizumab market.
Conclusion
The available clinical evidence suggests that tocilizumab is relatively safe with minimum side effects. Moreover, studies showed that when tocilizumab administered in combination of corticosteroids offers the modest mortality benefit in certain patients with COVID-19 who are severely ill. However, some adverse effects have been found related to the use of tocilizumab in patients with COVID-19 such as elevated liver enzyme levels and neutropenia or thrombocytopenia which are uncommon. Clinical studies also showed that the effect of tocilizumab in COVID-19 patients, may be prolonged and earlier outcomes have more relevance in view of the acuteness of presentation and rapid progression in the absence of immunomodulation in patients with hyper inflammation and ARDS.
Moreover, ongoing trials are evaluating the use of tocilizumab for the treatment of COVID-19. Government and pharmaceutical market players have also taken strategic initiatives to overcome the pandemic.
TABLE 1. EFFECT OF TOCILIZUMAB ALONE IN COMBINATION WITH CORTICOSTEROIDS AND PERCENTAGES OF EFFECTIVENESS
|
S.No. |
Comment |
Survival Percentage |
Patient Conditions |
|
1. |
Compared with the usual care, tocilizumab reduced deaths by |
4% |
Among patients with COVID-19 who required oxygen and had evidence of inflammation |
|
2. |
Among patients with COVID-19 who receives glucocorticoids in combination with tocilizumab |
79% |
Greater likelihood of quicker improvement |
|
65% |
Less mortality rate |
||
|
71% |
Less invasive mechanical ventilation |